Ionisation energies
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
What is the unit of ionisation energy?
kJ/mol⁻¹
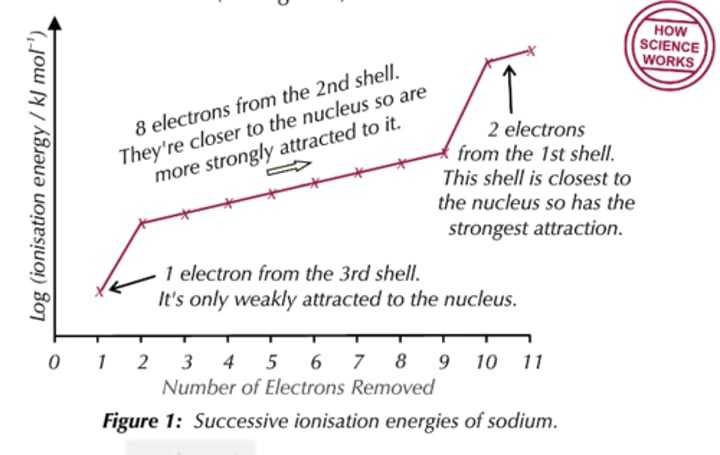
Ionisation energy of sodium
Why does magnesium have a higher ionisation energy than aluminium?
Because of their sub-shell structures
Electron configuration of magnesium
1s²2s²2p⁶3s²
Which orbital does the first ionisation energy remove an electron from?
3s orbital
Electron configuration of aluminium
1s²2s²2p⁶3s²3p¹
Which orbital does the first ionisation energy remove an electron from?
3p orbital
Which orbital is further away from the nucleus: 3s or 3p?
3p
Which orbital is higher in energy: 3s or 3p?
3p
How is the electrostatic attraction between the 3p orbital's electrons and the nucleus different compared to that of the 3s orbital's electrons?
The electrostatic attraction is lower
What does this mean in terms of ionising?
It is easier to ionise an electron in the 3p orbital than in the 3s orbital
What is Hund's rule?
Electrons will fill an unoccupied orbital before they pair up
Why is it easier to remove an electron from a doubly occupied orbital?
Because electrons in the same orbital repel each other
Explanation as to why the first I.E. of sulfur is lower than that of phosphorus

What are ionisation energies?
The energy value associated with removing one electron from the atom
When writing the ionic equation for ionisation energy of oxygen, do you start with O₂ or O?
O because you are dealing with purely atoms
Ionic equation for the ionisation of oxygen
O₍g₎ → O⁺₍g₎ + e⁻
What would be the second ionisation energy of oxygen?
O⁺₍g₎ → O²⁺₍g₎ + e⁻
What does the first ionisation energy mean?
The energy required to remove 1 electron from each atom of 1 mole of gaseous atoms to form 1 mole of gaseous ions
What is the definition of the second ionisation energy?
The energy required to remove 1 electron from each 1⁺ ion of 1 mole of gaseous ions to form 1 mole of gaseous 2⁺ ions
What three factors affect ionisation energy?
Nuclear charge, distance from the nucleus and shielding
What does a greater nuclear charge mean for removing an electron?
More energy is needed to remove the electron
How does an increased nuclear charge affect the ionisation energy?
Increases the I.E
What type of attraction is between electrons and the nucleus?
Electrostatic attraction
How does an increased distance from the nucleus affect the ionisation energy?
Decreases the I.E
What happens to the ionisation energy as the principle quantum number increases?
Decreases the I.E.
How does more shielding between electrons affect the ionisation energy?
Decreases the I.E
How does the first ionisation energy change down a group and why?
I.E. decreases because the atomic radius increases, making the electron easier to remove
What is another reason for the first ionisation energy decreasing down a group?
Going down a group there are more electrons between the nucleus and the outer electrons, making the outer electrons experience greater shielding
Ionisation energy for group 1
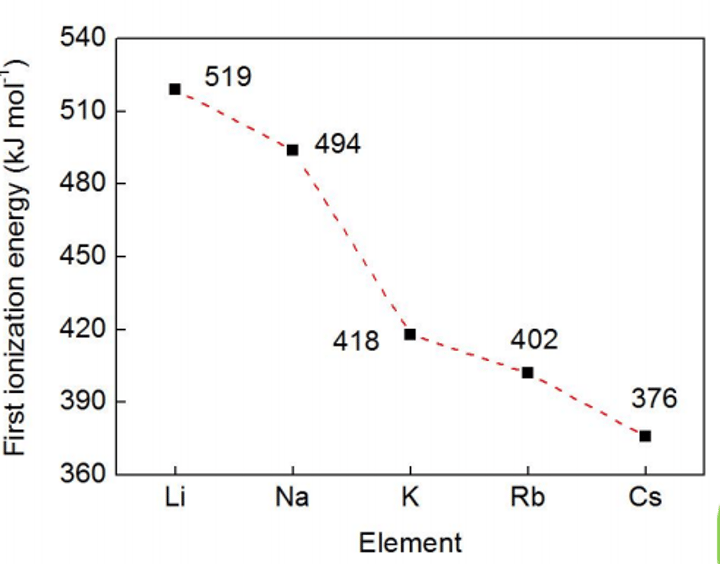
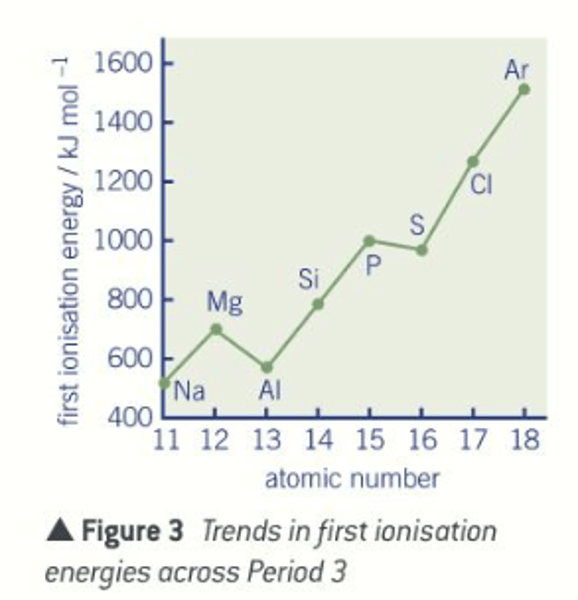
How does the first ionisation energy change across a period and why?
Increases because across a period the atomic radius decreases and the nuclear charge increases
Ionisation energy of period 3