LC and GC-MS - Drug metabolism assessment by mass spectrometry
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
What is the MS characterisation of metabolites?
Metabolite I → M+
Metabolite II → M+
Metabolite III → M+
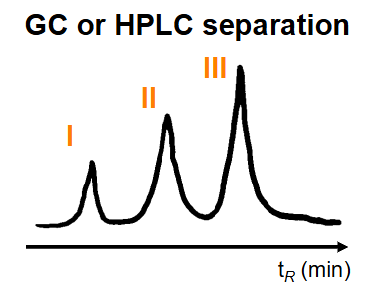
What are the advantages and disadvantages of GLC-MS and HPLC-MS?
Advantages - small sample size ~ 20 µL, mixtures separated, M+ determined separately, can be automated, high throughput
Disadvantages - capital equipment costs, GLC metabolites require chemical derivatization, HPLC requires volatile mobile phase; buffers? NO!!
What does phase I metabolites do?
Produces polar group or nucleophile ready for phase II
Right hand side is more hydrophilic
+ or - is for addition or loss of proton (using atomic mass)
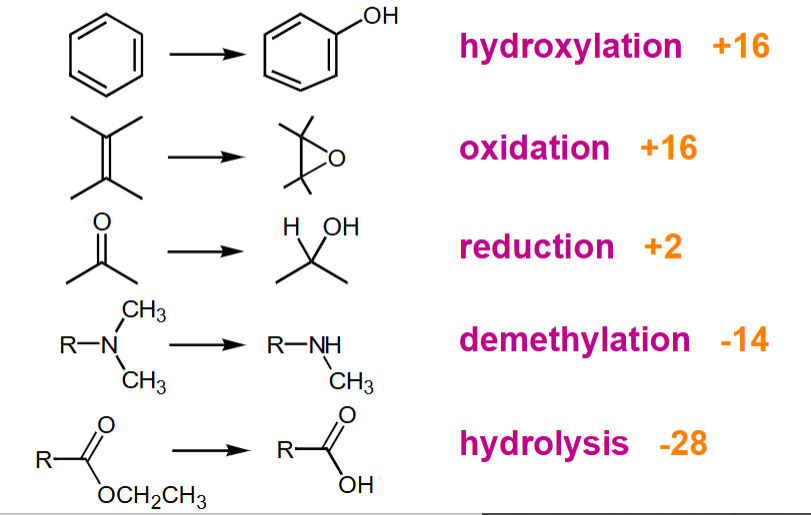
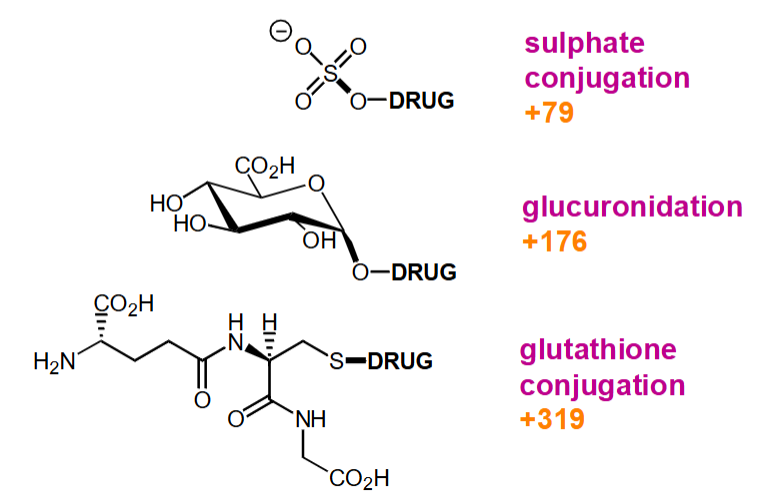
What does phase II metabolites do?
Produces highly polar groups or conjugates for excretion
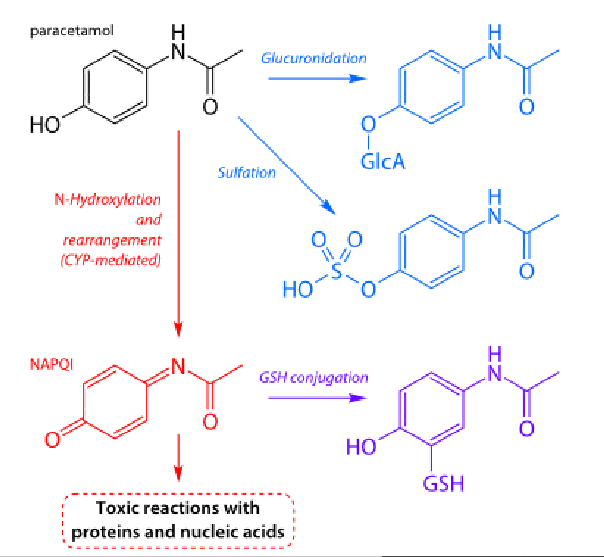
What are the different pathways for paracetamol metabolism?
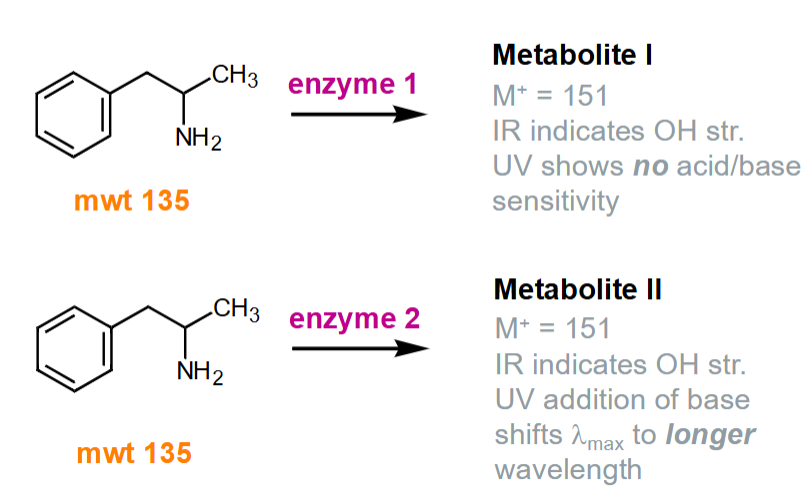
What are the different pathways for amphetamine metabolism?
Phase I - hydroxylation happens in the side chain as there is no acid/base sensitivity
Phase II - hydroxylation happens in the benzene ring as there is acid/base sensitivity
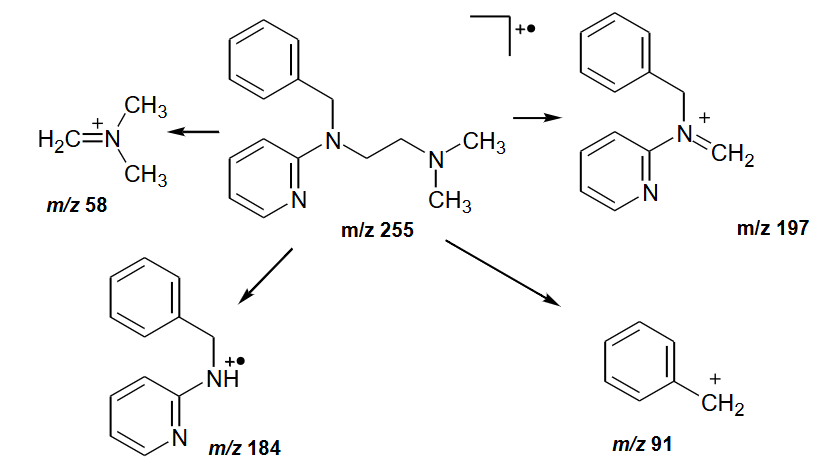
Three metabolites can result on metabolic conversion of tripelenamine, they show the same overall fragmentation pathway as tripelenamine, but some peaks occur at different masses due to metabolism.
Metabolite I shows M+ = 241 and peaks at m/z 197, 184, 91 and 44
Metabolite I shows M+ = 165 and peaks at m/z 107, 94 and 58
Metabolite Ill shows M+ = 271 and peaks at m/z 213, 200, 91 and 58
Select the processes responsible for formation of each drug metabolite
Oxidation at the benzene ring.
Oxidation at the pyridine ring.
Oxidation at both the benzene and pyridine rings.
De-benzylation of the drug.
De-methylation of the drug.
Can be oxidised in the pyridine and benzene rings and +16
Can be de-methylated in the CH3 and -14
Can be de-benzylated in the benzene and -90
Metabolite I = 5
Metabolite II = 4
Metabolite III = B, each piece that has pyridine ring would be 16 bigger