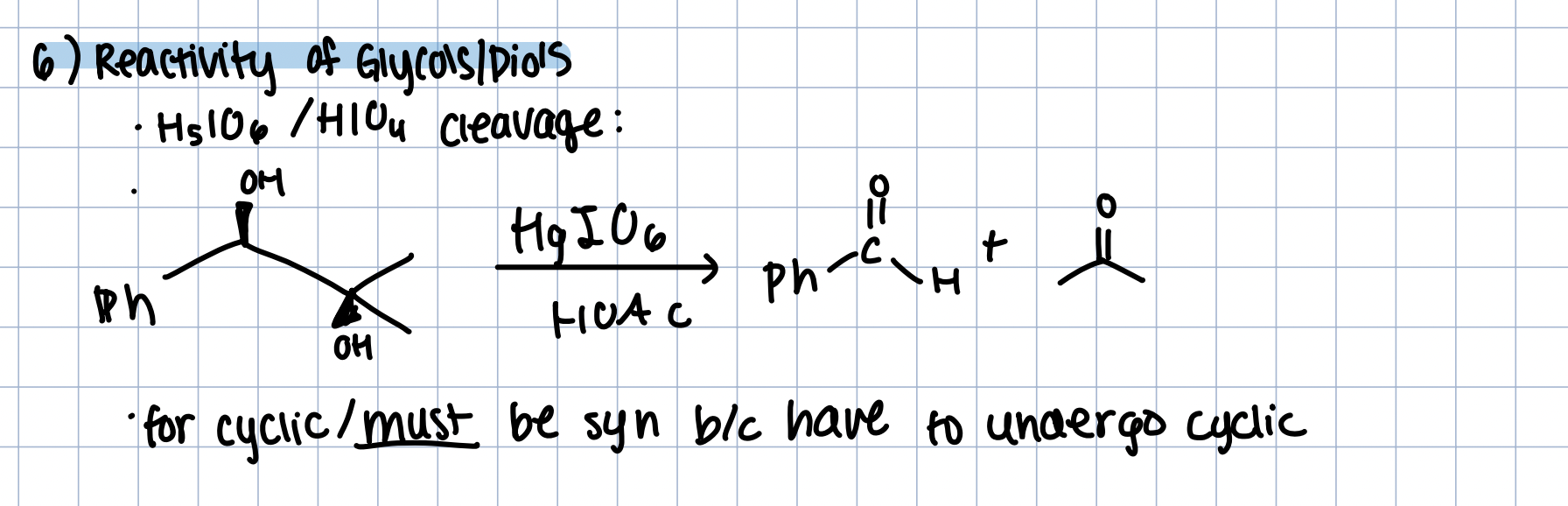CHEM 3570 - Prelim 3, Alcohols, Thiols, Ethers, Epoxides, Glycols
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
thiol
R-SH
more branched alcohol means..
less acidic (less stable conjugate base due to hindrance of solvation)
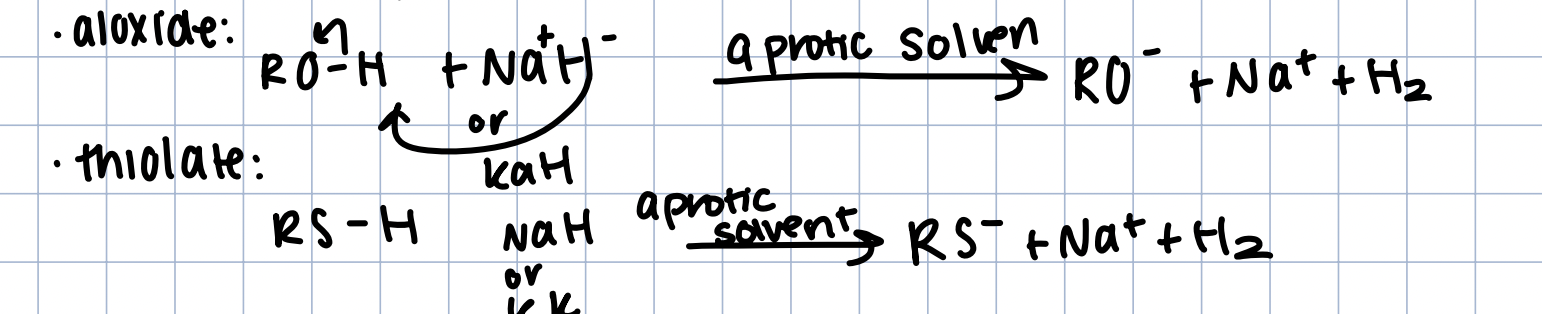
Formation of Thiolate/Aloxides
React with NaH/KH with aprotic solvent
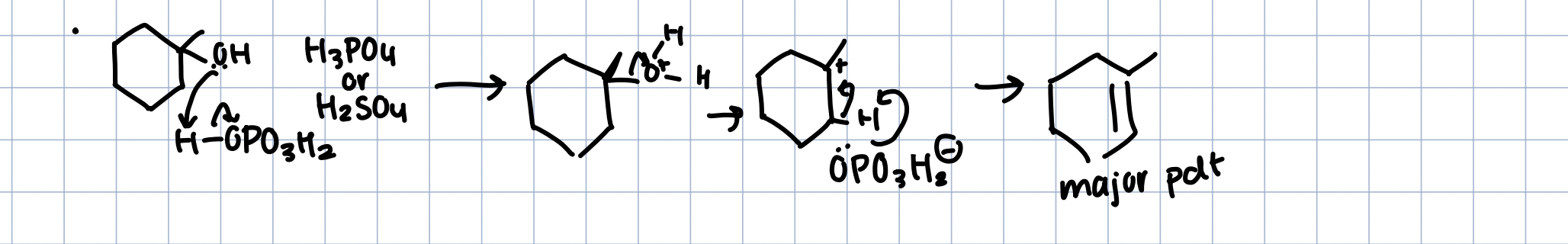
Dehydration/Protonation of Alcohols
Makes a better leaving group
H3PO4 or H2SO4
becomes E1 rxn
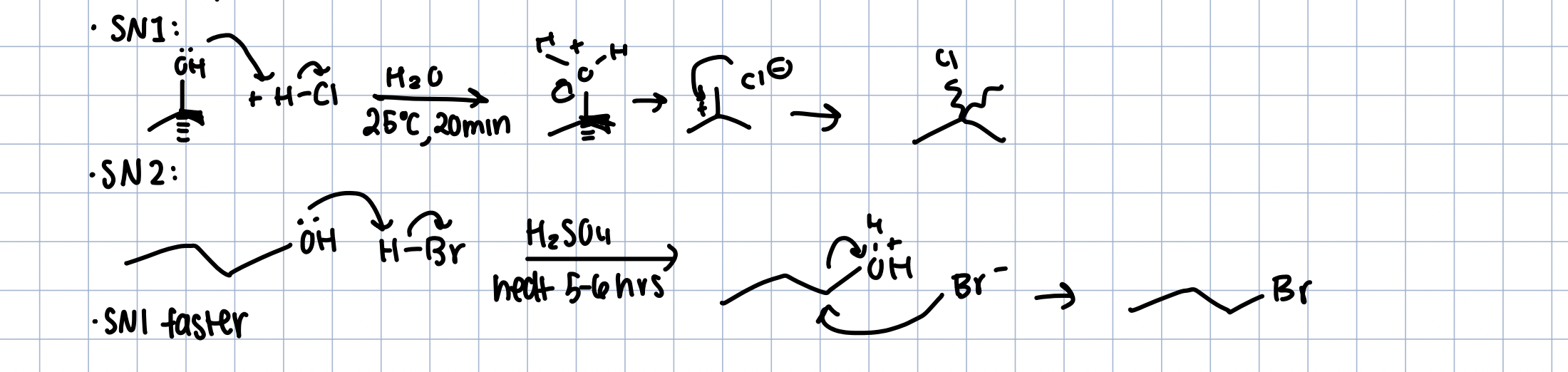
Alcohols with HX
can happen with Sn1 and Sn2
protonate alcohol to good leaving group
sn1 faster
For SN1: H-X, H2O
For Sn2: H-x, Aprotic
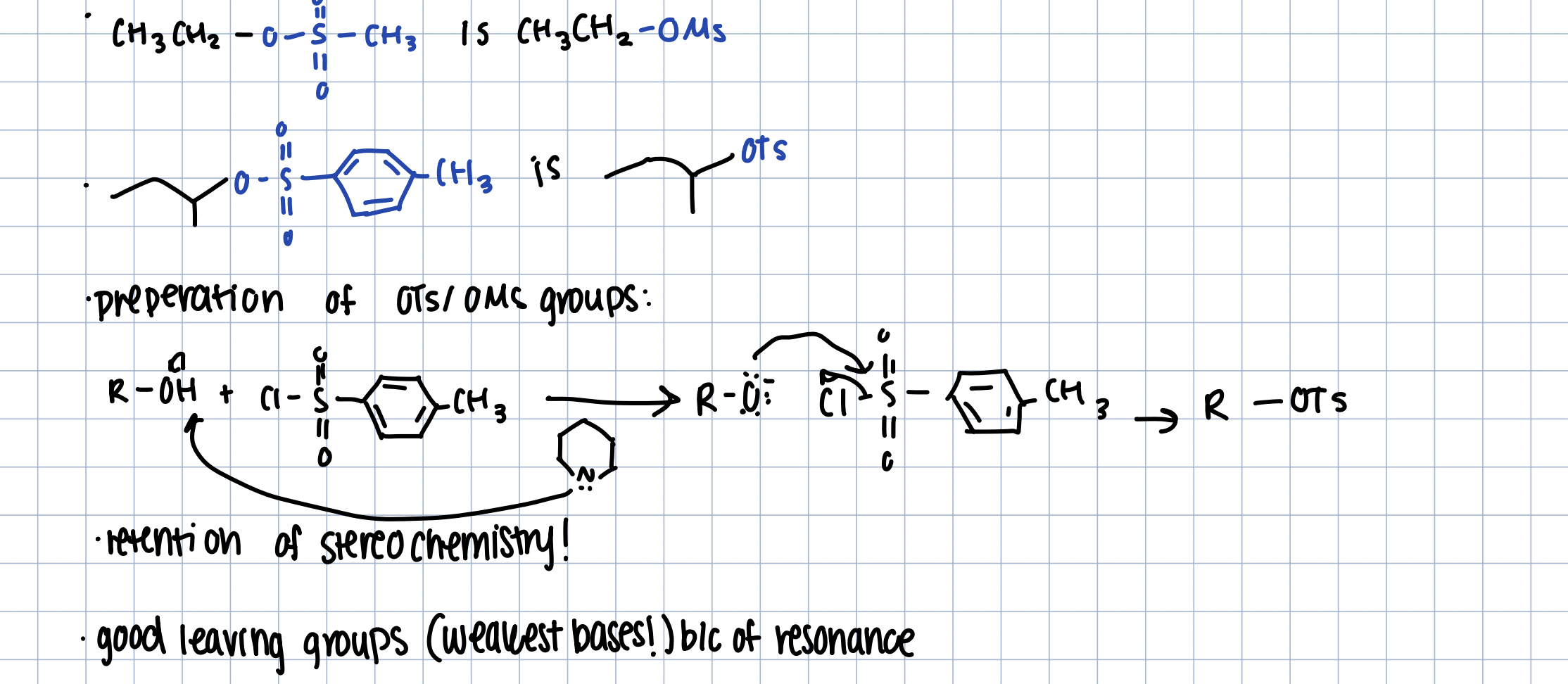
Preparation of OTs/OMs groups
TsCl and Pyridine
retention of stereochemistry
they are good leaving groups/weak bases
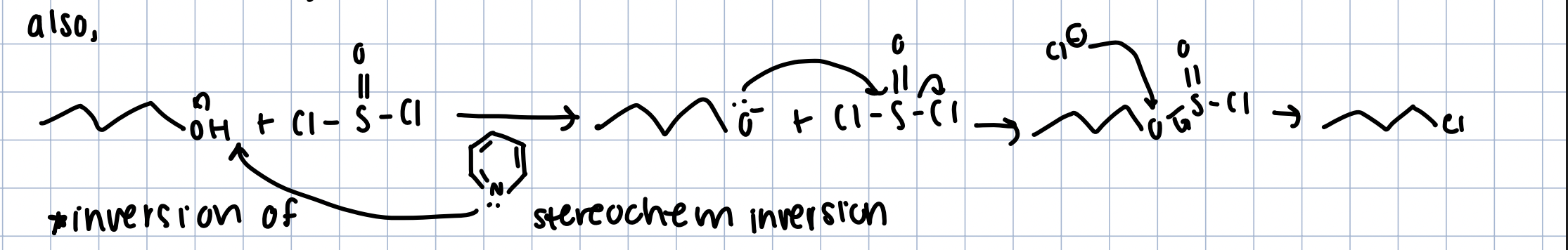
Alcohols - Chlorides (inversion)
SOCl2 in pyridine
like SN2
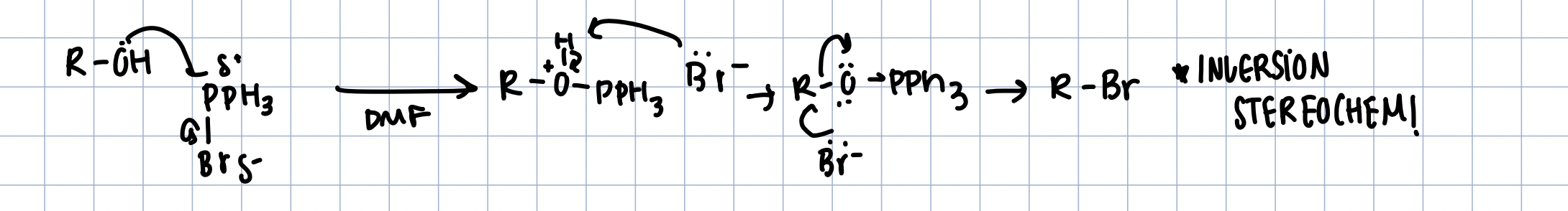
Alcohols - Bromides (inversion)
PBr in DMF
like SN2
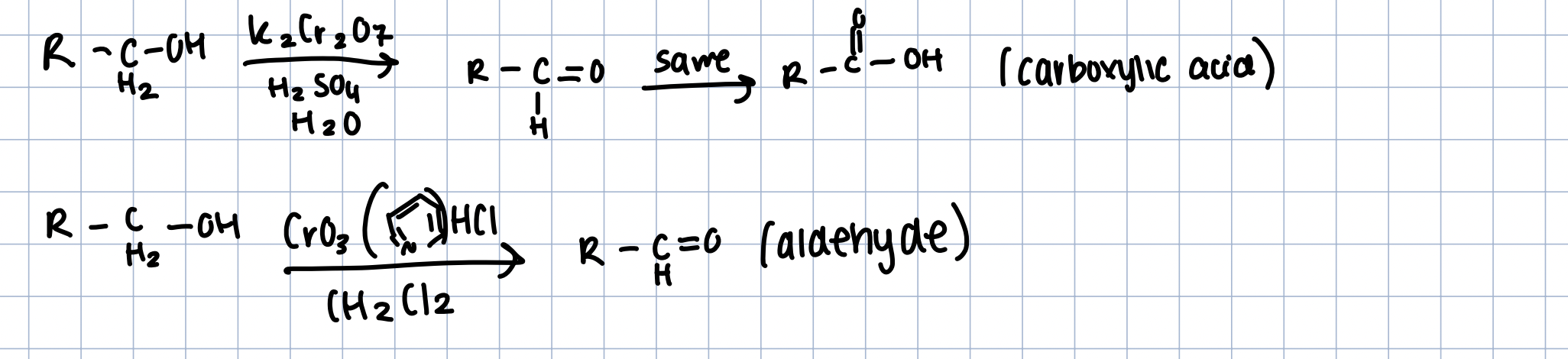
Oxidation of Alcohols (overview of degrees)
3º - no oxidation bc no alpha hydrogens
2º - ketone product
1º - aldehyde or carboxylic acid
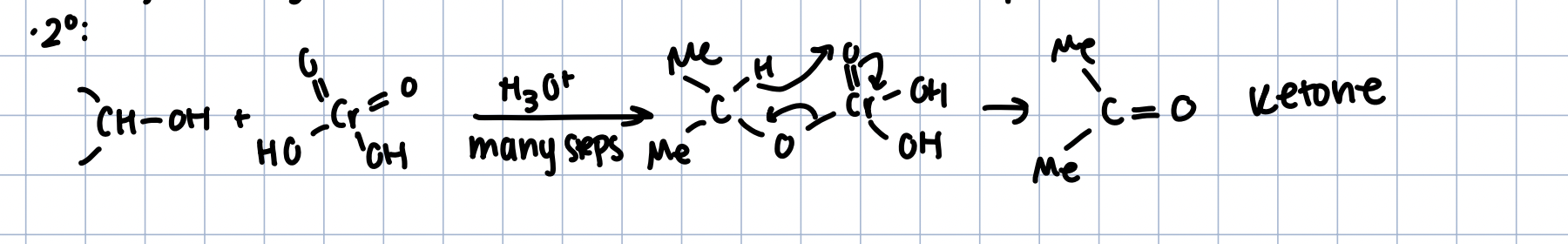
Oxidation of 2º Alcohols
CrO42- in acidic conditions, or CrO3 in pyradine
Oxidation of 1º Alcohols
K2Cr2O7 in H2SO4 and H2O - overoxidize to carboxylic acid
CrO3(pyridine)HCl in Ch2Cl2 - Aldehyde
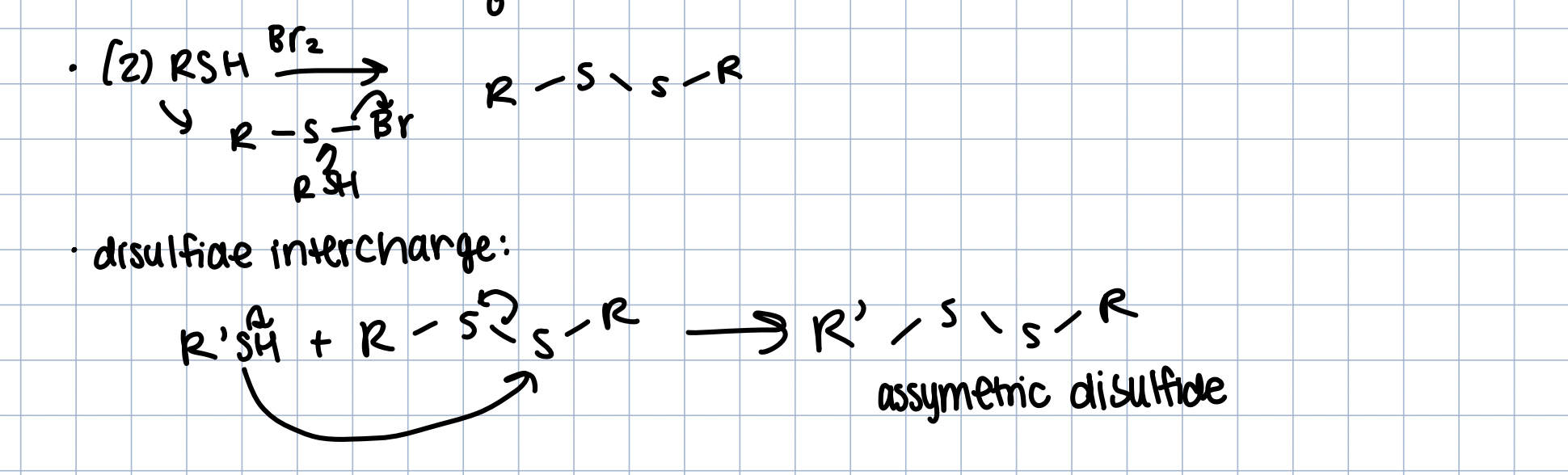
Oxidation of Thiols - Disulfide
(2RSH) in Br2 or I2
weak oxidant
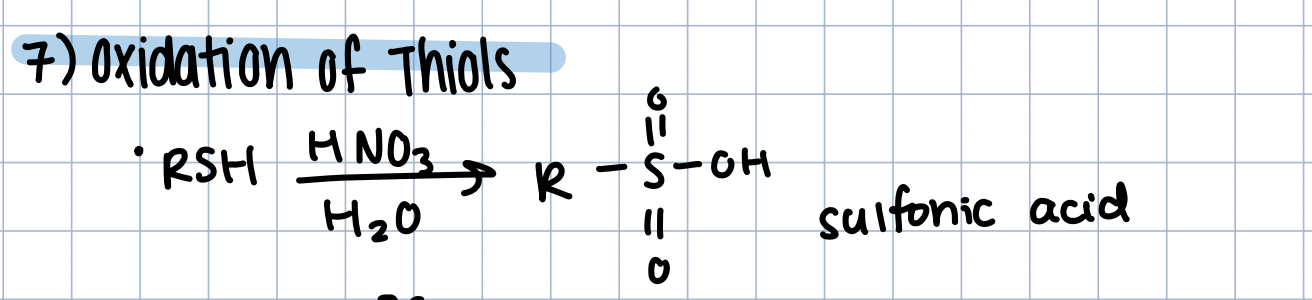
Oxidation of Thiols - Sulfonic Acid
RSH in HNO3/H2O
stronger oxidation
glycol
1,2 diol
Synthesis of Ethers (2 ways)
Sn2 williamson ether, alkoxymercuration-reduction
SN2 Williamson Ether/Sulfide Reaction
Strong base (NaH), then primary alkane
convert to alkoxide (or thiolate), then SN2 to get the ether/sulfide
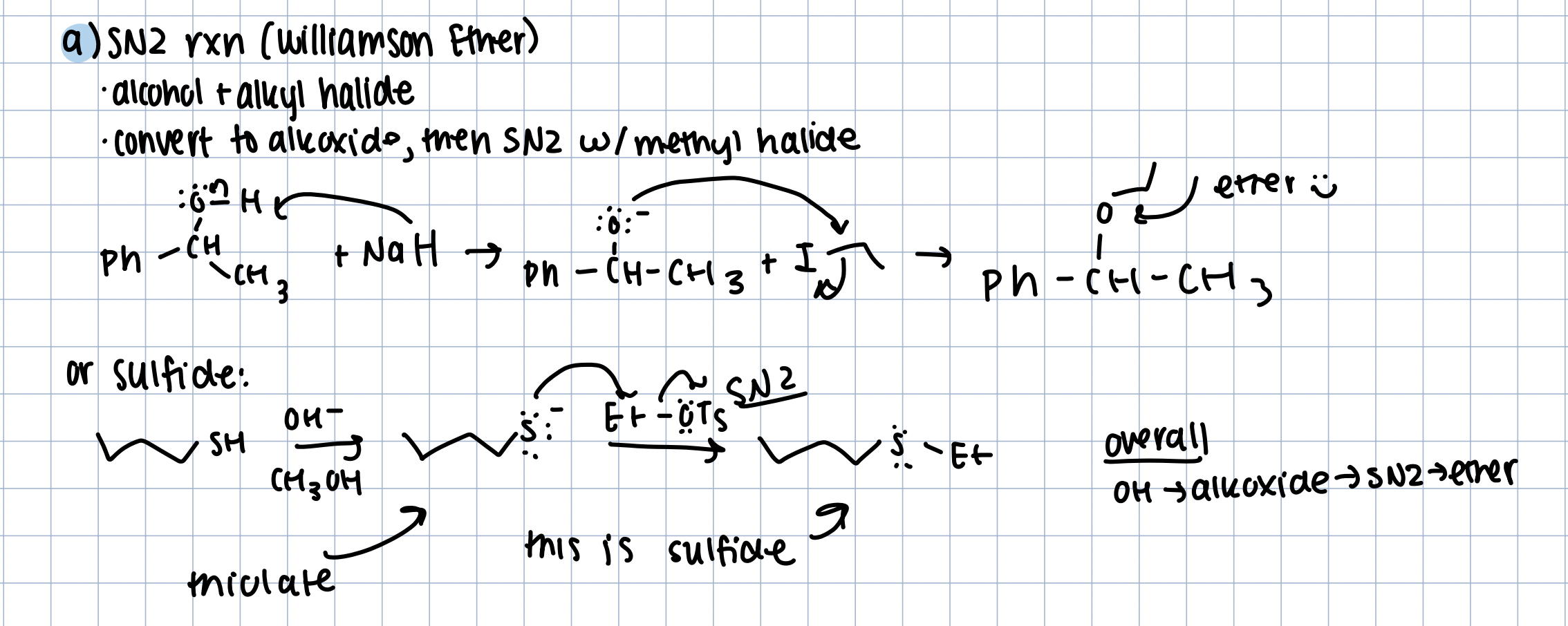
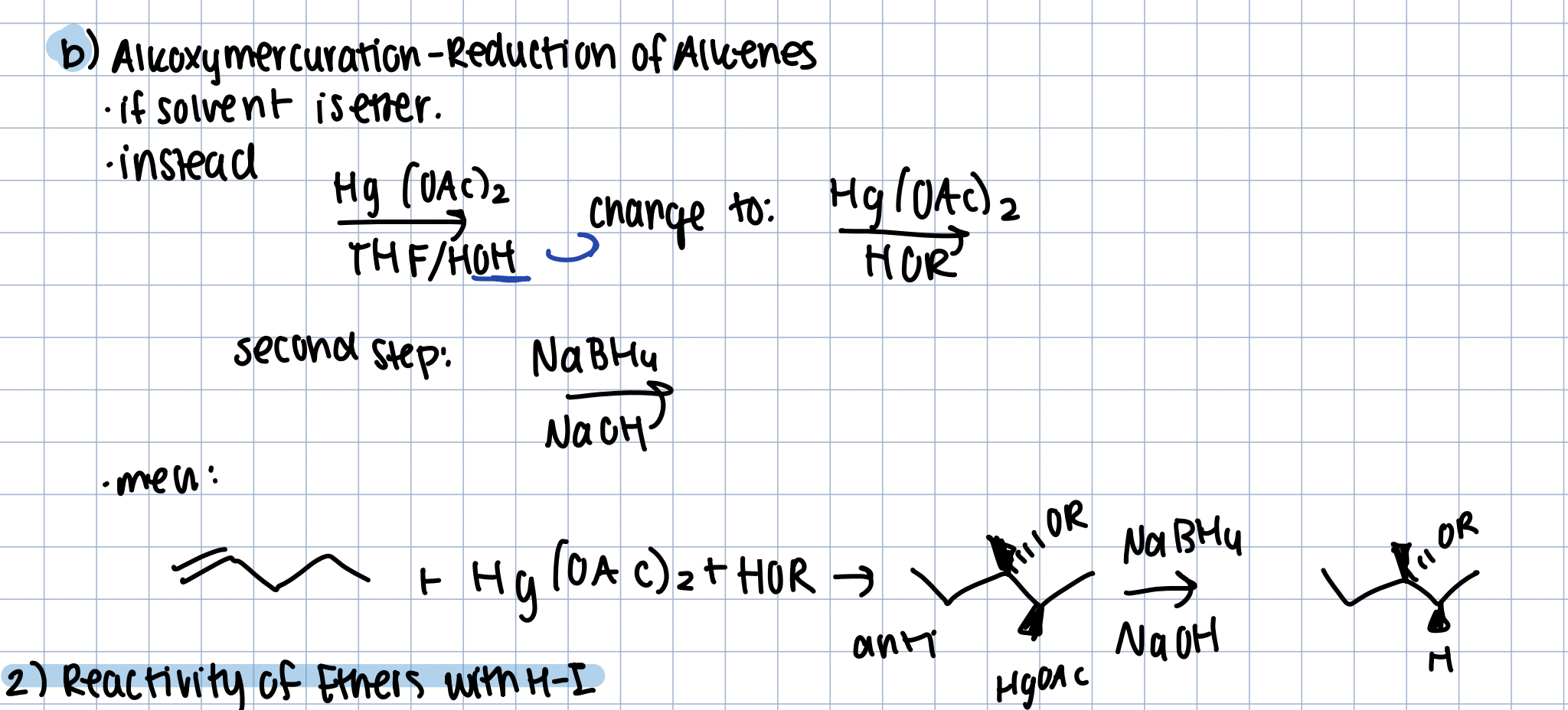
Alkoxymercuration-Reduction of Alkene to obtain an Ether
Hg(OAc)2 in HO-R, then NaBH4 in NaOH
anti addition of the OR and HgOAc, then becomes ether
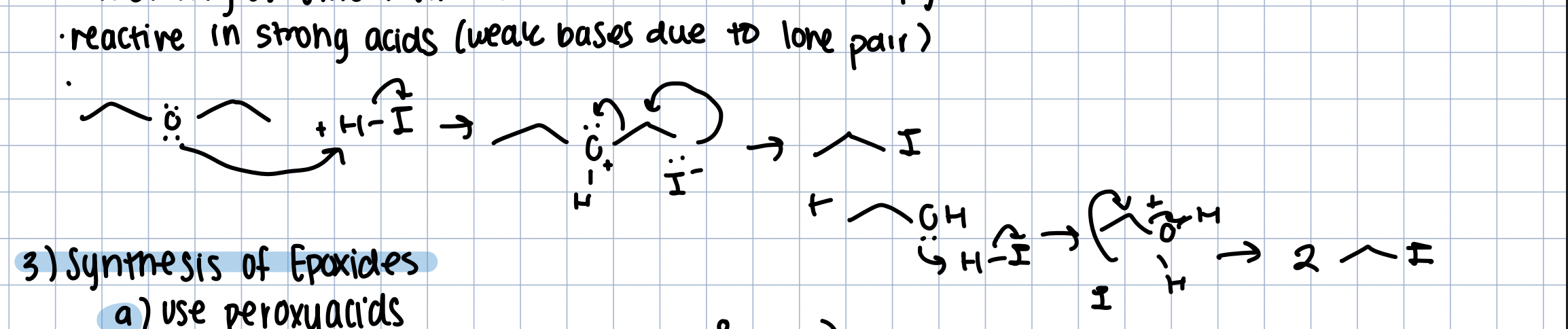
Reactivity of Ethers with H-I
Ether is reactive in strong acid
Is protonated by H+, SN2 rxn, then again the alcohol is protonated to good LG and SN2 again
Synthesis of Epoxides (2 ways)
One step peroxyacid (mCPBA), Halohydrins (2 steps)
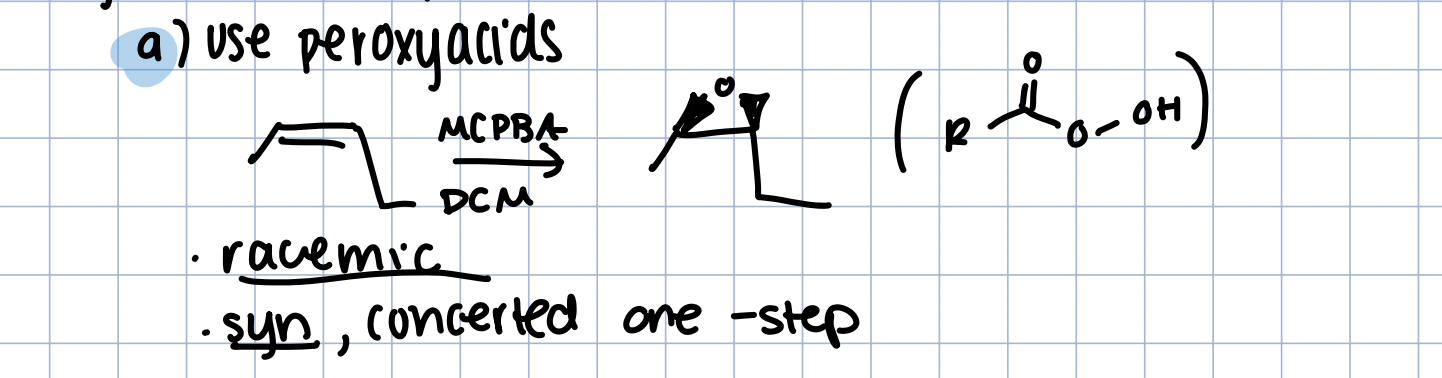
Synthesis of Epoxide with peroxyacid
using mcPBA in DCM
one step concerted
syn addition
forms racemic
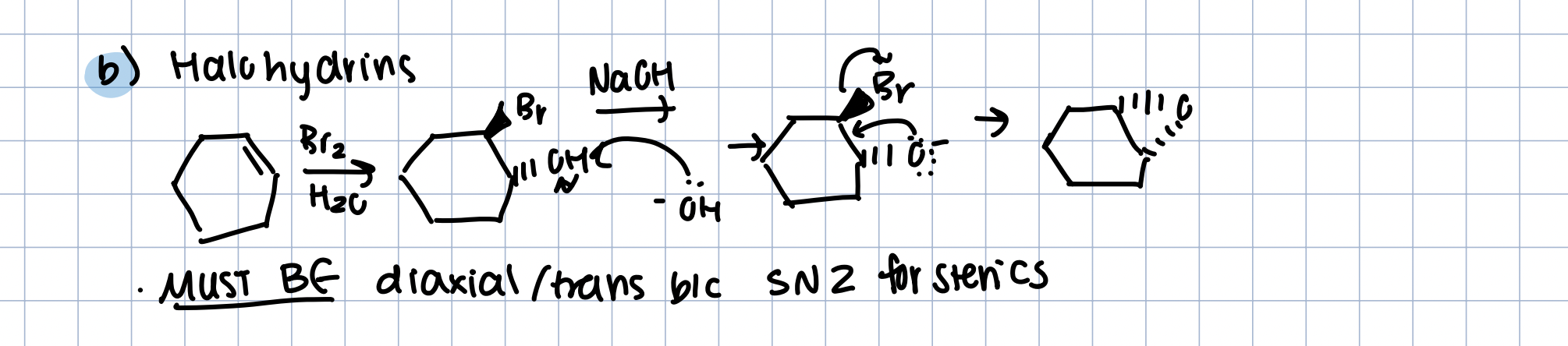
Synthesis of Epoxide with Halohydrins
Br2 in H20, then NaOH
turns to halohydrin (anti b/c halonium ion), then SN2 with OH-
must be diaxial/trans for SN2 sterics
Reactivity of Epoxides (Openings) (2 ways)
Acid/Base, Grignard (both anti)
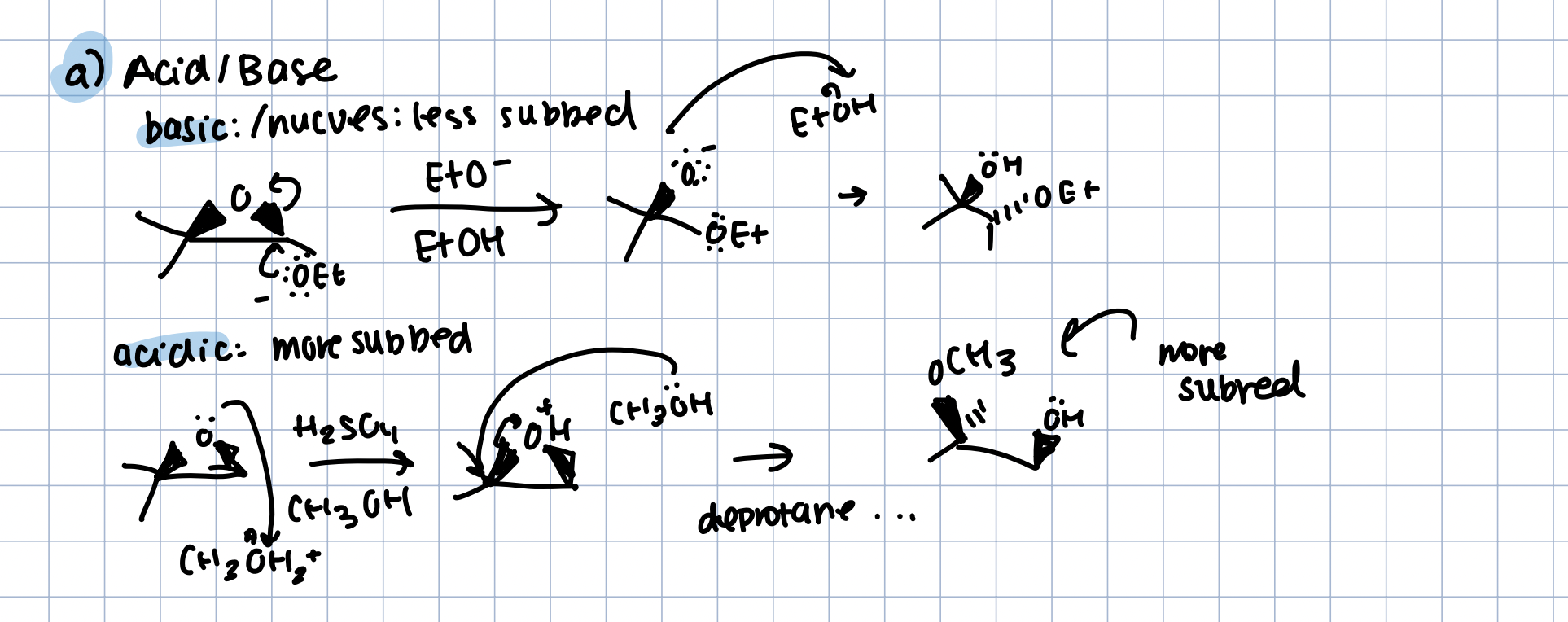
Acid/Base opening of Epoxides
Acid: Nucleophile in Conj. Acid
nucleophile attacks less subbed
Base: Acid in Nucleophile
nucleophile attacks more subbed
anti
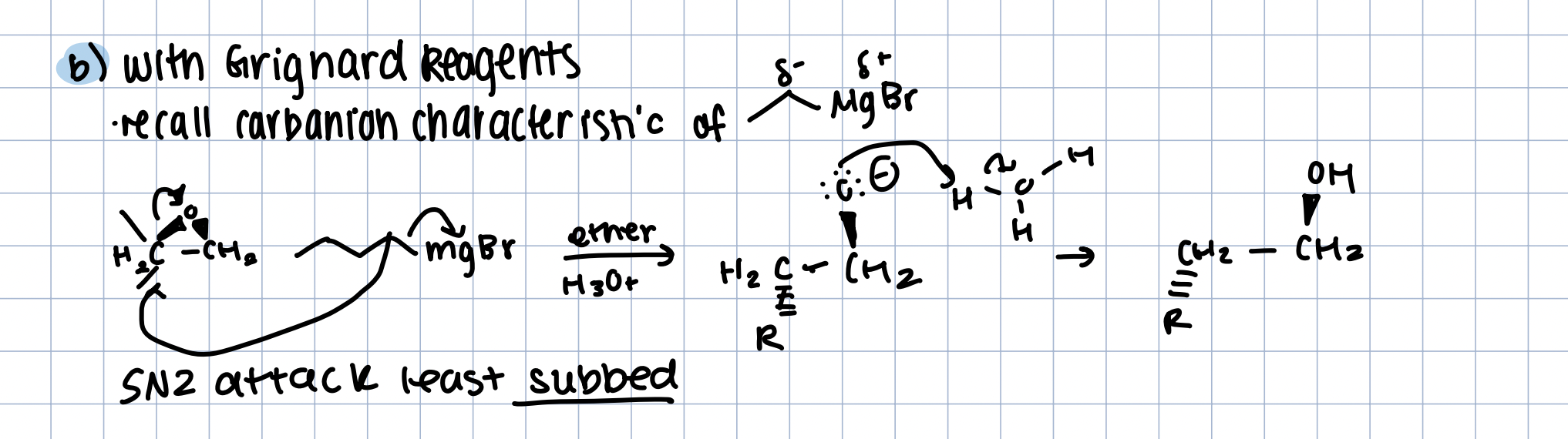
Epoxide Opening with Grignard Reagent
recall carbanion characteristic
R - MgBrin ether and H30+
Sn2 attack by R- MgBr, O- protonated by H3O+
SN2 will attack least subbed
Synthesis of Glycols/Diols (2 ways)
Acid/Base catalyzed epoxide opening (trans)
Dihydroxylation of an Alkene (cis)
Synthesis of Glycol with Acid/Base Epoxide opening
Acid: NaOH in H2O
Base: H3O+ in H2O
product will be trans

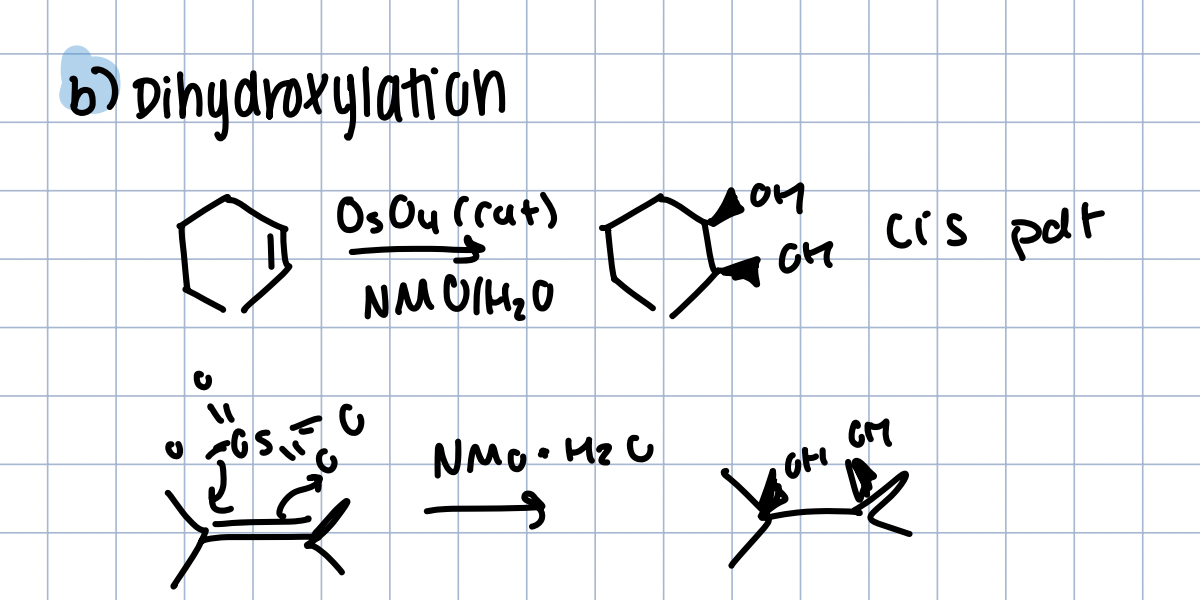
Synthesis of Glycol with Dihydroxylation
OsO4 in NMO/H2O
cis product
Reactivity of Glycols/Diols
H5IO6 OR HIO4 in HOAc
cleavage
must be a syn diol because undergoes cyclic intermediate