Edexcel IAL Chemistry Unit 1
1/204
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
205 Terms
Mole (mol)
The unit for amount of substance; contains 6.02 times 1023 particles.
6.02 times 1023 mol-1.
Relative Atomic Mass (Ar)
The mass per mole of a substance (gmol-1).
The volume occupied by 1 mole of any gas at a specific temperature and pressure (24 dm3 at RTP).
pV = nRT (where p is pressure in Pa, V is volume in m3, T is temp in K).
The amount of solute (mol) dissolved in a volume of solution (dm3).
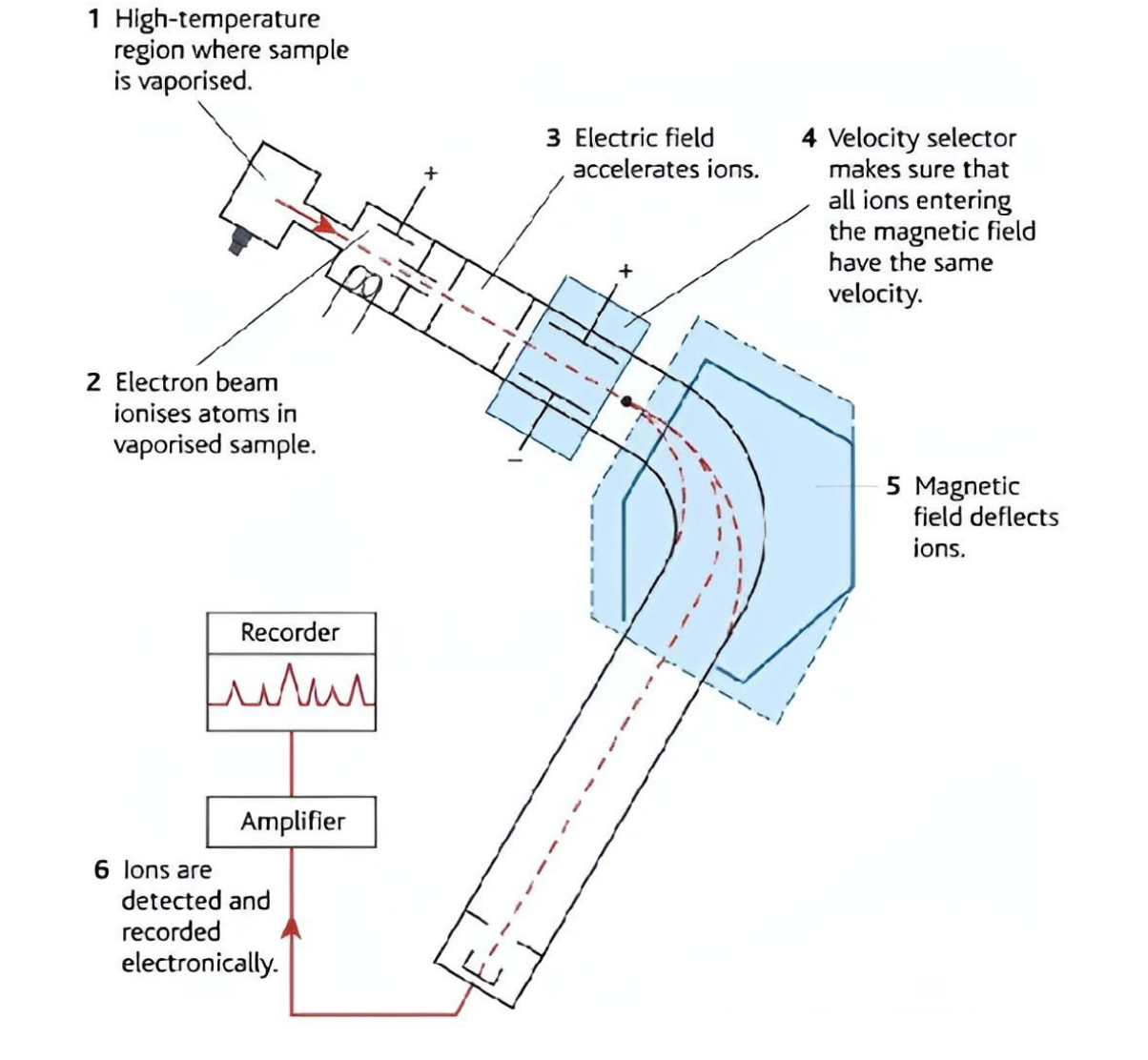
Mass Spectrometer Step 1
Vaporisation: The sample is turned into a gas.
Deflection: Ions are deflected by a magnetic field according to their mass-to-charge (m/z) ratio.
Molecular Ion Peak
The peak with the highest m/z ratio, representing the relative molecular mass (Mr).

s-orbital Shape
Spherical.
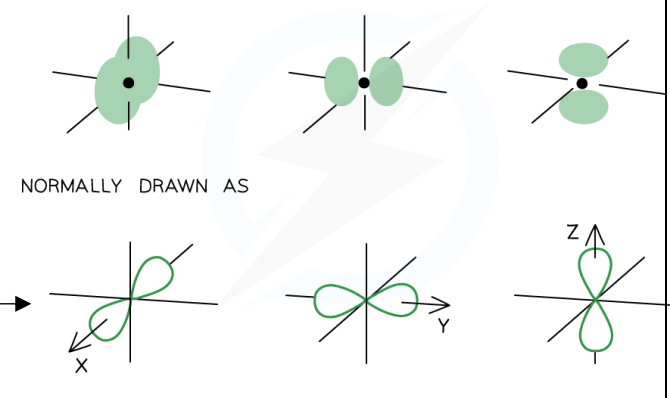
p-orbital Shape
Dumbbell-shaped.

First Ionisation Energy
The energy required to remove one mole of electrons from one mole of gaseous atoms to form one mole of 1+ gaseous ions.
The strong net electrostatic force of attraction between oppositely charged ions.
A family of compounds with the same functional group and general formula, differing by CH2.
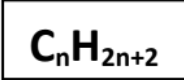
Saturated
Contains only single carbon-carbon bonds (Alkanes).
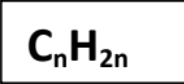
Unsaturated
Contains at least one C=C double bond (Alkenes).
Hazard: Toxic
Represented by a skull and crossbones; can cause death if swallowed or inhaled.

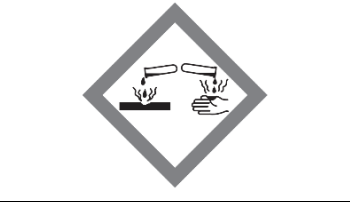
Hazard: Corrosive
Can damage skin or eyes on contact and destroy metals.
Ions with the same number of electrons (e.g., N3-, O2-, F-, Na+, Mg2+, Al3+.
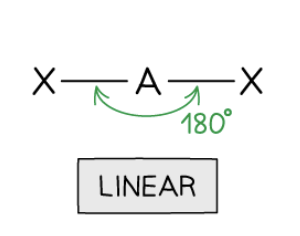
Linear Shape
2 bonding pairs, 0 lone pairs, 180° bond angle.
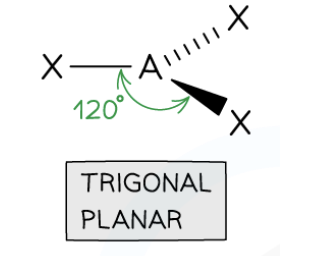
Trigonal Planar Shape
3 bonding pairs, 0 lone pairs, 120° bond angle.
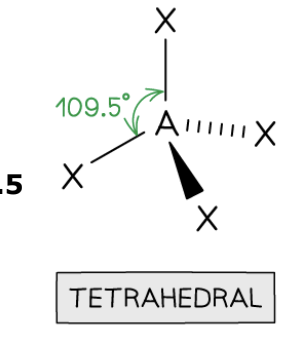
Tetrahedral Shape
4 bonding pairs, 0 lone pairs, 109.5° bond angle.
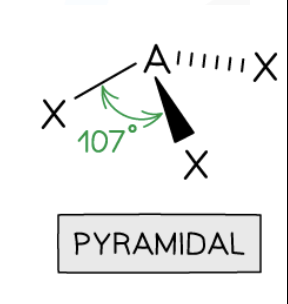
Trigonal Pyramidal Shape
3 bonding pairs, 1 lone pair, 107° bond angle (e.g., NH3.
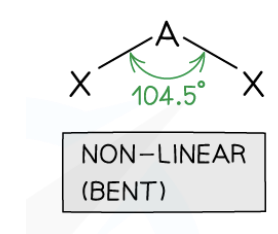
V-Shaped / Bent
2 bonding pairs, 2 lone pairs, 104.5° bond angle (e.g., H2O).
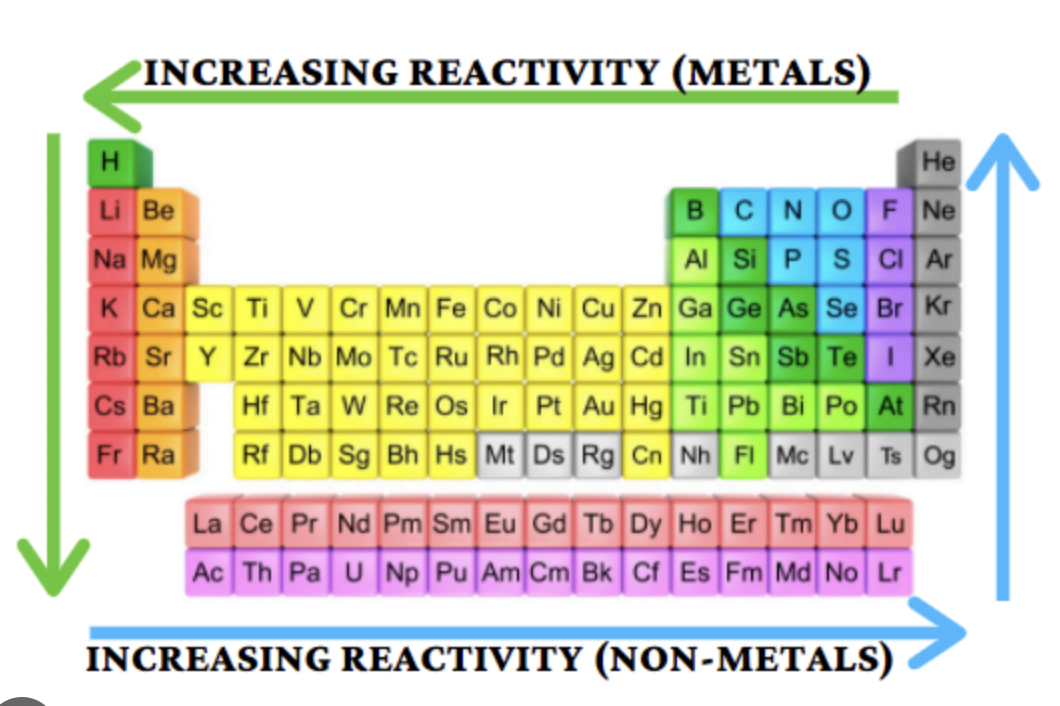
Group 2 Reactivity Trend
Increases down the group
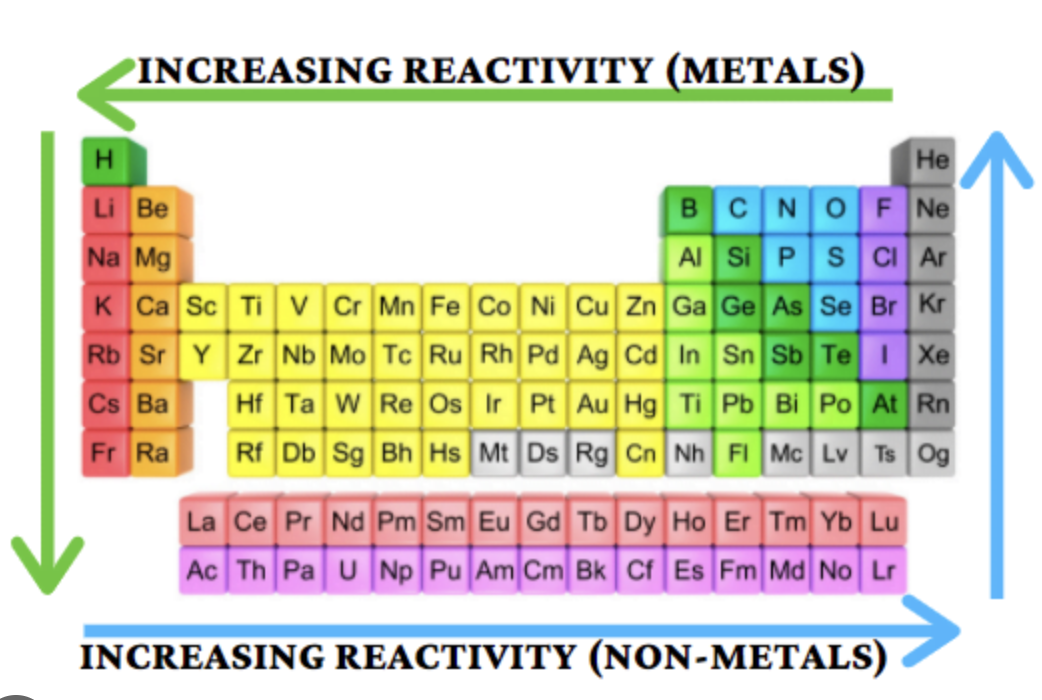
Group 7 Reactivity Trend
Decreases down the group
Alkane + Halogen (UV)
Halogenoalkane + Hydrogen Halide
Alkene + Hydrogen (Nickel)
Alkane
Alkene + Bromine Water
1,2-dibromoalkane (Colourless)