Intro to physical science final
1/61
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
62 Terms
Immersed an object in a large tank of water and then let it go. What kind go information would you need to know about the object in order to predict in advance whether it will rise to tip and float, stay, or sink to the bottom?
The information I would need to know about the object is the objects mass and volume to find the objects density. Would need to know if the object is less than. more than, or equal to the density of water to guess what the object will do when placed into the water.


When you place an object completely under water it displaces a volume of water equal to its own volume. Since both the object and displaced water have the same volume, comparing the mass of the object to the mass of the water displaced the same as comparing the density of the object to the density of the surrounding water.
Three platic bags filled with different liquids in a tank of water.
The water in the tank has a density of 1.00 g/ML
Table record whether each plastic bag sinks, floats, or remains where it was placed
Plain water- 1.00 g/ml -Remains where it was placed
Salt water- 1.20g/ml -Sinks
Rubbing alcohol- 0.786 g/ml -floats


Three platic bags filled with different liquids in a tank of water.
The water in the tank has a density of 1.00 g/ML
Table record whether each plastic bag sinks, floats, or remains where it was placed
Plain water- 1.00 g/ml
Plain water- 1.00 g/ml -Remains where it was placed


Three platic bags filled with different liquids in a tank of water.
The water in the tank has a density of 1.00 g/ML
Table record whether each plastic bag sinks, floats, or remains where it was placed
Salter water-1.20 g/ml
Salt water- 1.20g/ml -Sinks


Three platic bags filled with different liquids in a tank of water.
The water in the tank has a density of 1.00 g/ML
Table record whether each plastic bag sinks, floats, or remains where it was placed
Rubbing alcohol- 0.786 g/ml
Rubbing alcohol- 0.786 g/ml -floats

If the density of the object is greater than the density of water, would the
object sink, float, or remain submerged where it is?
Sinks

if the density of the object is less than the density of water, would the object
sink, float, or remain submerged where it is?
Floats

If the density of the object is equal to the density of water, would the object
sink, float, or remain submerged where it is?
Remains submerged where it is

Although the demonstration was done i n a tank o f water, the results can be made more general. Whenever an object is immersed in any fluid (liquid or gas), to decide whether it will rise up, sink down, or remain where it is, you need to compare its density to the density of the surrounding fluid.
The object will rise up and float if its density is less than the density of the
surrounding fluid.
• The object will sink if its density is greater than the density of the surrounding
fluid.
• The object will remain where it is if its density equals the density of the surrounding
fluid.

Suppose a block of material floats i n liquid A , but sinks in liquid B. What can
you say about the relative densities of the two liquids and the material out of
which the block was made?
-The material has a density that is less than liquid A because it floats in liquid A.
-The material has a density that is more than liquid B because it sinks in liquid B.

Hydrogen gas, despite its high flammability,
was once used to fill airships. Following the
Hindenburg disaster on May 6, 1937, hydrogen
was replaced with nonflammable helium gas.
Compare the densities of air and helium
(C5A1 Table 7). Why do you think helium is
used to fill modern-day airships, such as the
Goodyear blimp?
The density of air is 0.00121 and the density of helium is 0.00016. Helium is used to fill modern day airships because heliums density is less than airs, which causes the airship to rise up or float.

a) Would you expect a block of steel to sink or float in water? Why?
I would expect a block of steel to sink because a block of steel is more than the density of water.

b) Why does a steel ship float? (Hint: Think about what else takes up space
in the ship besides steel.)
A steel ship floats because steel ships are hollow. The steel ship has air because
it is hollow. Airs density is less than waters, which causes the steel ship to float.
c) Why might the density of a material and the density of an object constructed
from that material be different?
The density of a material and the density of an object constructed from the material might be different because like the steel ship it is hollow, which changes its density. The steel material doesn’t have any hollow spaces or air pockets like an object might have, which makes the density different than the objects.
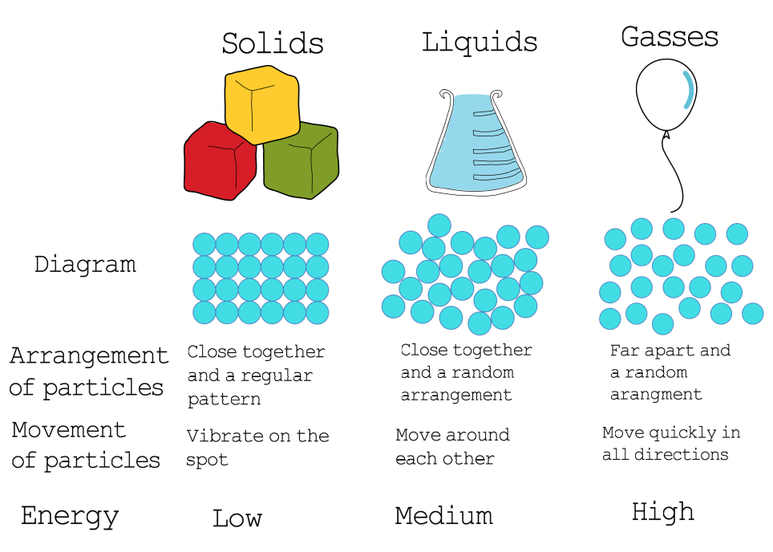
Draw a picture of how you think liquids differ from gases in terms of particles
-Liquid particles have moderate attraction, keeping them close together but allowing them to flow
-gas particles have negligible attraction and move freely
-Gas particles are further a part liquids are closer
-Gasses move faster
-Luqids are slower, but still more
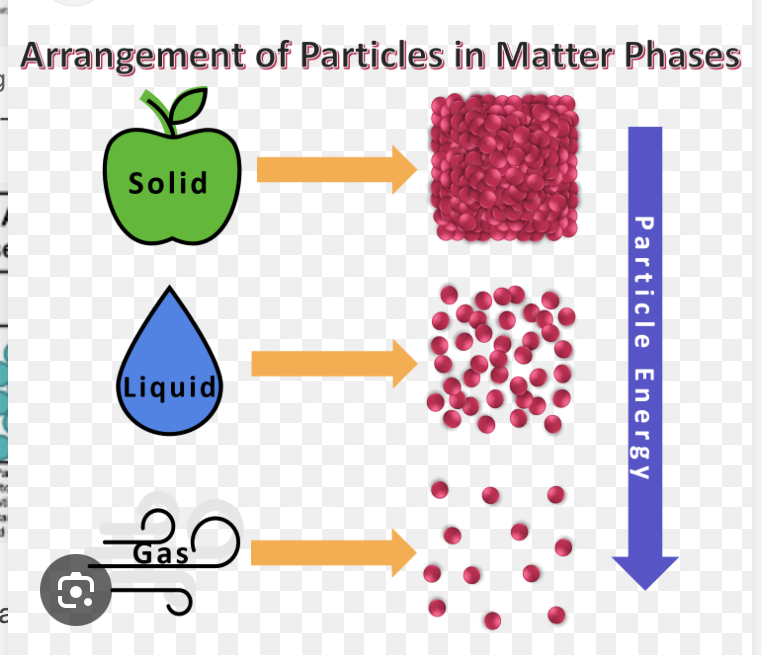
Solids different from liquids in terms of particles
Solid particles have very strong attractions, keeping them locked in fixed positions and vibrating in place, resulting in a definite shape and volume.
Liquid particles have weaker attractions, allowing them to remain close together (maintaining a definite volume) but slide past one another, allowing them to flow and take the shape of their container
-Solid partcles are closer together
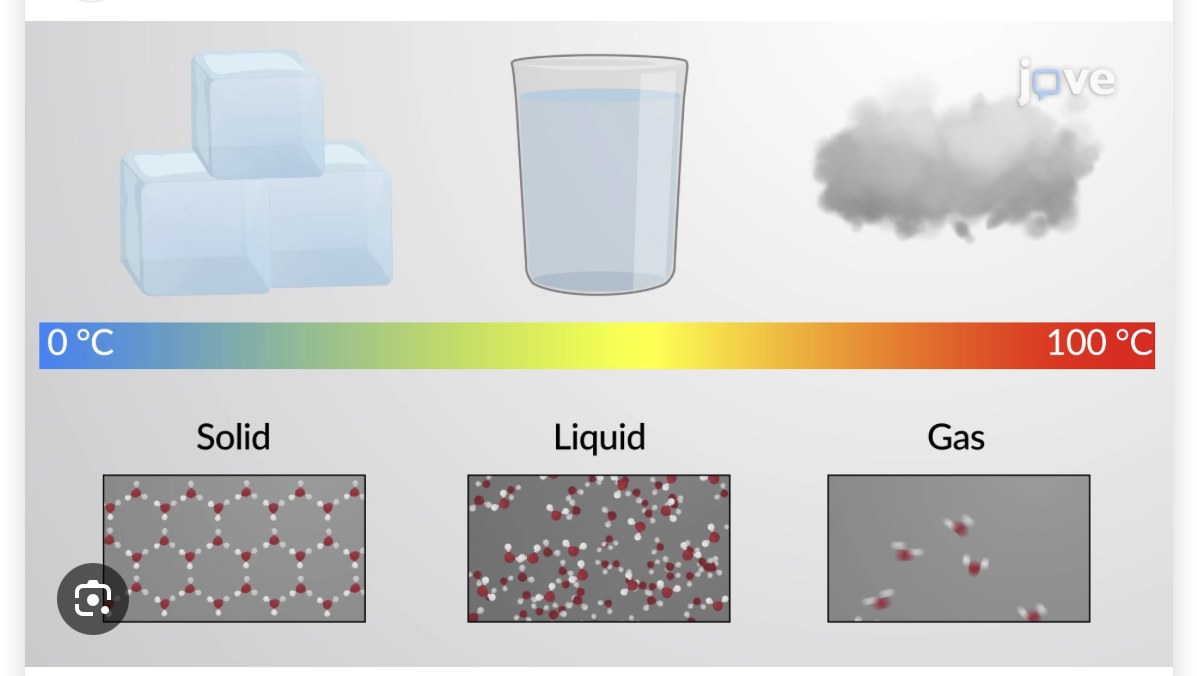

1.Add one drop of food coloring and allow the beaker to sit undisturbed for a few minutes. Do not swirl or stir the water
2.Observe the water and food coloring in the beaker again.
3.Evidence for particle motion in liquids?
1.It looks like marble coloring or a lava lamp
2.All the color has spread evenly
3.Yes since the color spreads

Based on food color observations how do you think the motion and spacing of particles might be different in liquid compared to gases?
Liquids are more dense
They spread out fairly evenly
Food coloring: Sinks to the buttom not mixing equally. More on the butoom, but more equally spread

Based on your observations how do you think the spacing of particles might be different in solids as compared to liquids?
Solids have less space compared to liquids
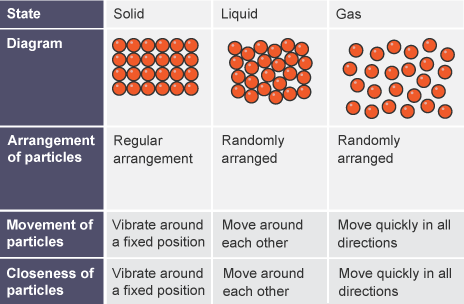
Equal volumes of gases at the same temperature and pressure have equal numbers of particles. Why do they have different masses (and densities)?
Distance of the particles makes them have different masses and denies because they are different materials.
In terms of particles why is the solid state of a material denser than the liquid state of the same material?
The particles are more condensed closer together
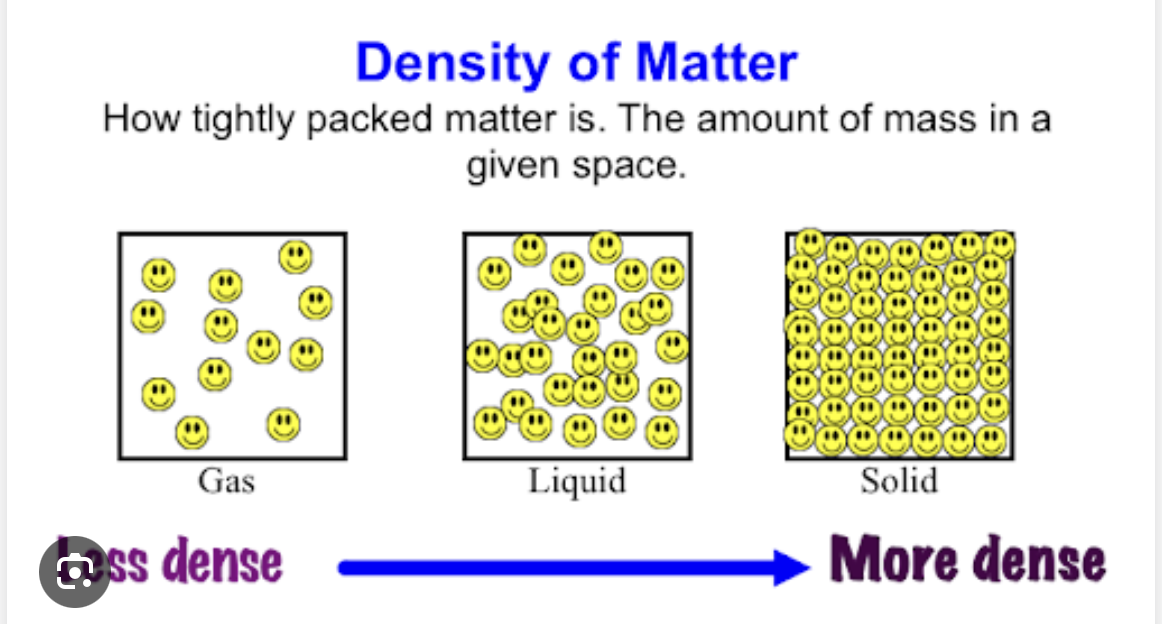
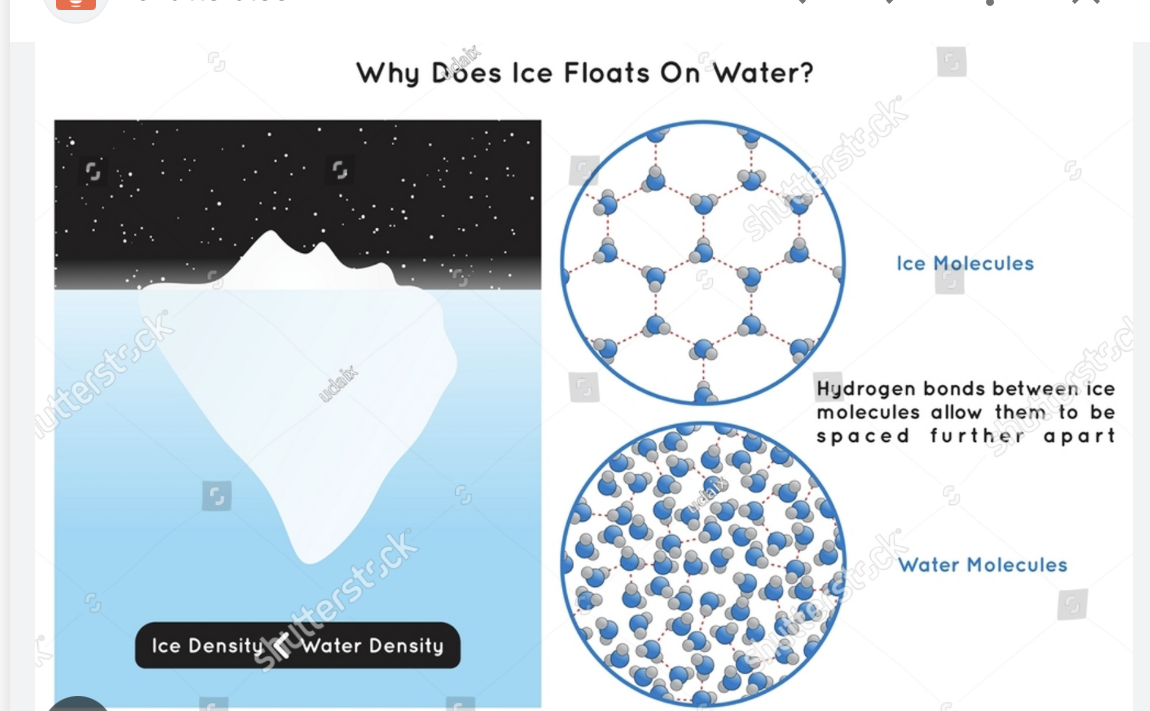
In terms of particles why does ice float in water? Briefly explain in terms of particles
Less dense air pockets too
Ice is less dense than water. Air pockets like in a ship
More strength of attraction
Solid the most
Least strength of attraction
Gass Particles
-Liquid is less than solid, but Gas is less-Not attracted.
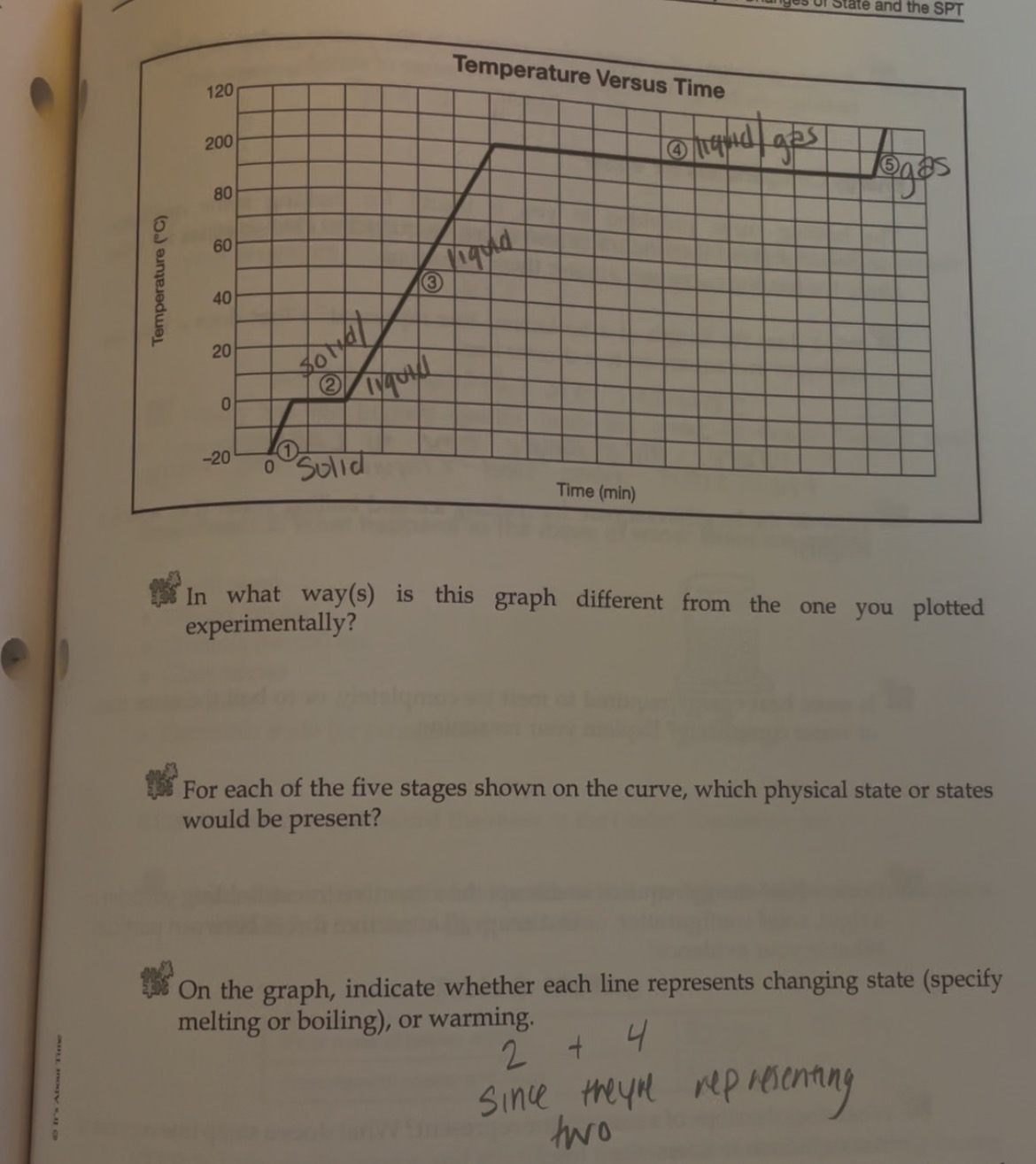
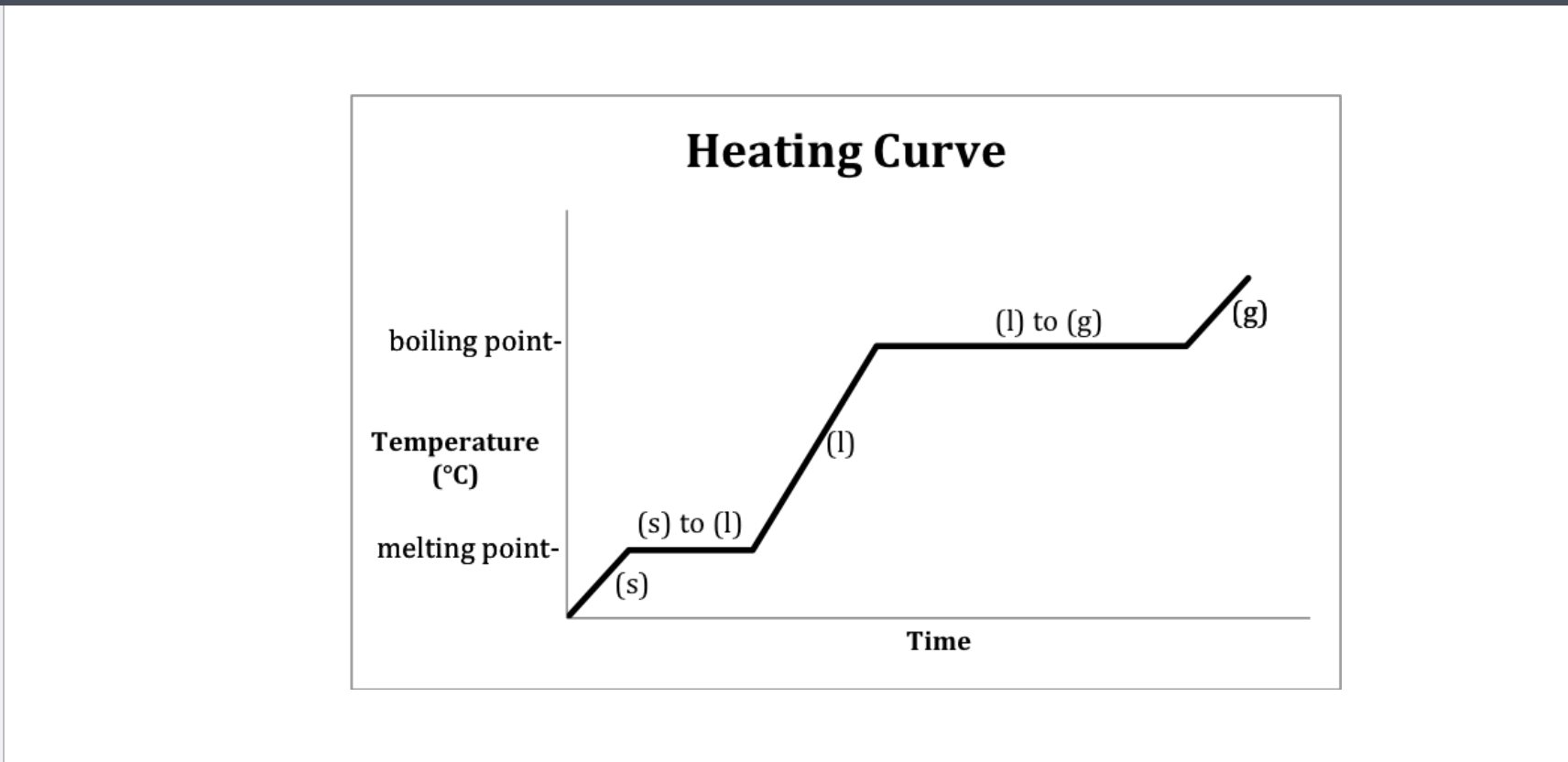
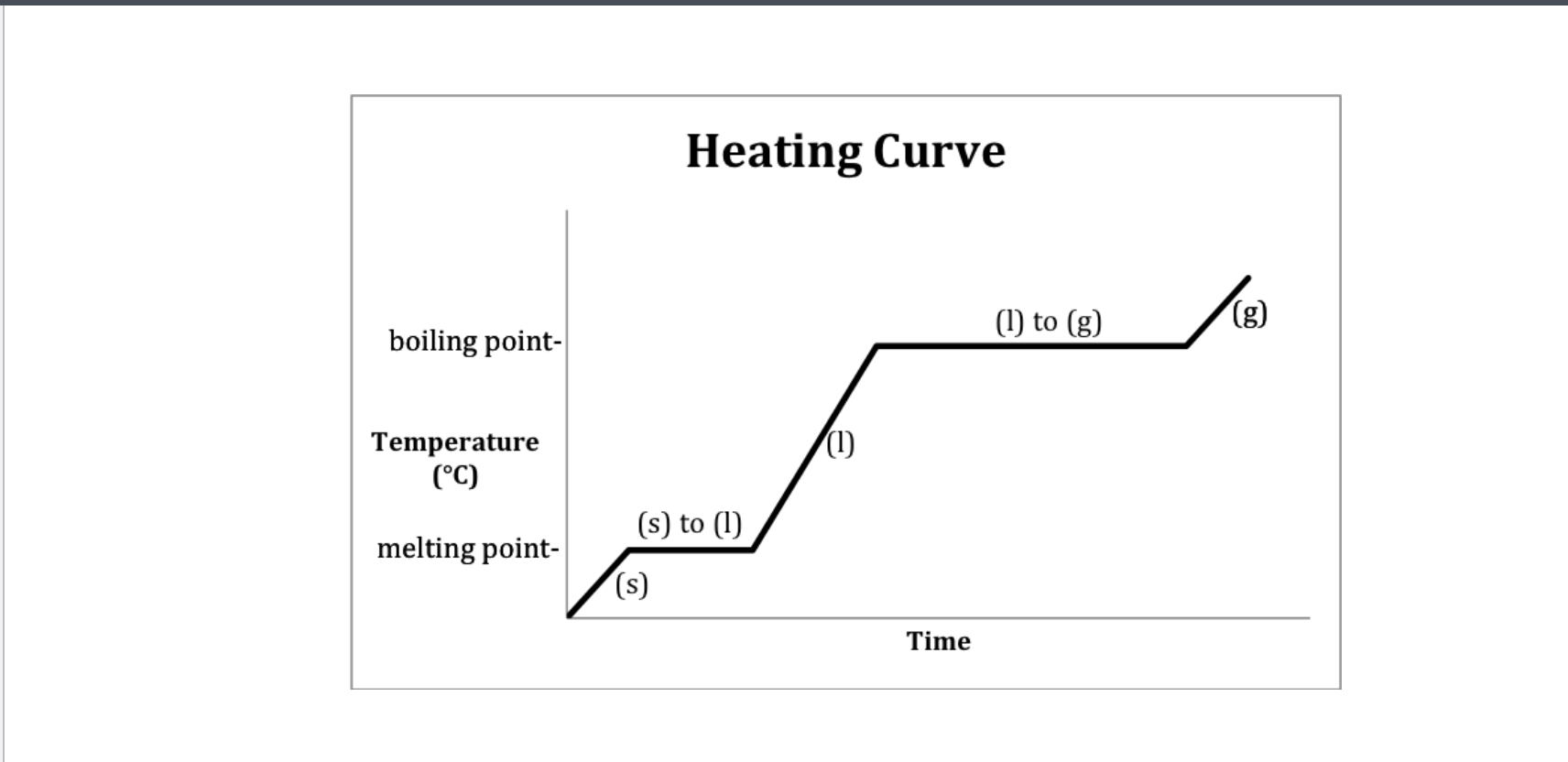
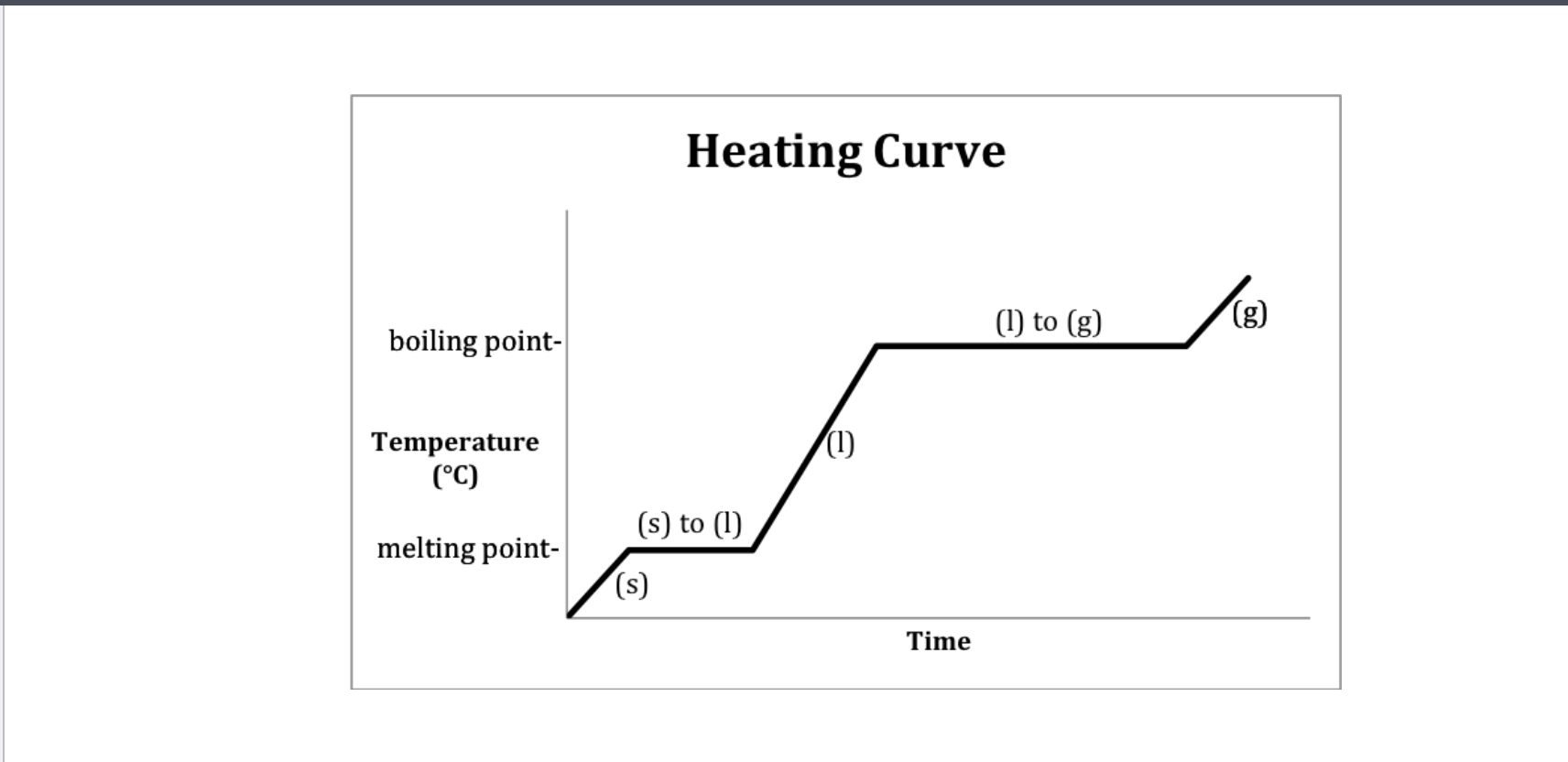
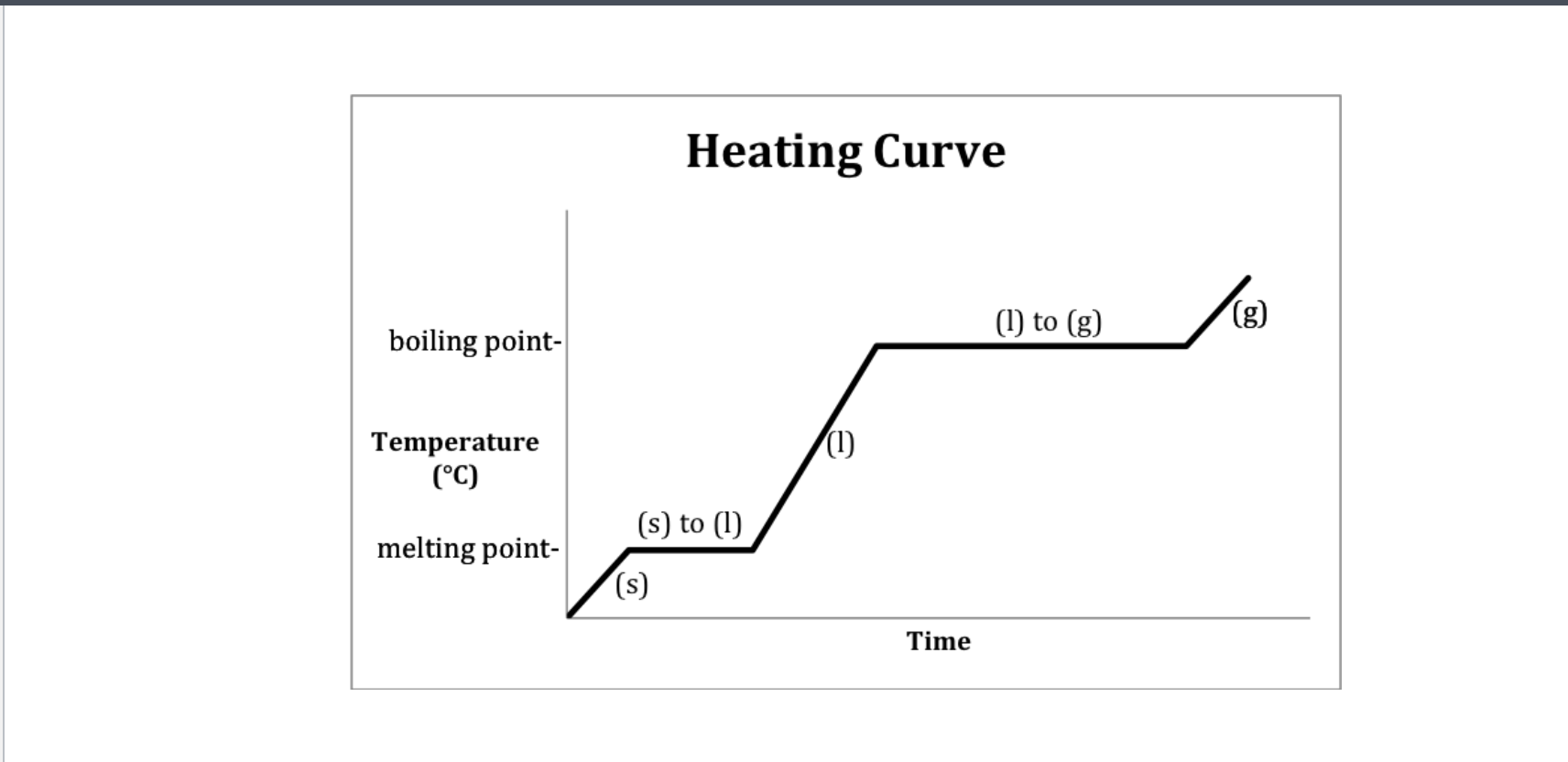
If only one state of a material is present in the system what is the result of heating a change in termperature or a change of state? -Heating curve
1,3,5 change in state
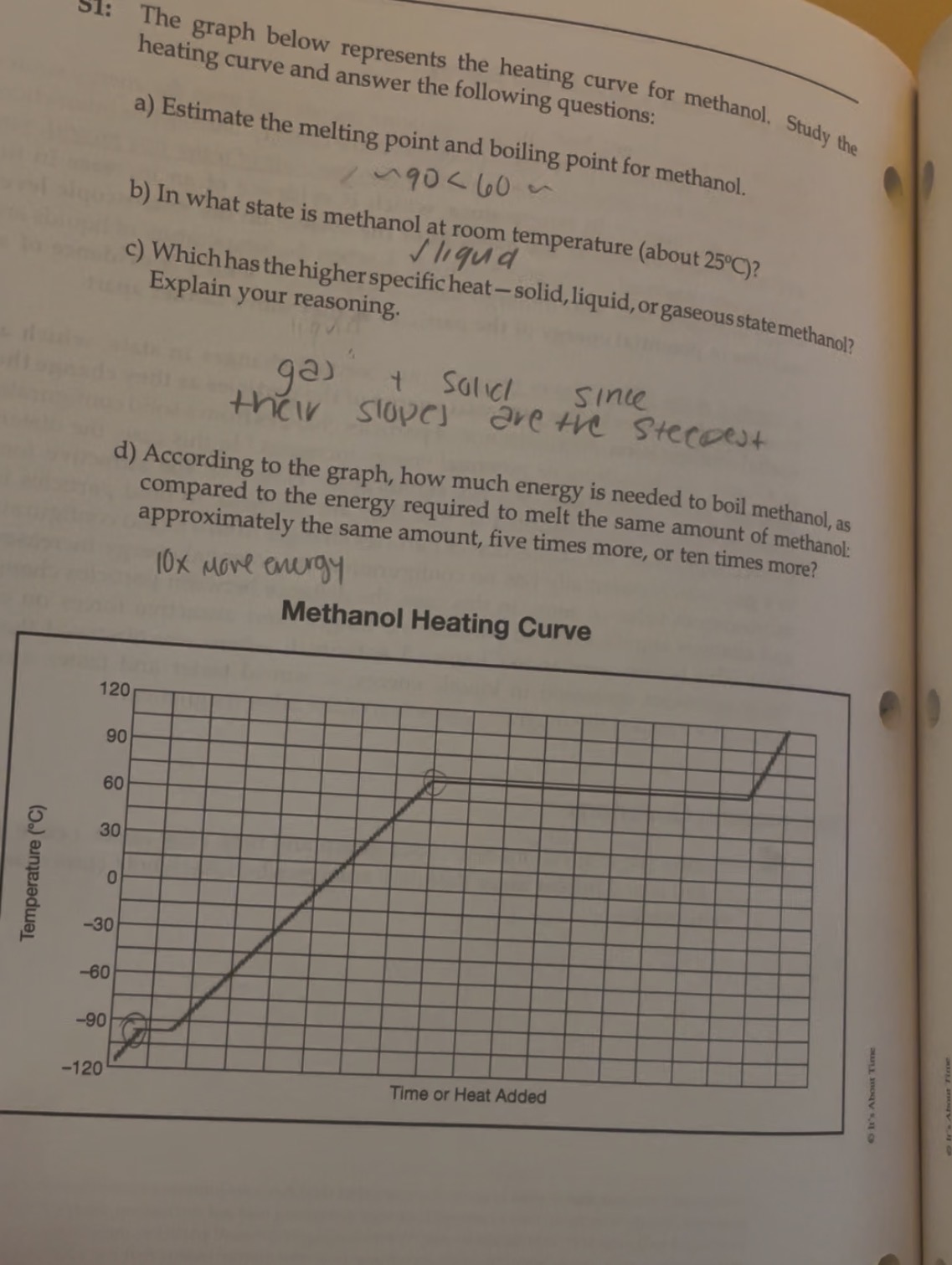
Methanol heating curve
Which has the highest specific heat-solid, liquid, and gaseous state methanol?
Gas and solid since their slopes are the steepest
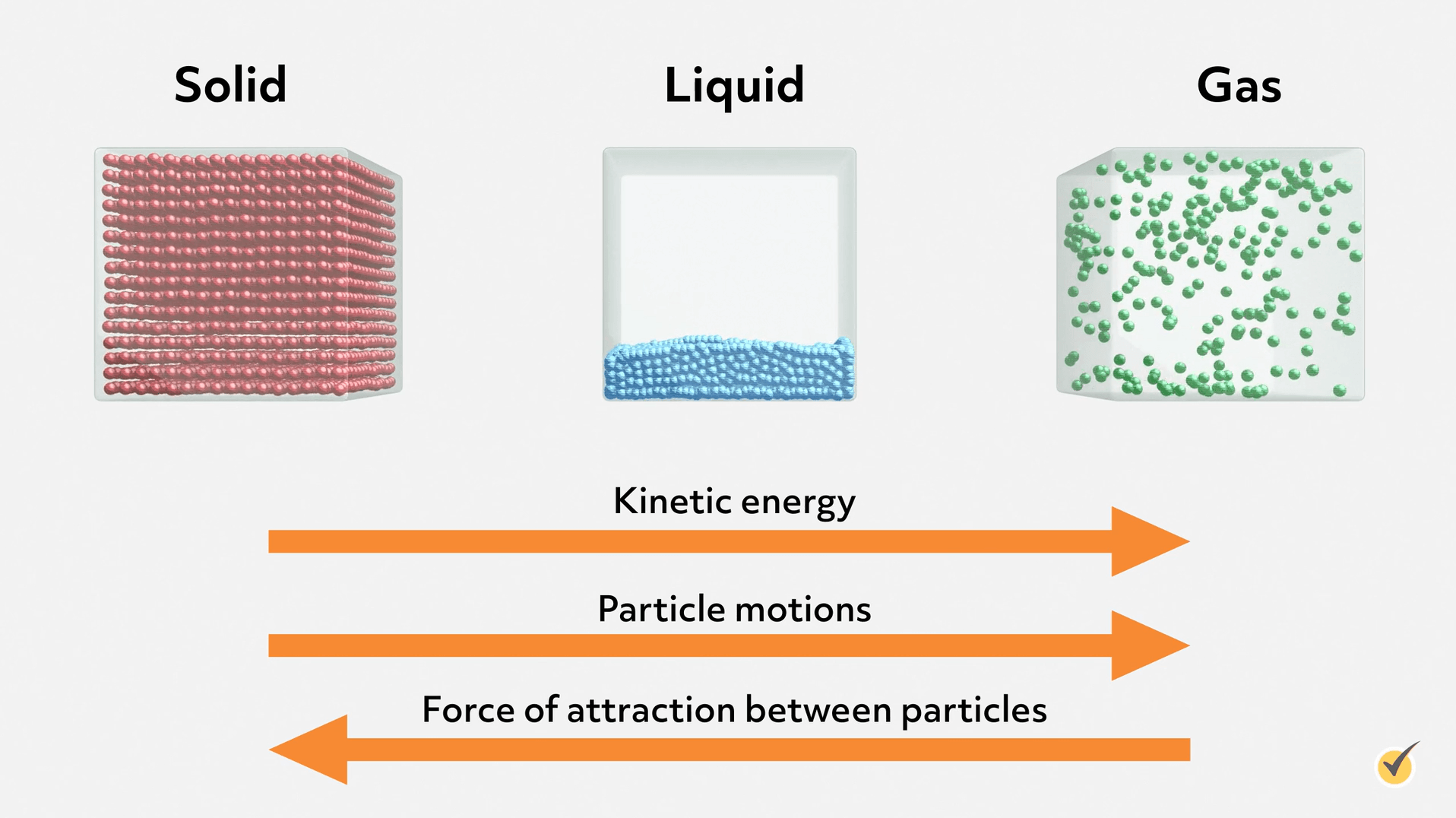
For 3 different materials at room temperature-one is gas, one is liquid and the other is solid-how would you rank the strength of attractive forces between particles for these materials? Explain your reasoning?
Solid strongest
Liquid
Gas weakest
Shaking up Italian Dressing
Italian salad dressing consisting of equal volumes of water and oil are mixed together. The mixture was allowed to sit undisturbed for awhile
What happens to the mixture after sitting undisturbed for awhile?
Seperates

Shaking up Italian Dressing
Italian salad dressing consisting of equal volumes of water and oil are mixed together. The mixture was allowed to sit undisturbed for awhile
Which material sinks to the buttom and which floats to the top?
Water sinks, oil floats


Shaking up Italian Dressing
Italian salad dressing consisting of equal volumes of water and oil are mixed together. The mixture was allowed to sit undisturbed for awhile
According to your observations which has greater density- oil or water?
Water has a greater density, why oil floats on top of the oil
Shaking up Italian dressing
When italian dressing settles after being shaken, the oil rises to the top, while the denser vinegar, water, and herbs settle at the bottom. This separation occurs because oil is less dense than water and vinegar.
Top: Oil (olive oil, vegetable oil).
Bottom: Vinegar, water, herbs, and spices.
Tip: If olive oil solidifies in the fridge, let it sit at room temperature for a few minutes before shaking.

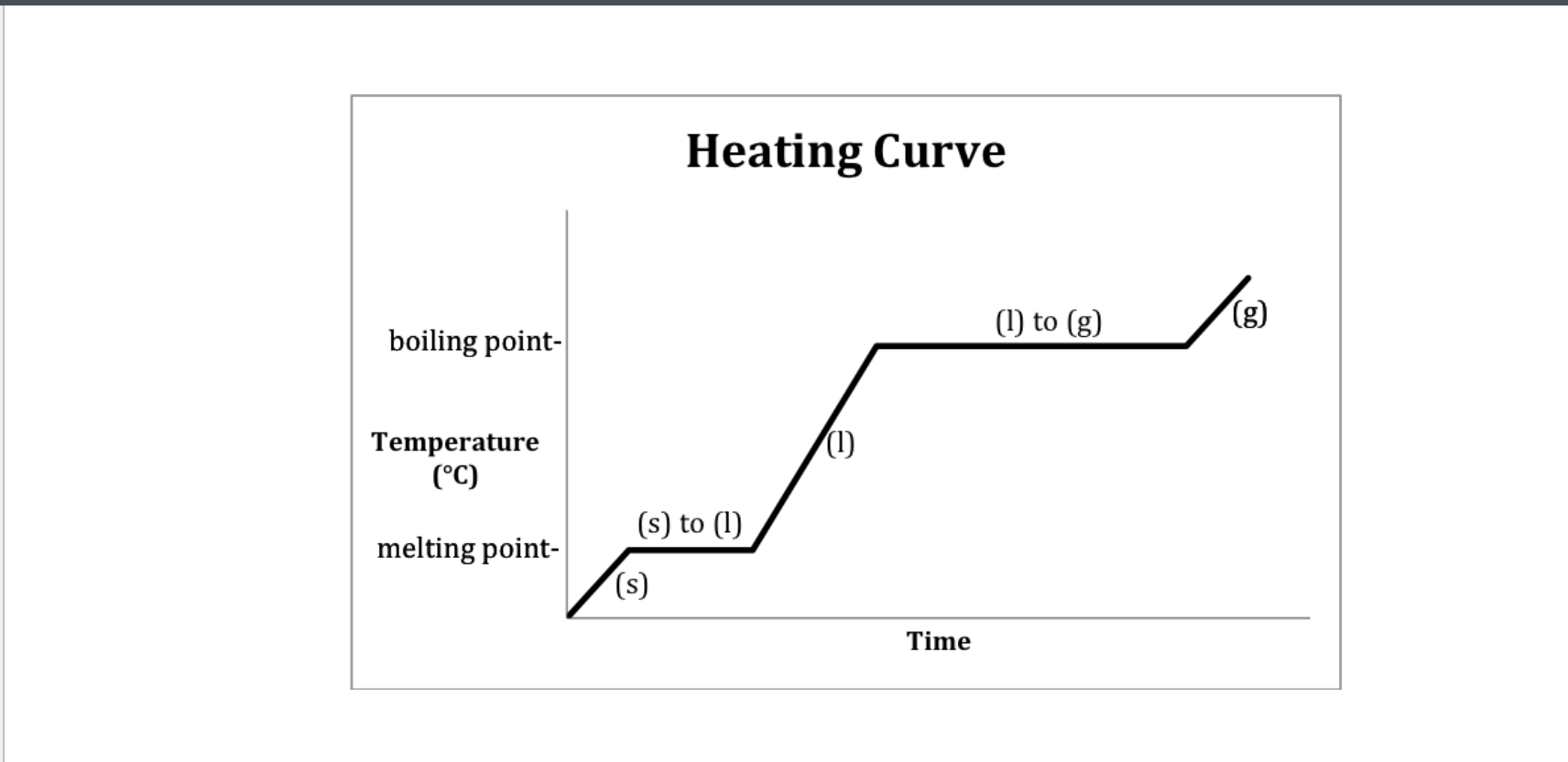
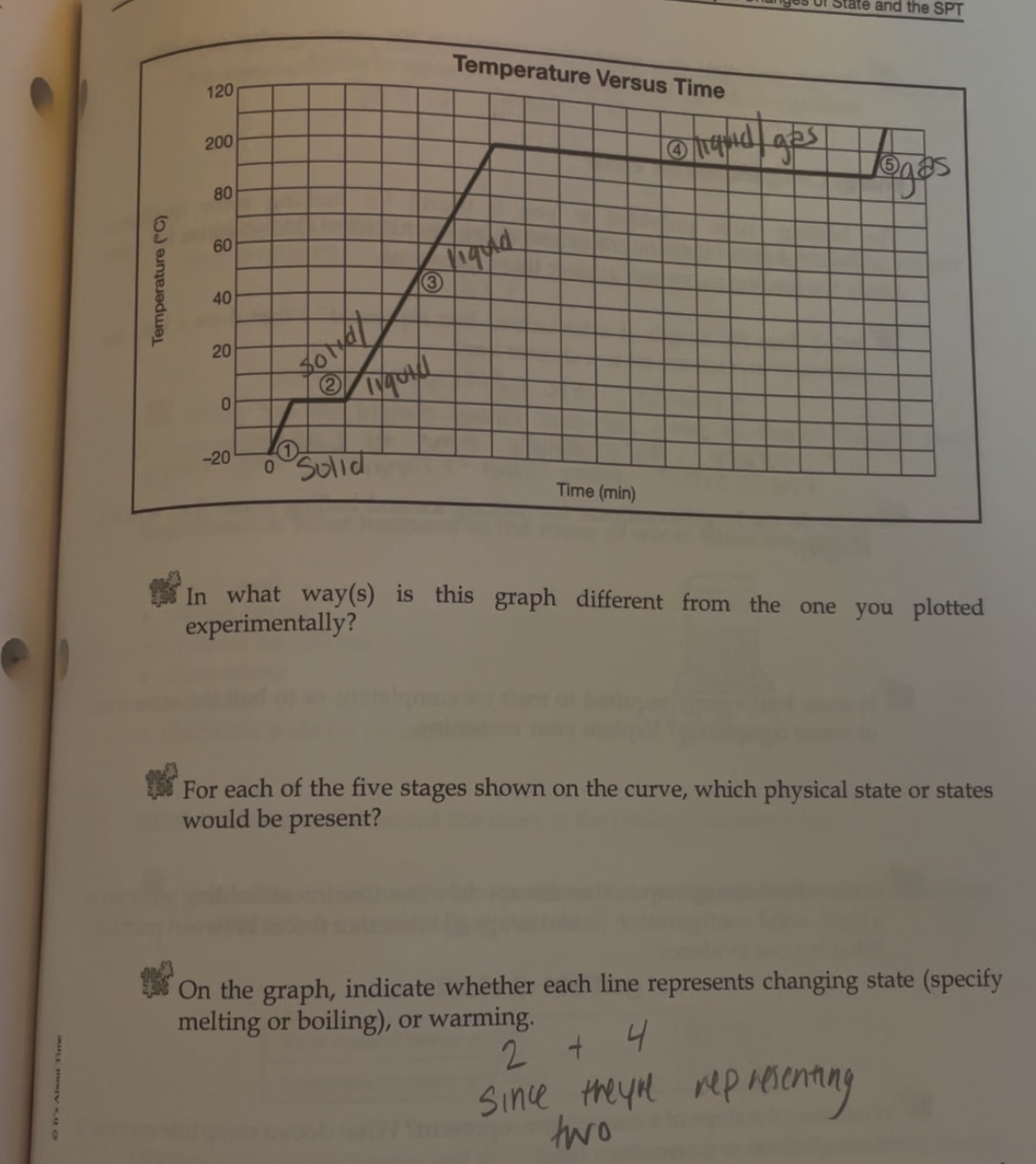
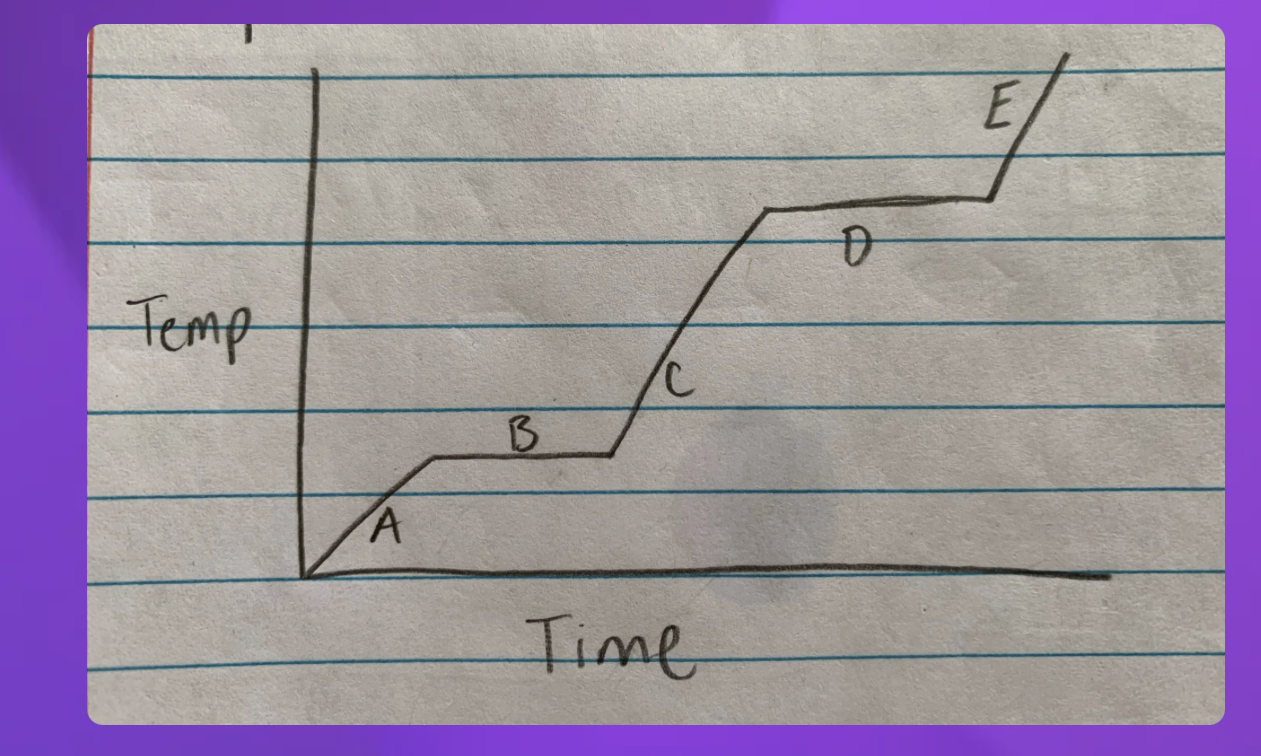
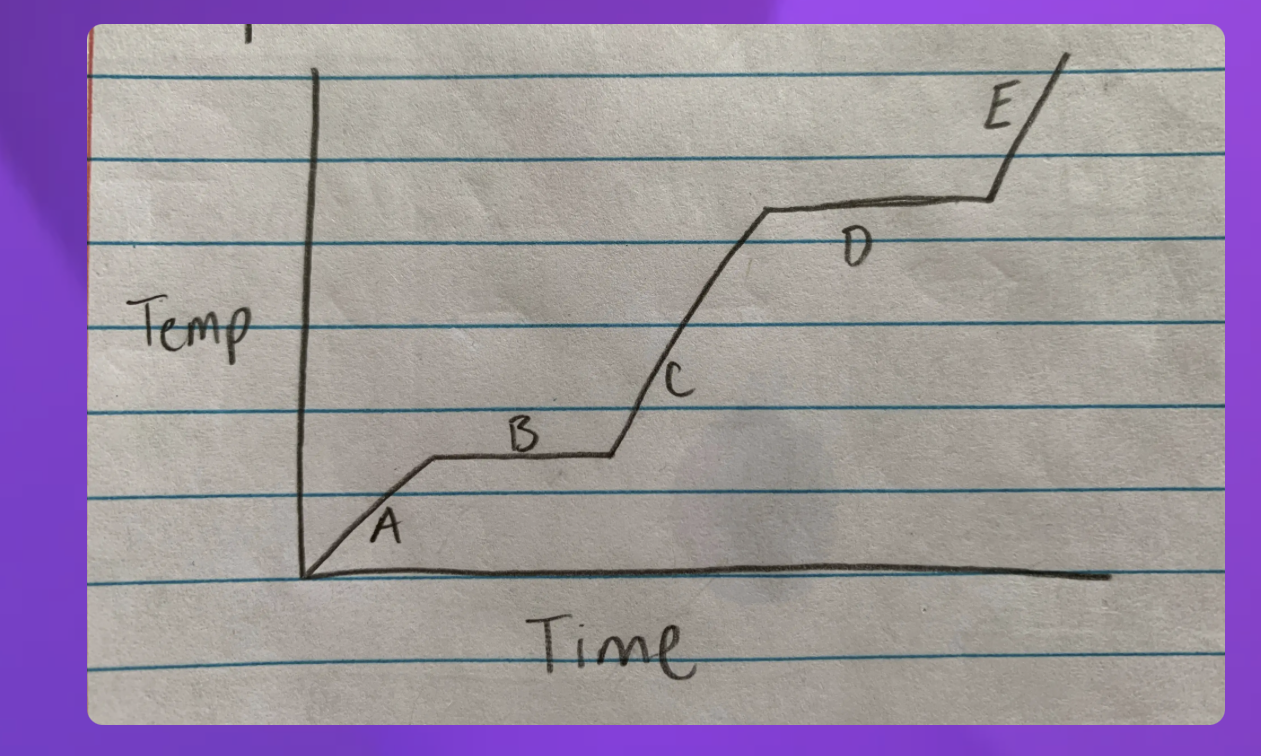
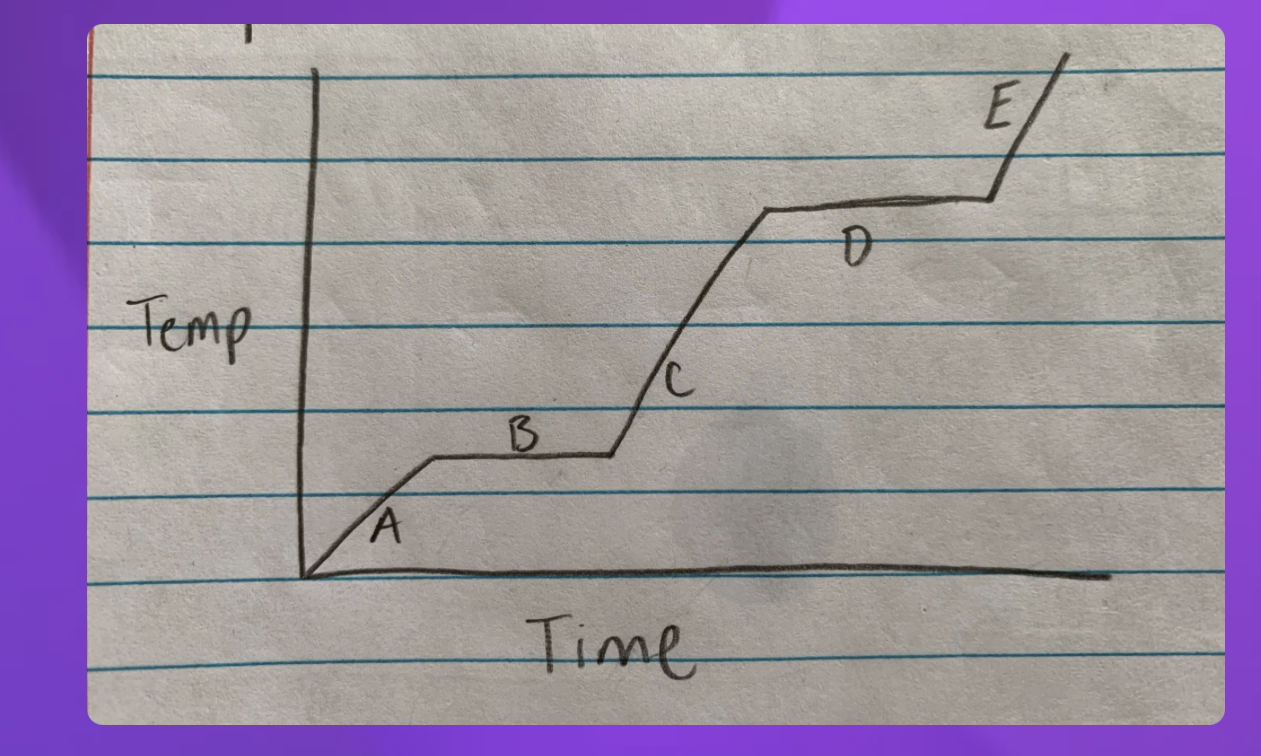
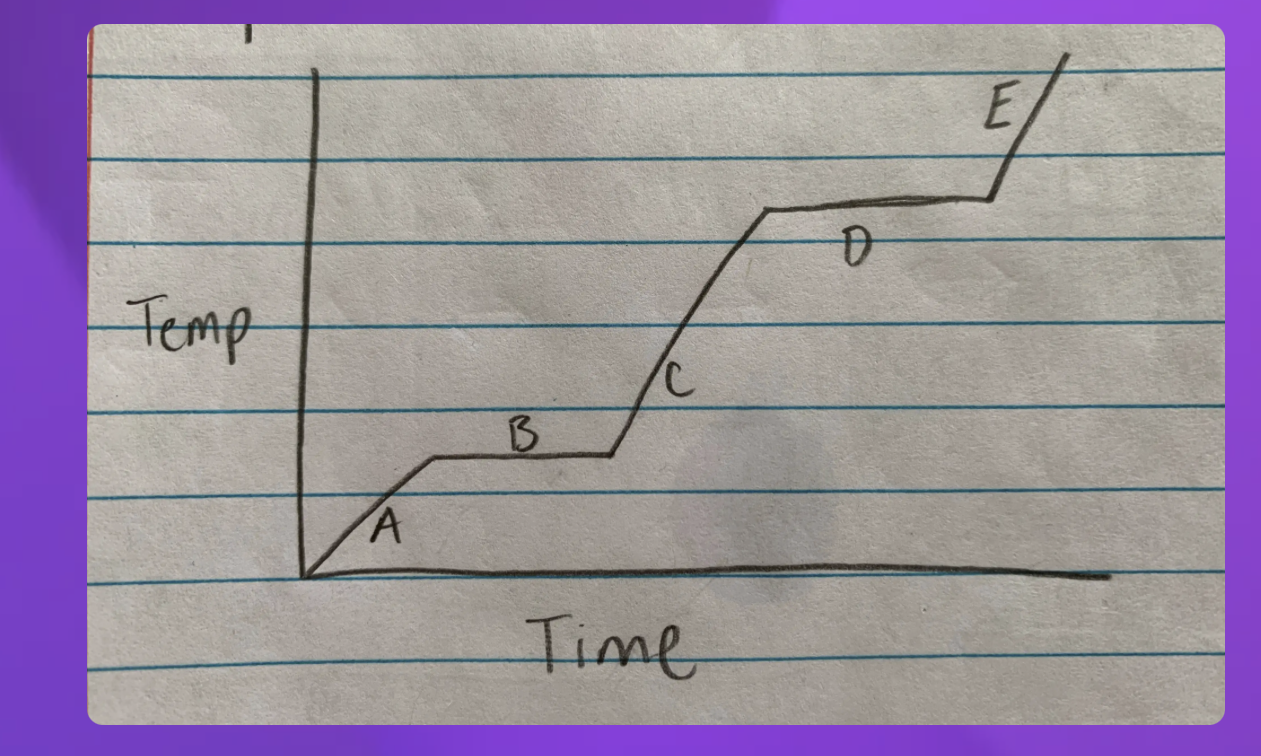
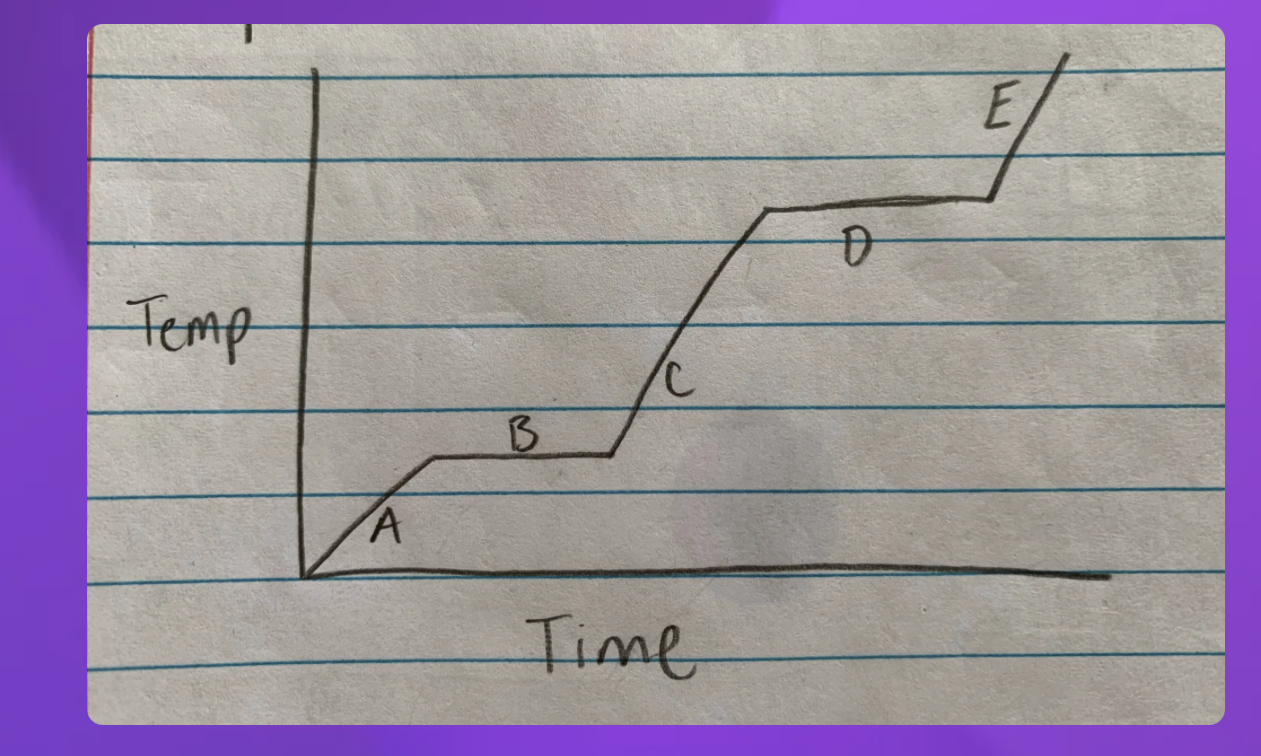
Heating curve
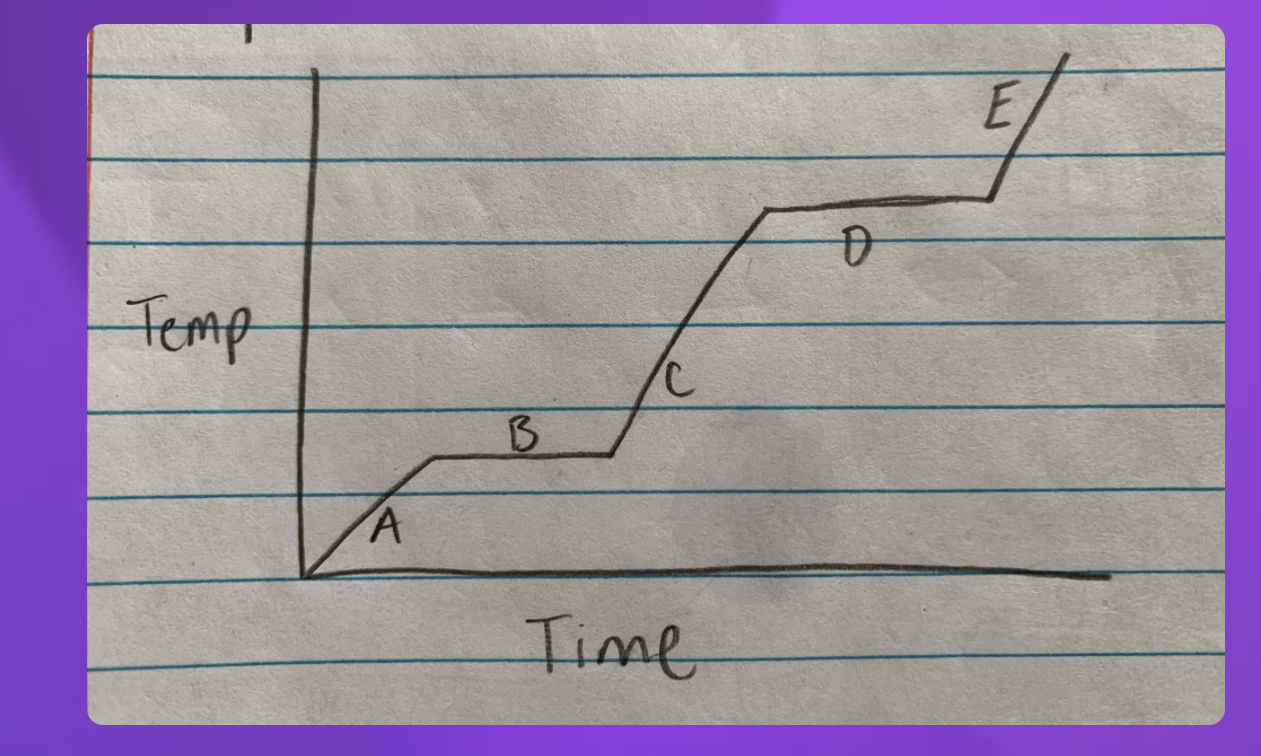
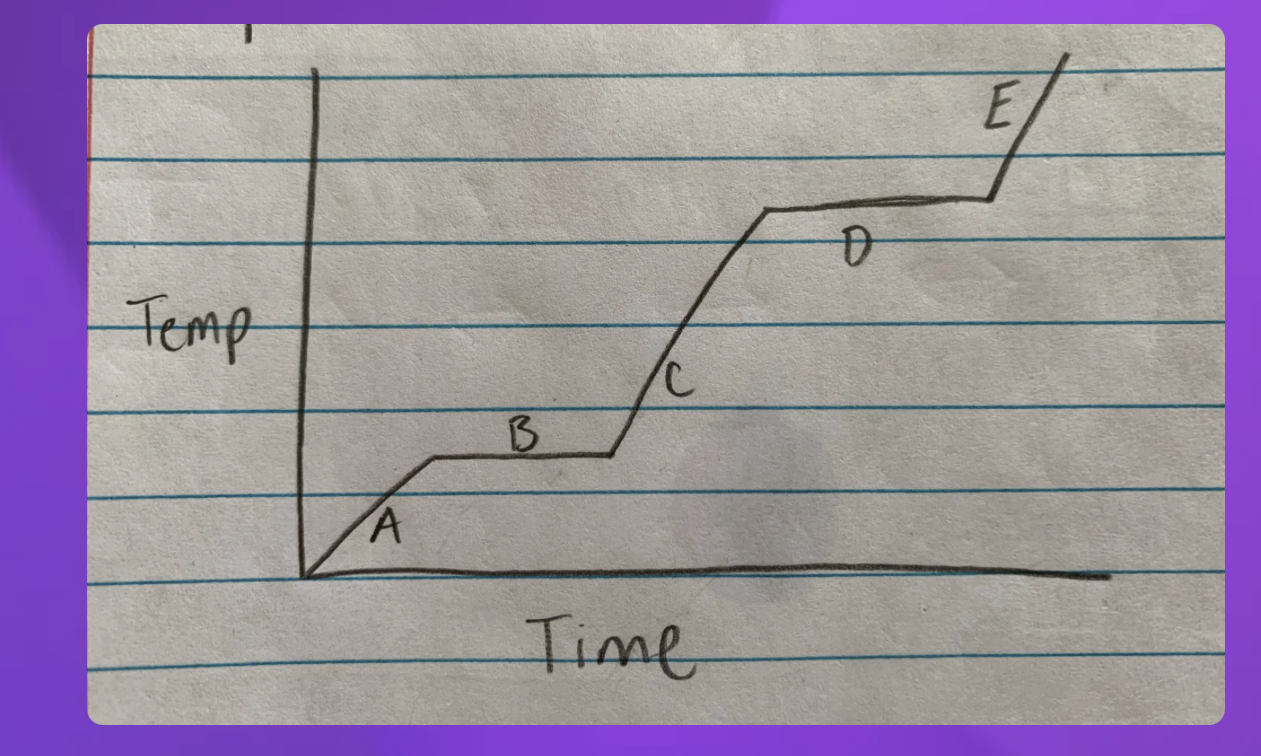
A- Solid B-Solid to liquid C-Liquid D-Liquid to gas E-Gas
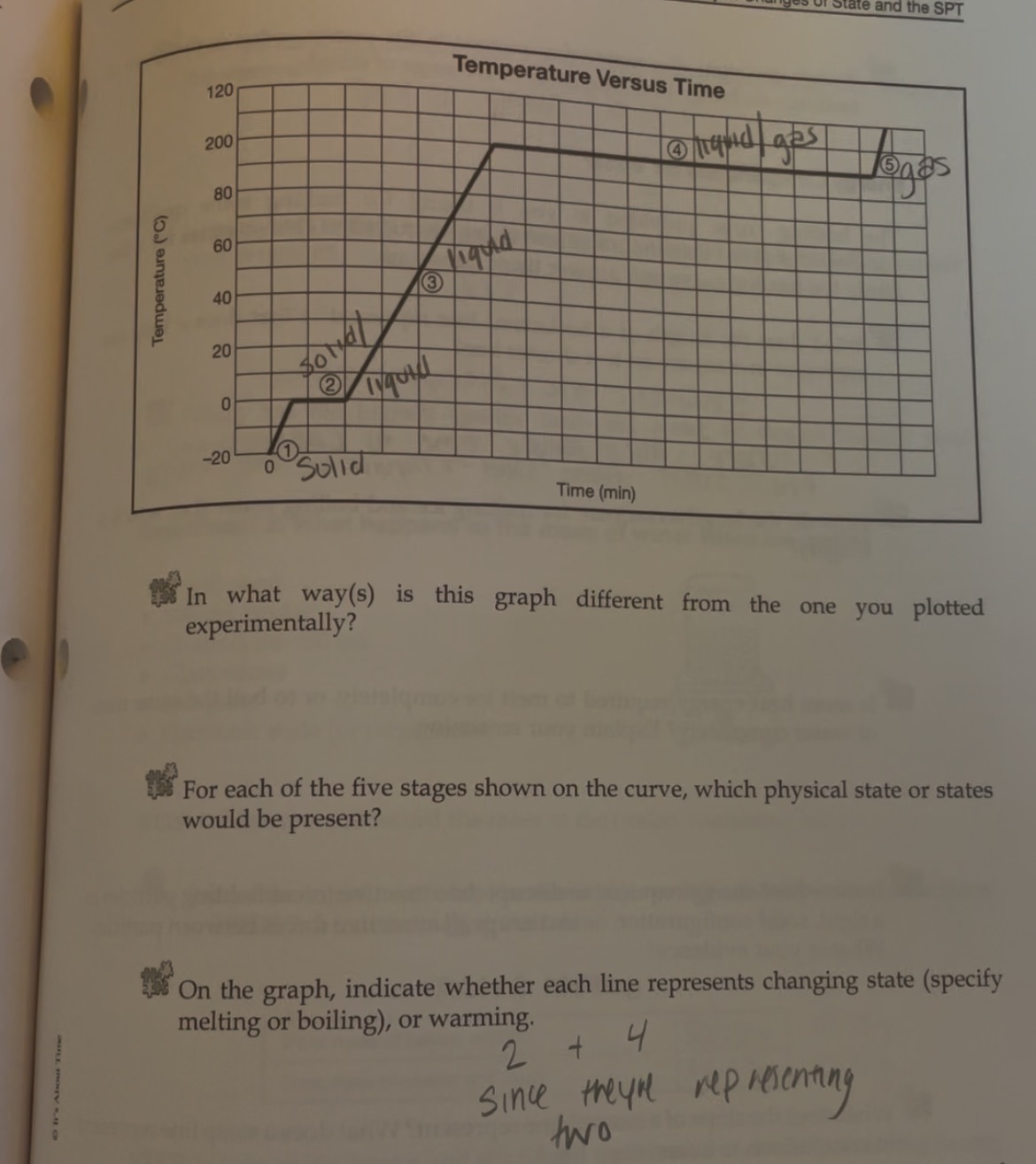
-Heating curve
-Indicate whether each line represents changing state (specifify melting or boilng)
2 and 4 since they are representing two
2-Solid to Liquid- Melting
4- Liquid to gas- Boiling
Sugar, Salt, Baking Soda
Sugar & Salt: These solids are denser than water. They sink and then dissolve, which increases the water's density.
Baking Soda: This solid is denser than water and sinks to the bottom. It is less soluble than salt or sugar and often fails to dissolve completely.

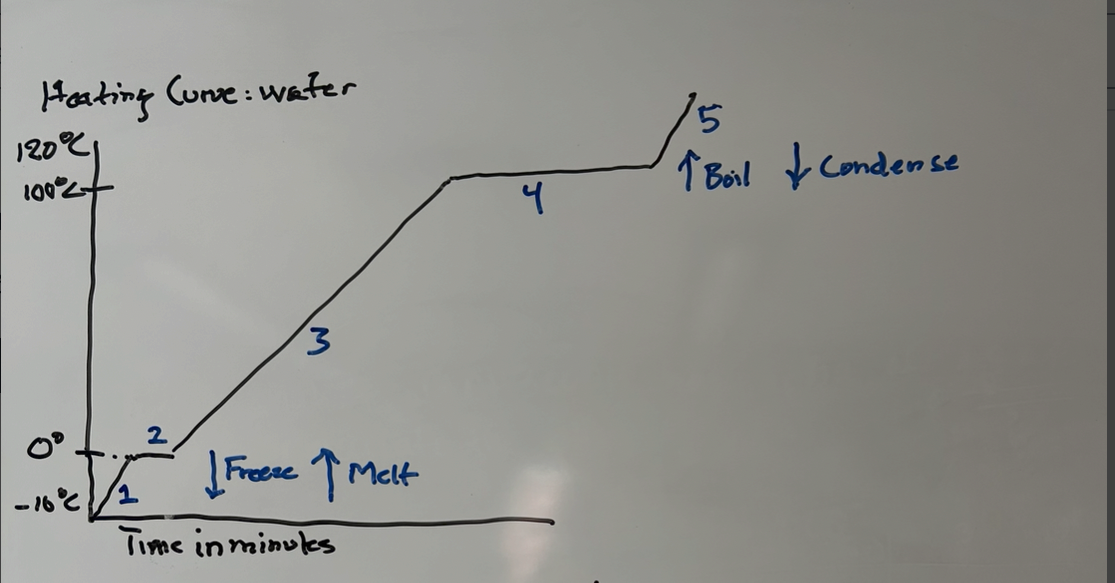
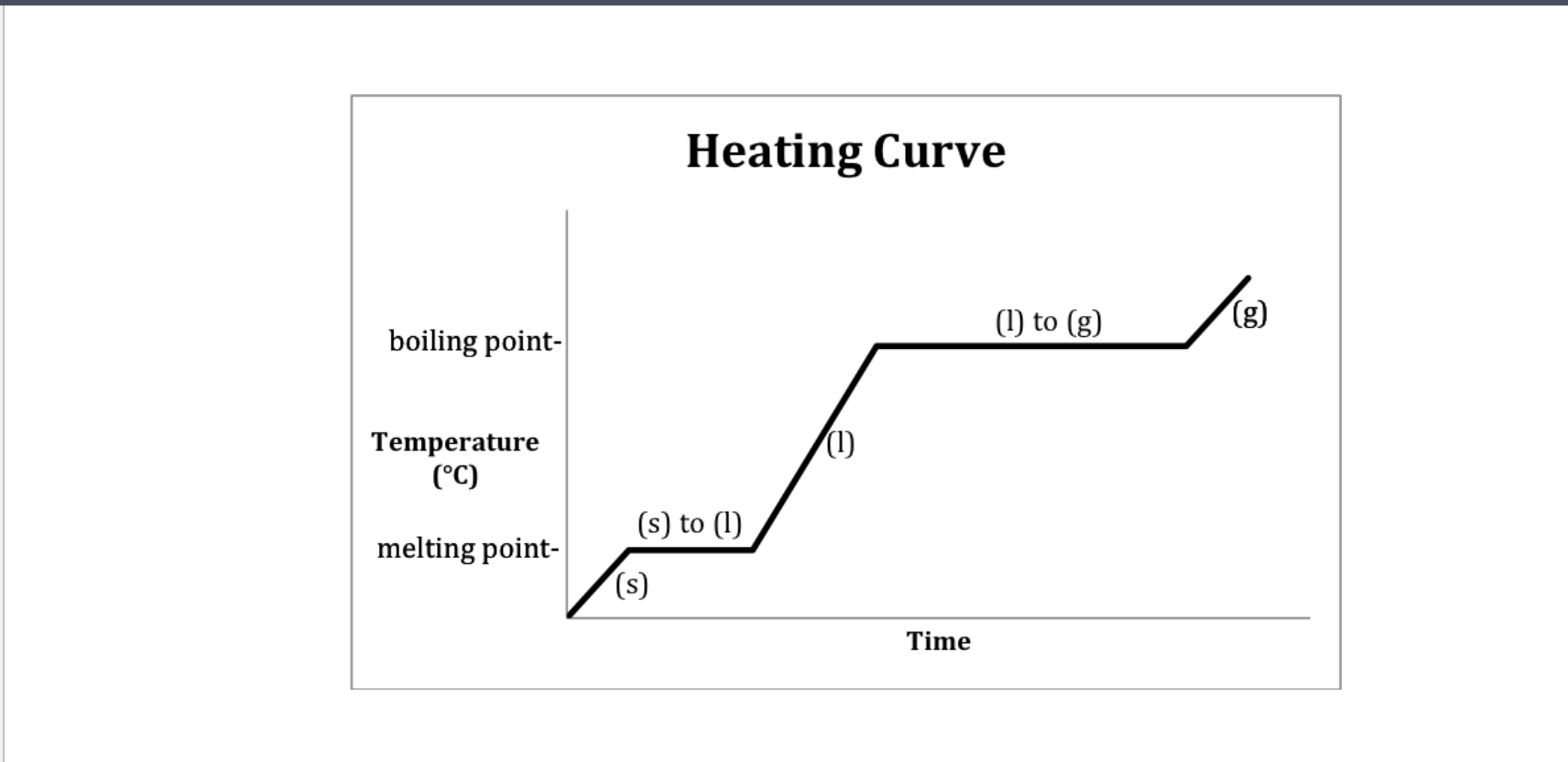
Heating curve-Water
Heating- 2-Melting 4-Boiling
Cooling- 2-Freezeing 4-Condensation (gas turning into liquid)
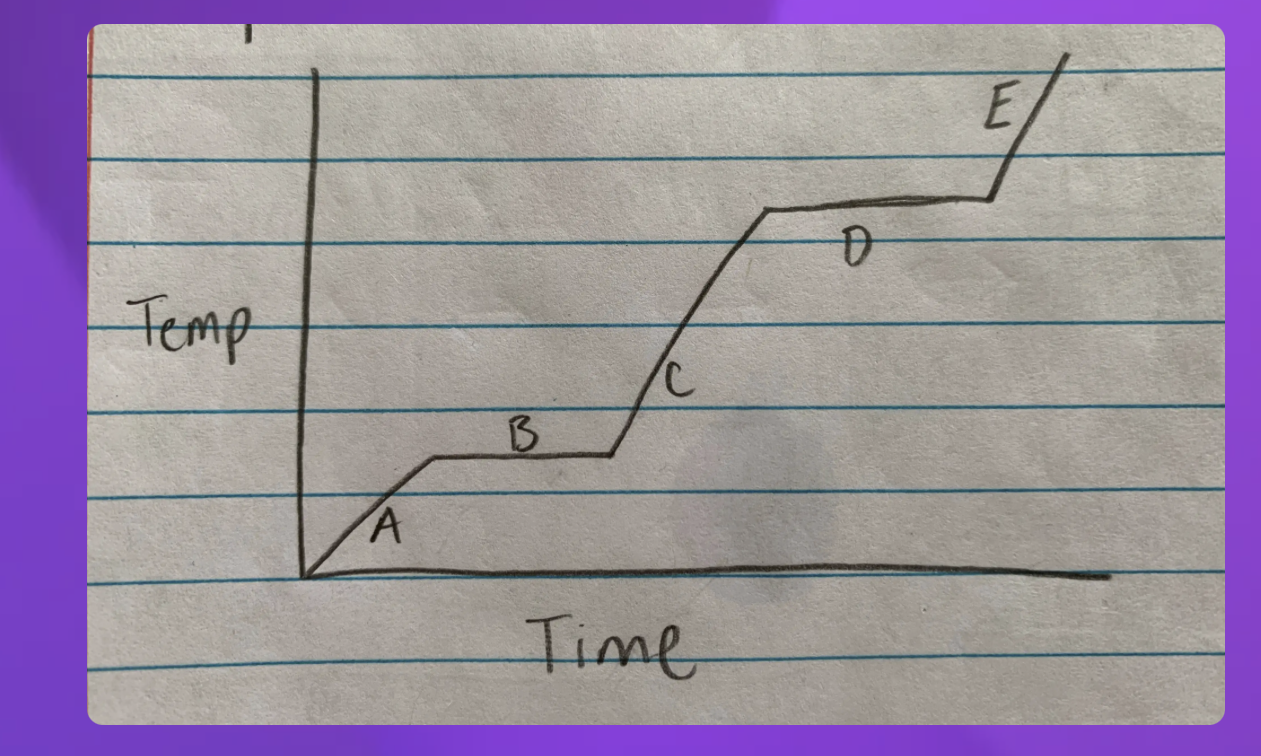
Which Point represents water at a solid?
A
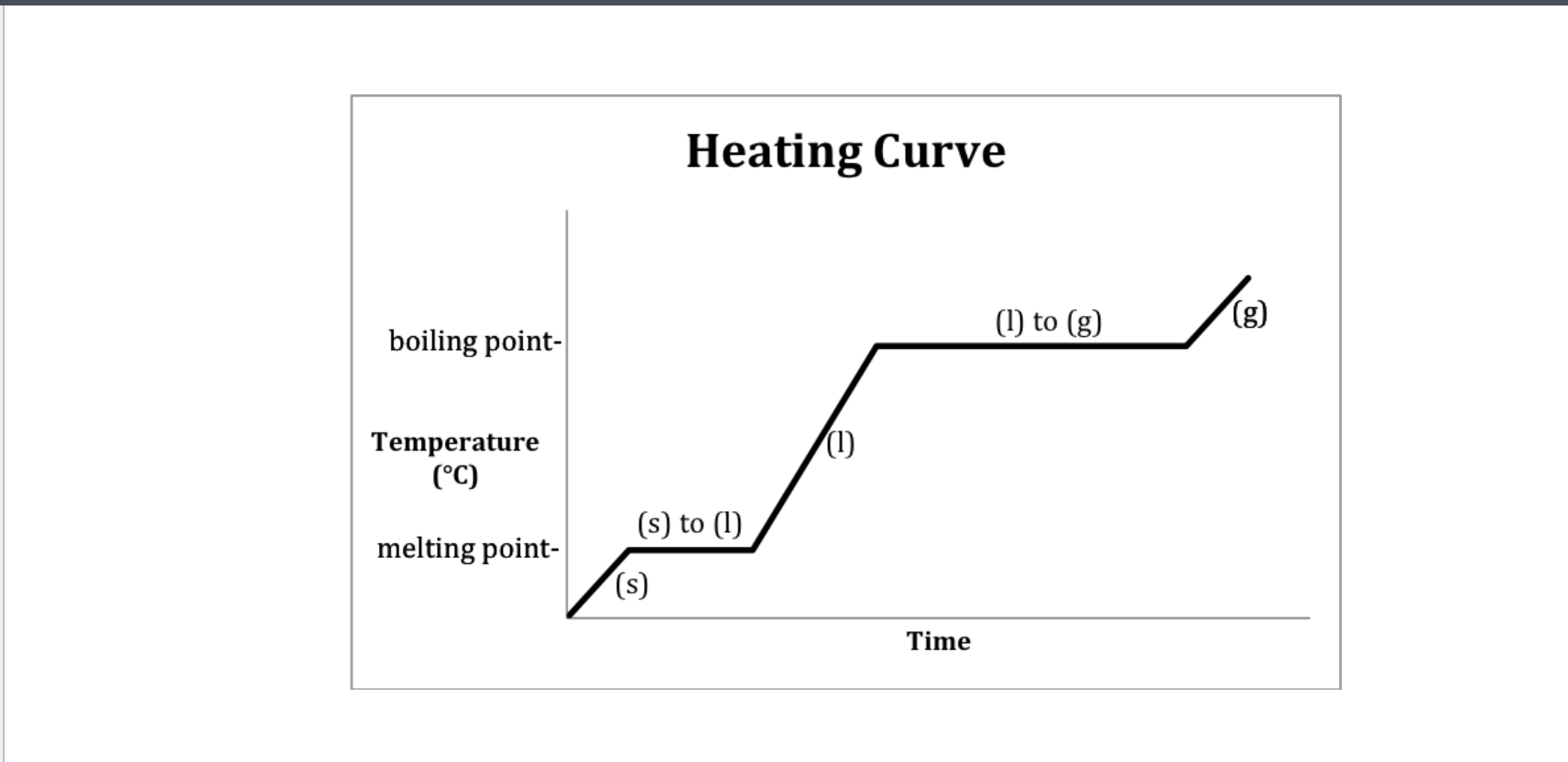
Which Point represents water as a liquid?
C
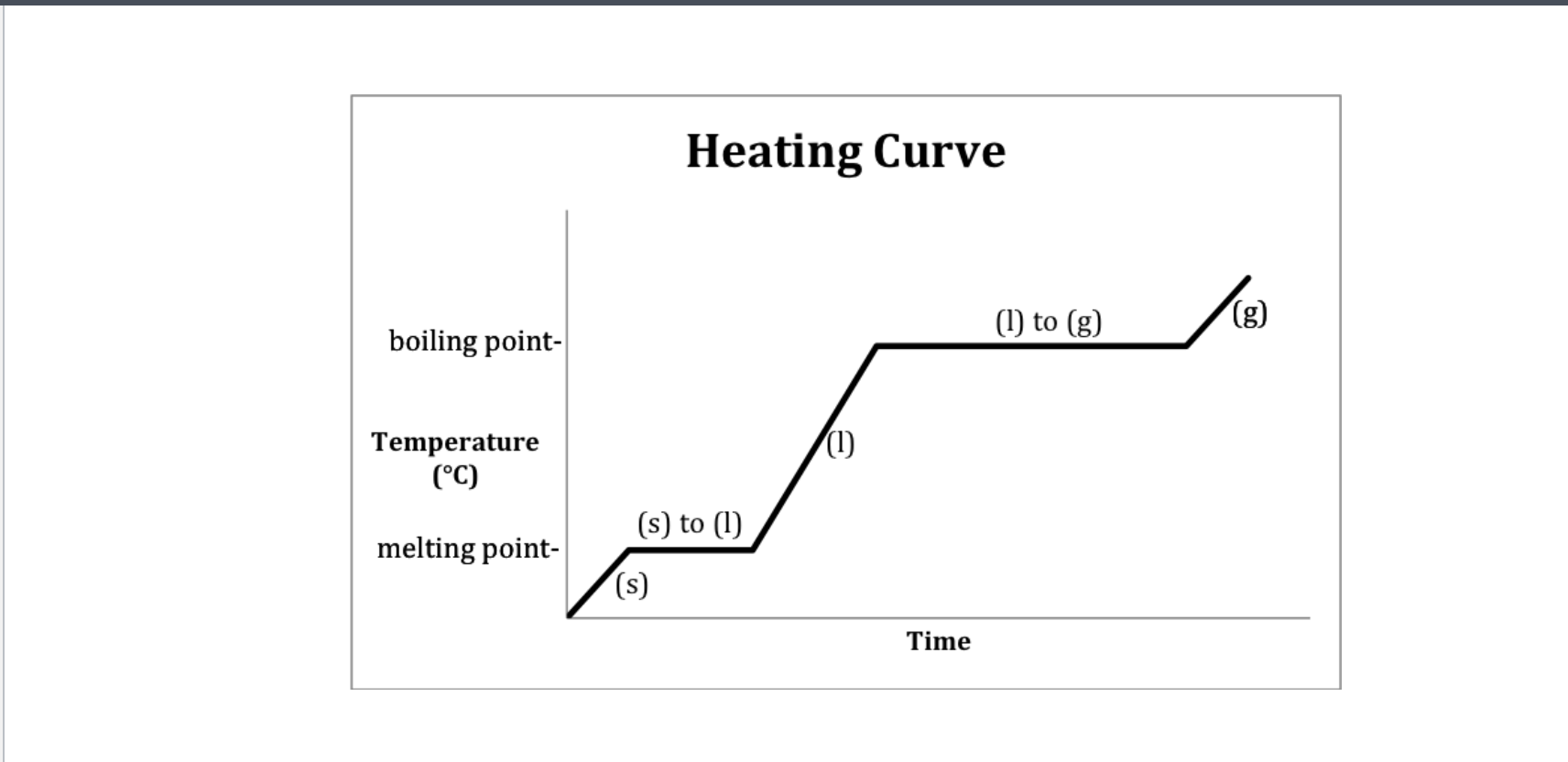
Which point represents water going from liquid to a gas? (boiling)
D
Which point represents water going from a solid to a liquid (melting point)
B
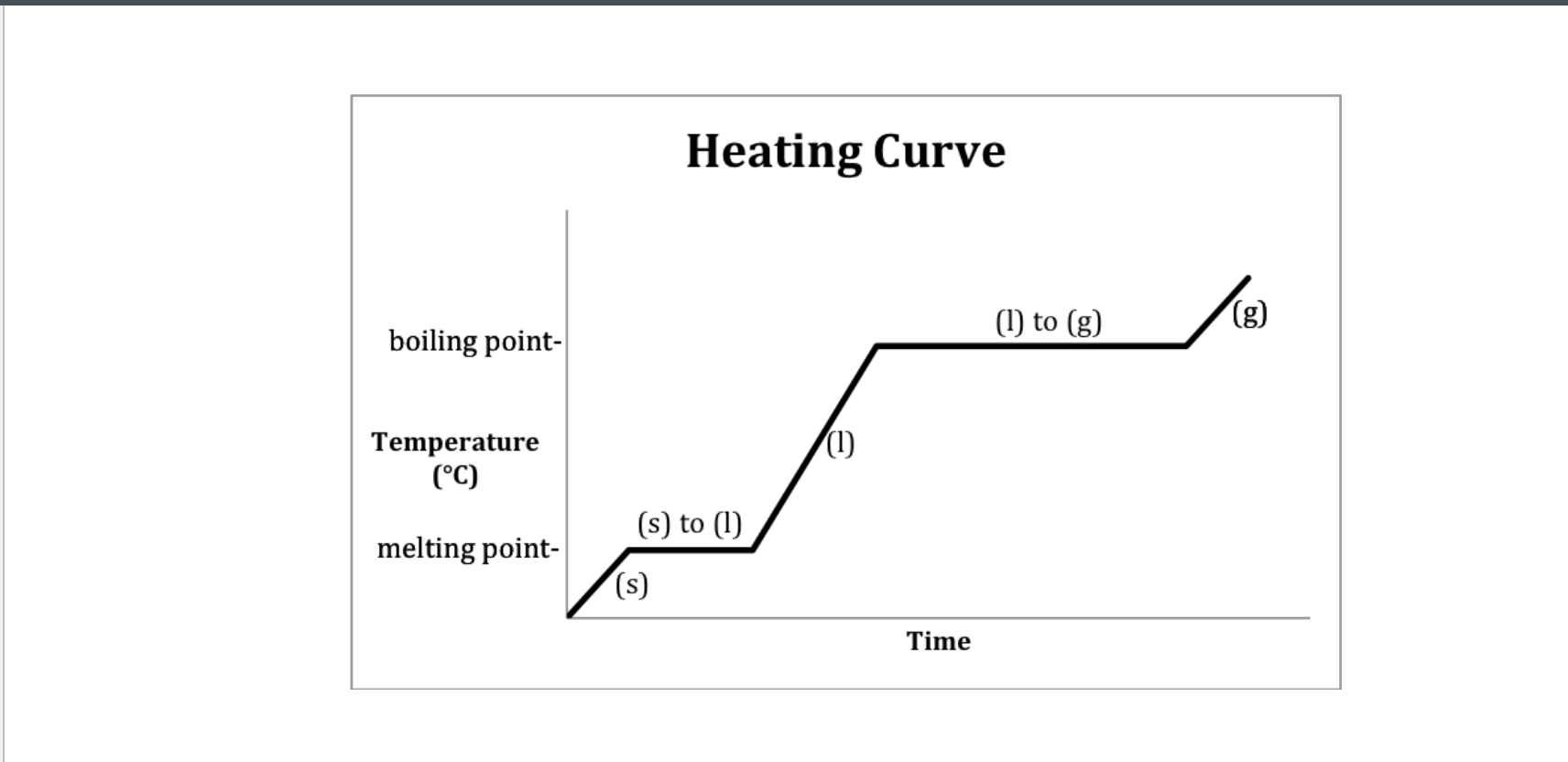
Which Point represents water as a gas?
E
Heat Diagram
B and D
element to the next
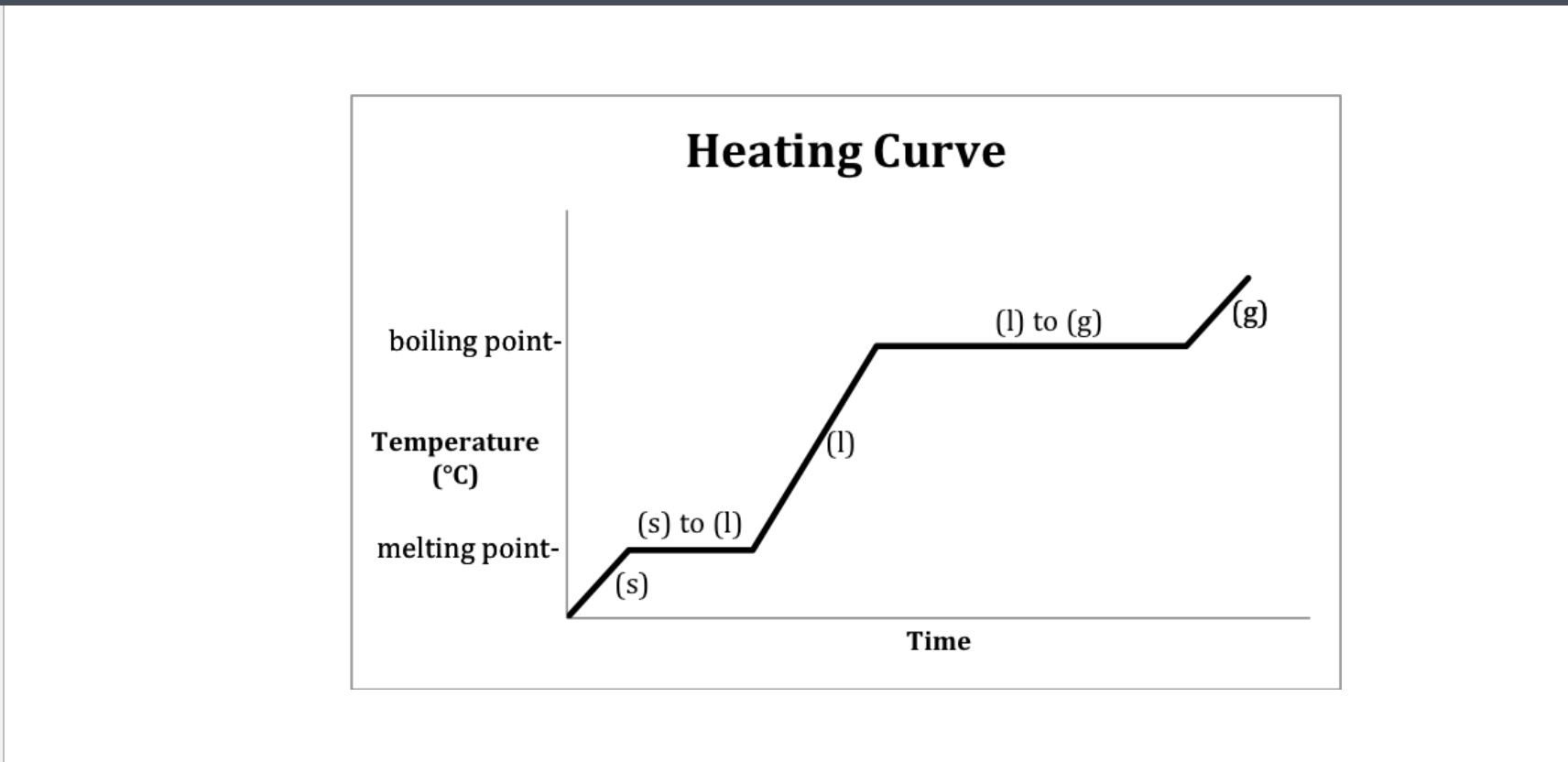
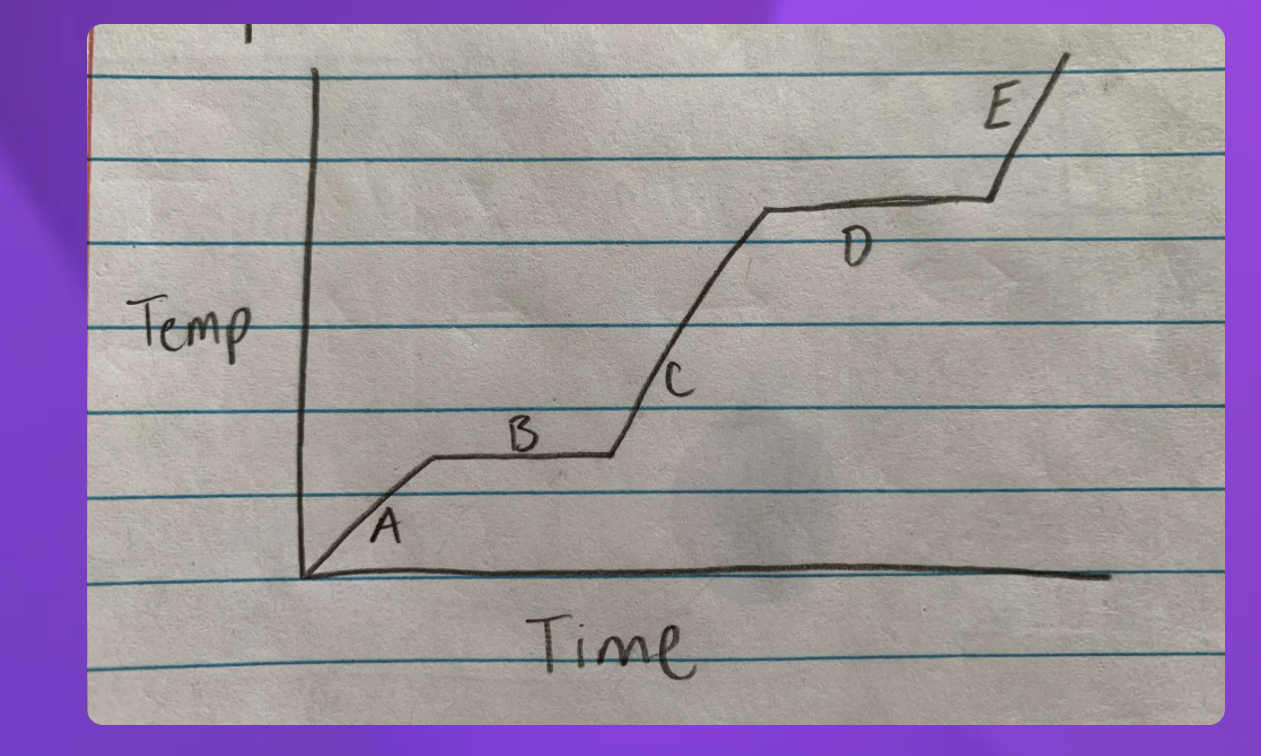
Heat digram- What is the hottest?
Gas
Heat Diagram- What is the coldest?
Solid
The graph can also go from a gas to a sold
2-Freezing (liquid to sold) and 4-condensation (gas to liquid)
Instead of melting 2-(Sold to liquid) and 4-Boiling (Liquid to gas)

If particles are cold they become?
Closer together (image on the right)

If particles are cold
Closer together- Very structured.
Think of an ice cube more a block and water more lose
Ice cube- particles close together. -water- Not as close together
Someone pours oil (density is 0.92 g/mL) and vinegar (density is 1,00 g/mL) into a bottle, which would float to the top?
Oil

Someone pours oil (density is 0.92 g/mL) and vinegar (density is 1,00 g/mL) into a bottle, which would float to the top?
-Oil would float to the top. Italian dressing example. Which one is more or less dense

The person adds a steel nut (its density is 7.75 g/ml) to the bottle where would it sit?
-Oils density is 0.92 g/mL -Vingars density is 1.00 g/mL
The bottom (Sinks)

The person adds a steel nut (its density is 7.75 g/ml) to the bottle where would it sit?
-Oils density is 0.92 g/mL -Vingars density is 1.00 g/mL (notes)
-Density is greater. See 7.75 much denser. Meaning it would sink.


The person adds a chunk of candle wax (its density is 0.95 g/mL) to the bottle where would it sit?
-Oils density is 0.92 g/mL -Vinegars density is 1.00 g/mL
The middle (floating on the vinegar)

The person adds a chunk of candle wax (its density is 0.95 g/mL) to the bottle where would it sit?
-Oils density is 0.92 g/mL -Vinegars density is 1.00 g/mL (notes)
-Chunk of candle wax- 0.95 less than the vinager, but still less than the oil- Would sit in the middle.

The person adds a piece of cork (its density is 0.32 g/mL) to the bottle where would it sit?
-Oils density is 0.92 g/mL -Vinegars density is 1.00 g/mL
The top (floating on both the oil and vinegar)

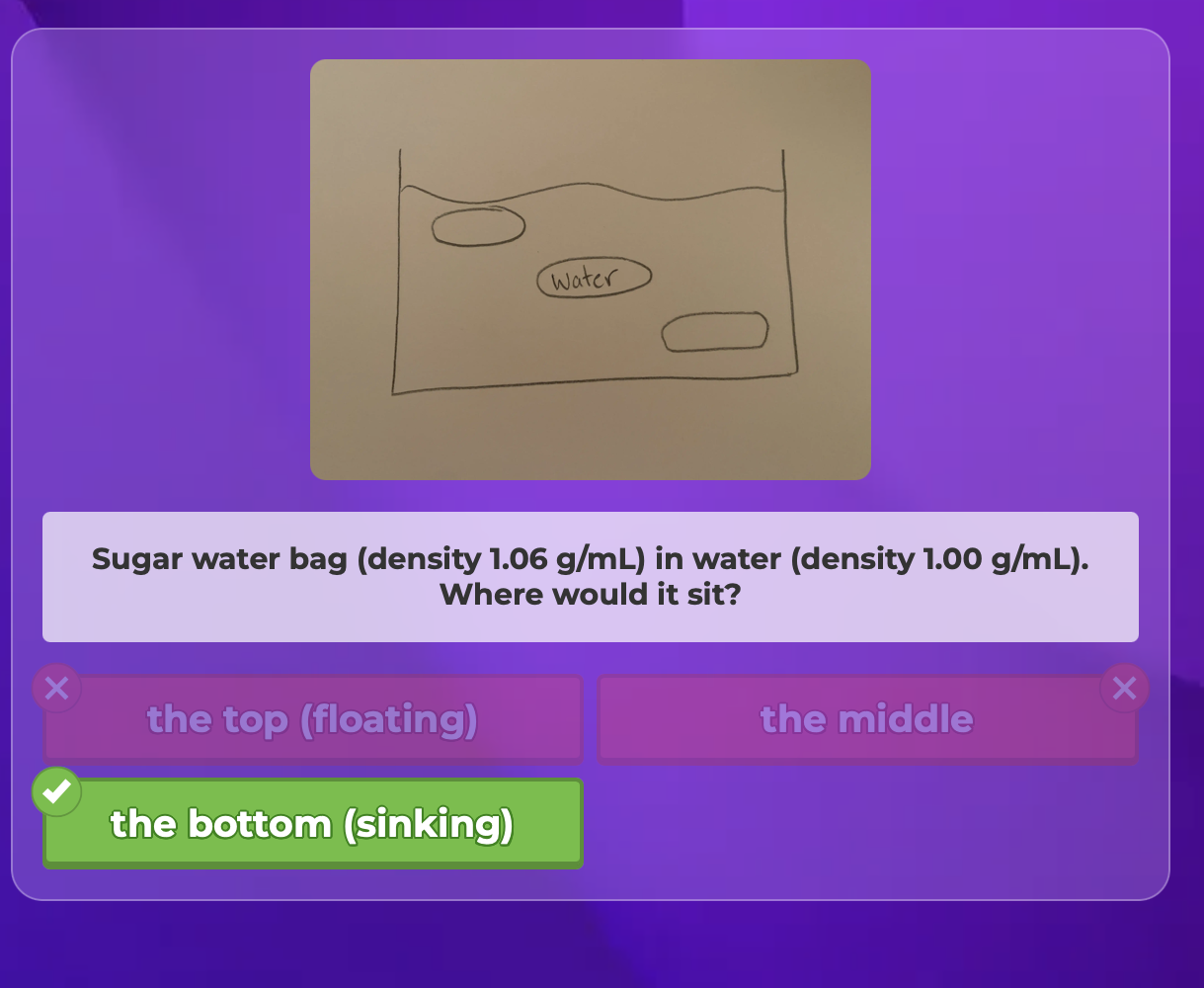
Three bags- One filled with water (density 1.00 g.mL)
One with sugar water (density 1.06 g/mL)
Rubbing achohl (density 0.82 g/mL)
-All bags are placed in a tub of water (density 1.00 g/mL)
Where would they all sit when placed in water?
Water bag (density 1.00 g/mL) in water (density 1.00 g/mL) Where would it sit?
The middle
-The water bag would sit in the middle because it is the same density as the water

Three bags- One filled with water (density 1.00 g.mL)
One with sugar water (density 1.06 g/mL)
Rubbing achohl (density 0.82 g/mL)
-All bags are placed in a tub of water (density 1.00 g/mL)
Where would they all sit when placed in water?
Sugar water bag (density 1.06 g/mL) in water (density 1.00 g/mL)
Where would it sit?
The bottom sinking
-The sugar water would sink to the bottom because it is more dense than water

Three bags- One filled with water (density 1.00 g.mL)
One with sugar water (density 1.06 g/mL)
Rubbing achohl (density 0.82 g/mL)
-All bags are placed in a tub of water (density 1.00 g/mL)
Where would they all sit when placed in water?
Rubbing achohl bag (density 0.82 g/mL) in water (density 1.00 g/mL). Where would it sit?
The top (floating)
-The rubbing acholh would float because it is less dense than water
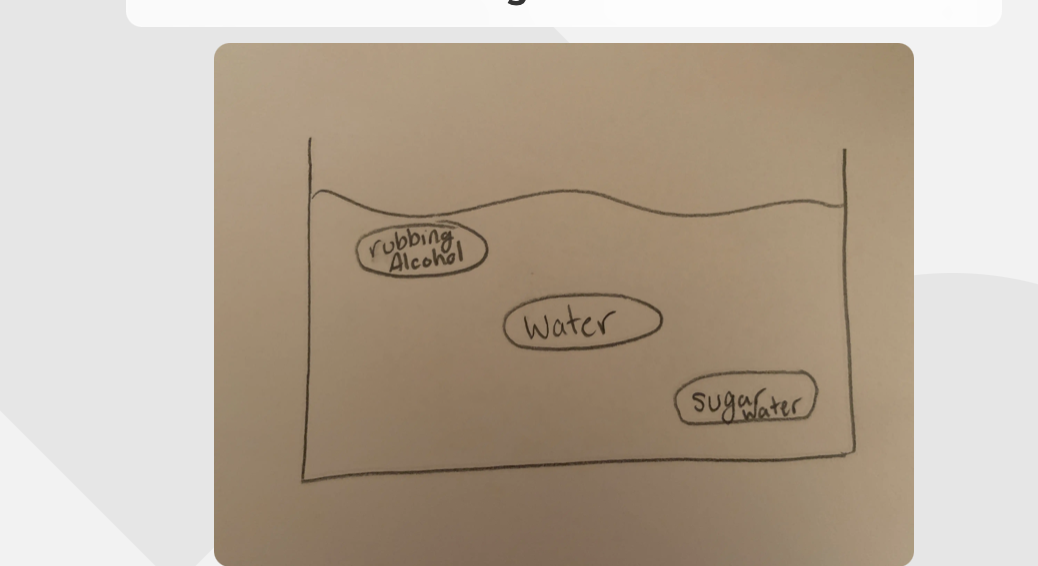
Where the bags would sit in water
-The rubbing acholh would float because it is less dense than water
-The water bag would sit in the middle because it is the same density as the water
-The sugar water would sink to the bottom because it is more dense than water
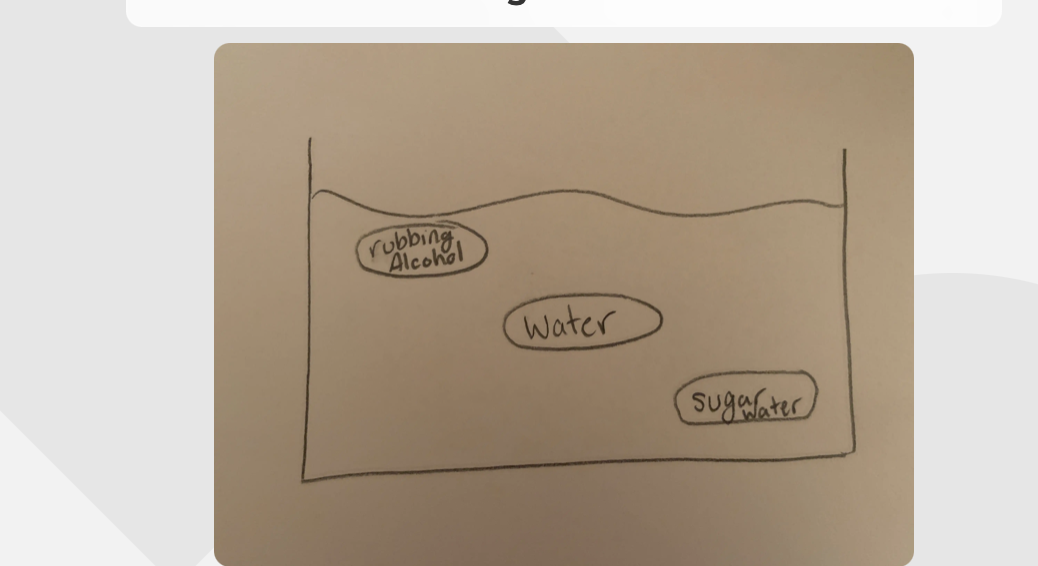
Three bags- One filled with water (density 1.00 g.mL)
One with sugar water (density 1.06 g/mL)
Rubbing achohl (density 0.82 g/mL)
-All bags are placed in a tub of water (density 1.00 g/mL)
Where would they all sit when placed in water?
-3 bags in a tub of water. -The middle- Can discount the weight of the bag, just the number of the density, bag of water in the middle.

Create a IO Heat diagram
-Need to know heat conduction interaction diagram. -Draw the diagram or label it. (the heat input output the example it gives is water on a hot day. Condemsation. If air was being cooled down heat is leaving the water droples heat energy is leaving the HTO-cooling down heat energy is leaving. Water in a glass- no. p.122 -Atomoshere cooling off on the side of the water glass.
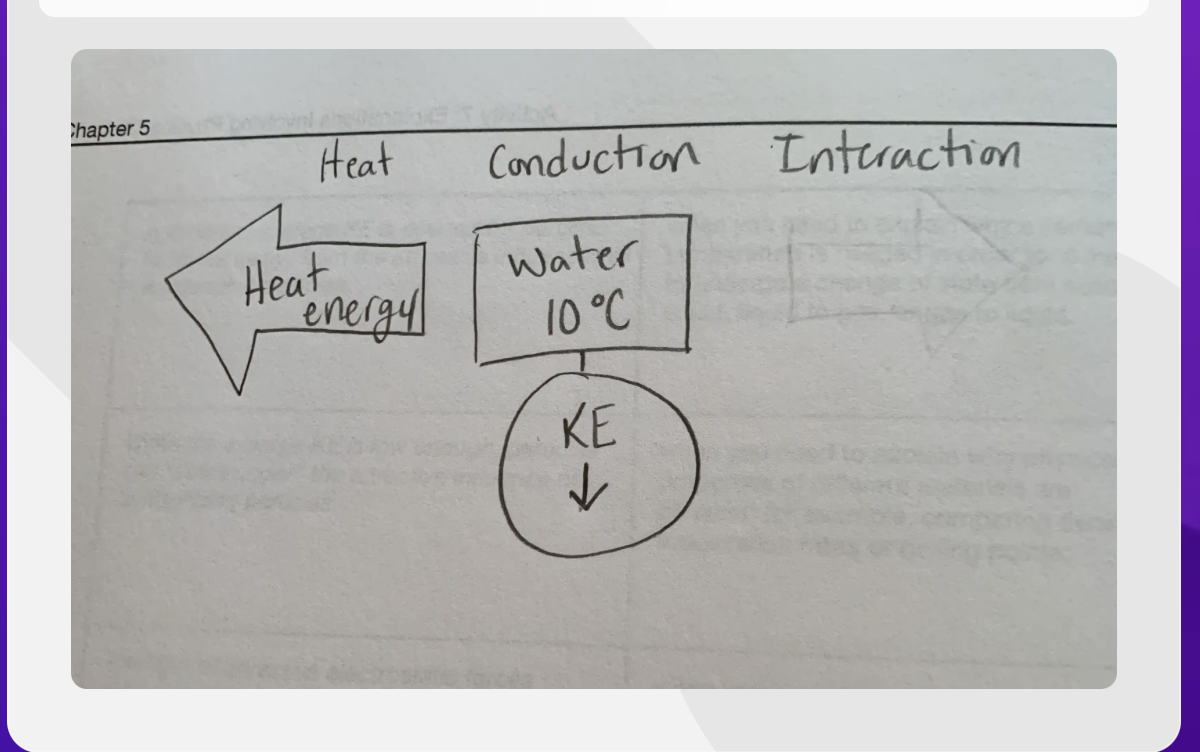
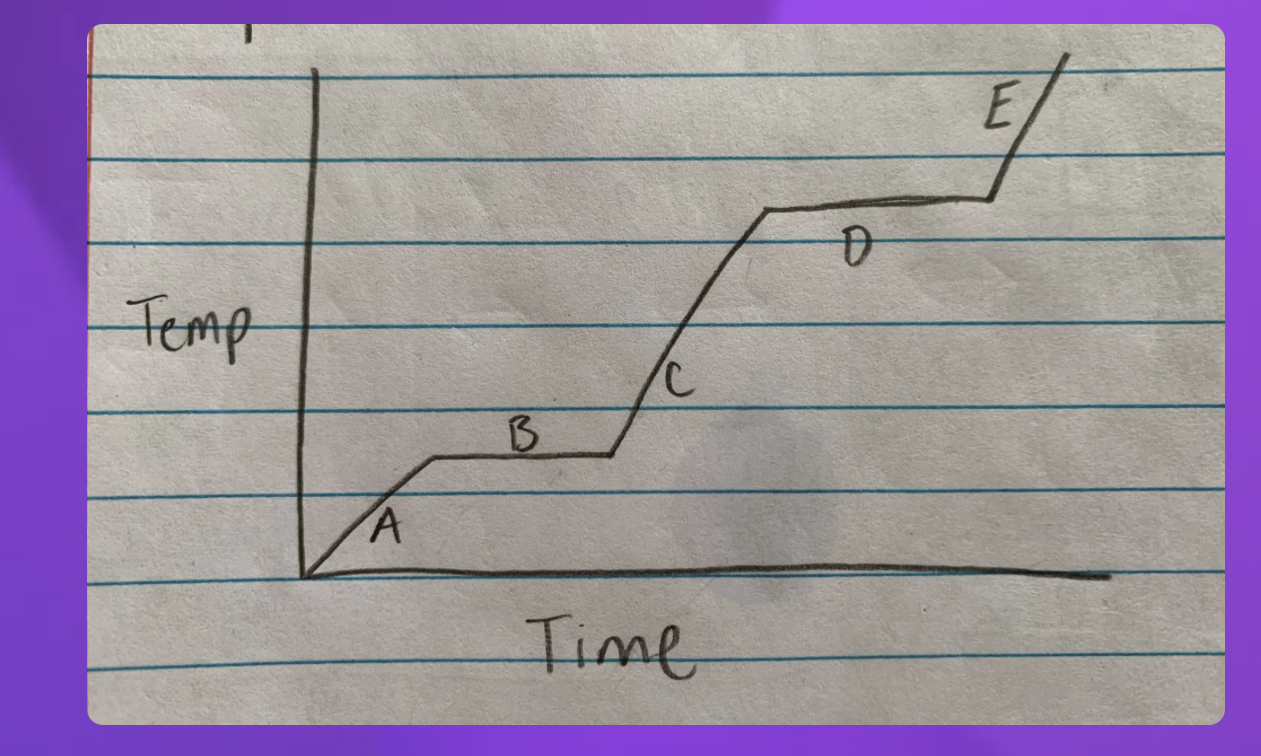
Heat energy- Phase changes
going up is the phase change
A- Going up-Soild
C- Going up- Liquid
E- Going up-Gas
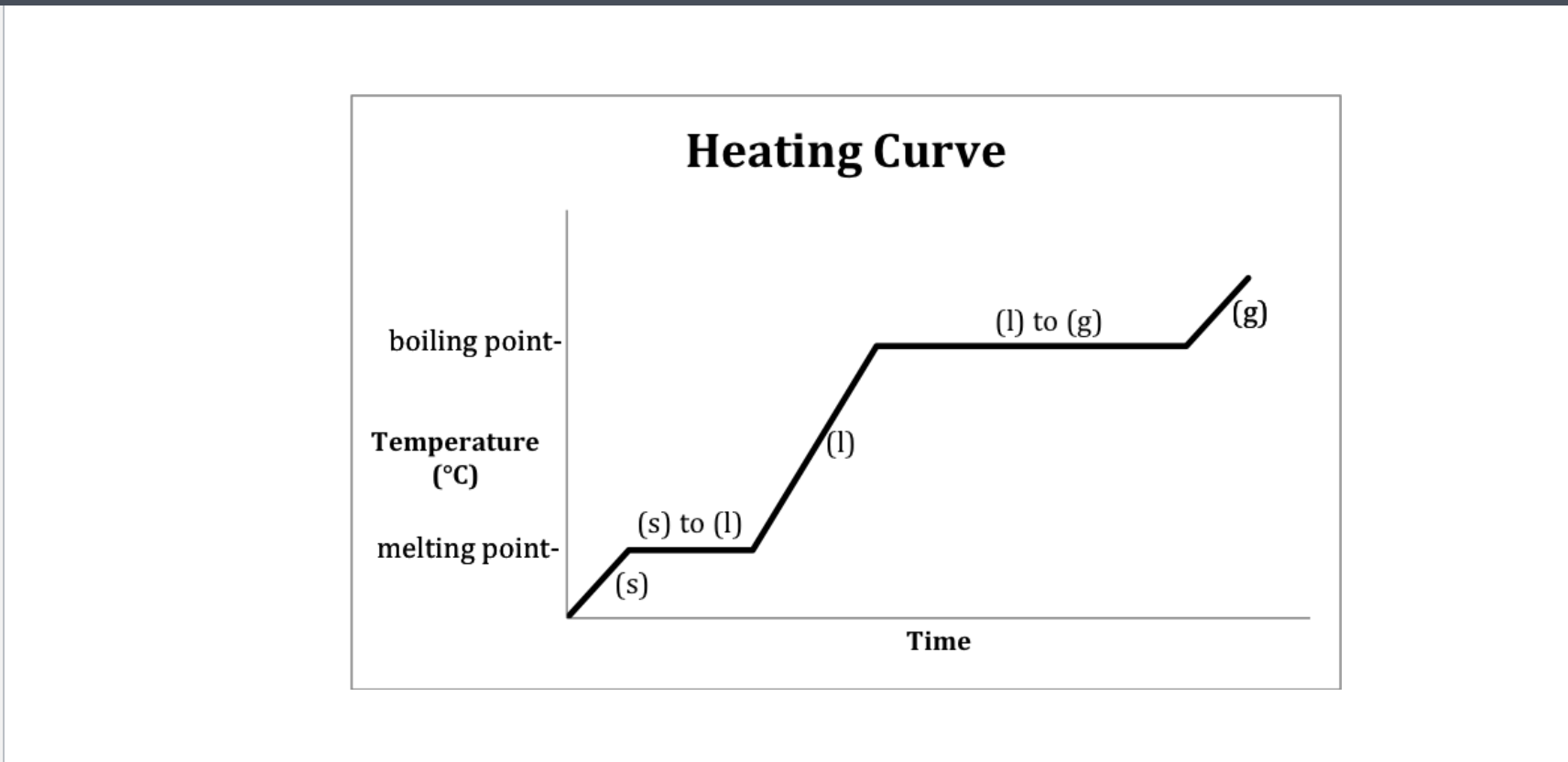

Condensation- For a heat IO diagram
Heat energy- arrow- facing away from conduction
Conduction- Water 10C- KE (kinetic energy) Going down
Interaction

Heat and Ball experiment