A&P Chapter 2: Chemistry of Living Things
1/58
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
59 Terms
Matter
anything that has mass/takes up space (ex: air, water, people, trees)
Elements
simplest form of pure substance made of one atom type (ex: oxygen, carbon, gold)
Compounds
pure substance made of two or more atom types (ex: water, salt, carbon dioxide)
Organization of matter (hierarchy)
Subatomic particles —> combine to form atoms —> bond together to make molecules —> make up living things
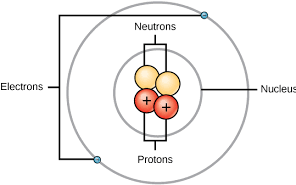
Characteristics of subatomic particles
Protons: +1 charge, found in nucleus, 1 amu
Neutrons: 0 charge, found in nucleus, 1 amu
Electrons: -1 charge, orbits around nucleus, much smaller
Atomic number
number of protons in an element (Z)
in a neutral element, the number of protons = electrons
Mass number
number of protons + neutrons in an atom (A)
Isotopes
atoms of an element that have the same atomic number, but different mass numbers (same protons, different neutrons)
How they’re used in biology/medicine: radioactive isotopes are used in biology to date geological fossils and in medicine to treat cancer, diagnose disease, find biochemical reaction/location, and DNA structure.
How do the location of electrons determine the chemical properties of an atom?
The number of electrons in the outer/valence shell determines how reactive an atom is (full valence shell = stable/non-reactive, incomplete valence shell = reactive/unstable) and the kinds of bonds it has. Basically how it interacts with other atoms.
[Different kinds of chemical bonds]
Ionic bond
forms when one atom gives an electron to another. the atom that loses the electron becomes positive, the one that gains it becomes negative.
ion: when an atom gains/loses electrons
cation: positive charge - atom loses electrons
anion: negative charge - atom gains electrons
[Different kinds of chemical bonds]
Covalent bonds
formed by sharing a pair of valence electrons = full outer shell
non-polar: equal sharing of electrons (balanced)
polar: unequal sharing of electrons (unbalanced)
[Different kinds of chemical bonds]
Hydrogen bonds
a weak bond between a partially positive hydrogen atom (already bonded) and a nearby electronegative atom
[Forms of energy in matter]
Kinetic energy
energy particles have from moving
ex: thermal (heat), electrical (electrons), radiant (x-rays)
[Forms of energy in matter]
Potential energy
stored energy based on position/arrangement of particles
ex: chemical (stores in bonds), nuclear (in the nucleus), elastic
[Forms of energy in matter]
Mechanical energy
sum of kinetic and potential energy
Energy interactions in the human body
energy from food/nutrients is converted into ATP through metabolism, which powers muscle movement, nerve signaling, and repair
[Chemical Reactions]
Metabolism
all chemical reactions in the body that sustain life (anabolism and catabolism make up metabolism)
[Chemical Reactions]
Anabolism
uses energy/ATP to build and repair body structures (consumes)
[Chemical Reactions]
Catabolism
breaks down molecules to release energy/ATP (releases)
[Chemical Reactions]
Activation Energy
the initial energy required to break bonds/start reaction
[Chemical Reactions]
Catalysts (enzymes)
increase the speed of a reaction (can be used in multiple reactions)
Structure of water and why it’s shape makes it a polar molecule
water is made of one oxygen atom covalently bonded to two hydrogen atoms in a V-shape. it’s polar because of uneven electron density/charge since the oxygen has a stronger attraction.
Chemical formula for creation of water from hydrogen and oxygen
2 H2(g) + O2(g) → 2 H2O(g)
the reactants are the hydrogen and oxygen gas
The Four Life-Supporting Properties of Water (and how they affect human life)
Cohesion of water: water molecules stick together via hydrogen bonds (cohesion) and stick to other surfaces = high surface tension (impact on humans: cohesion allows blood to travel against gravity + high surface tension helps lung function)
Resistance to change in temperature: takes more energy/heat to increase temperature. water can also moderate temperature. (impact on humans: prevents us from overheating during exercise)
Frozen Water Floats (Density): water is less dense when frozen = floats. (impact on humans: the surface of lakes freeze to allow aquatic life underneath to survive, we rely on those)
Solvent for Life: water’s polarity allows it to dissolve most substances (impact on humans: water is the solvent for blood, which transports nutrients and minerals)
Chemical properties of acids, bases, and neutral solutions + buffers
Acids: compound that releases H+ to solution (pH<7)
Bases: compound that accepts H+/releases OH-, removes H+ from a solution (pH>7)
Neutral solutions: have equal amounts of H+ and OH- ions (pH=7)
Buffers: help resist changes in pH by counteracting and making it neutral (ex: accepts H+ ions when they’re in excess and donates H+ ions when they’re depleted)
Special bonding properties of carbon (allows it to form an endless variety of organic molecules)
carbon has four covalent bonds (tetravalency), bonds with itself (catenation), can make multiple kinds of bonds (single, double, triple), and bonds with many elements.
The 4 types of macromolecules
carbohydrates, proteins, nucleic acids, and lipds
Carbohydrates
formed from building blocks/monomers of glucose. used as fuel/building material.
[Carbohydrates]
Monosaccharides
single sugars (monomers) that offer immediate energy, also used as building blocks. Ex: glucose, ribose, glyceraldehyde.
[Carbohydrates]
Disaccharides
double sugar made of two monosaccharides covalently bonded (condensation), used for energy source and energy transport. Ex: maltose, lactose, sucrose.
[Carbohydrates]
Polysaccharides
long polymers made of monosaccharides that are used for energy storage (starch and glycogen) and structural support (cellulose and chitin).
Lipids
hydrophobic compounds that are insoluble in water but dissolve in non-polar solvents. Consist of triglycerides (glycerol + fatty acids), phospholipids, steroids, and waxes.
Structure/properties of Saturated VS Unsaturated Fatty Acids
Saturated: have only a single C-C bond in their fatty acid tails. solid at room temperature
Unsaturated: have one or more double bonds between carbons = “kinks” in tails. Liquids (oils) at room temperature
Steroids VS Anabolic Steroids
Steroids: treat inflammation and conditions like asthma/allergies
Anabolic steroids: synthetic drugs that mimic testosterone to treat hormone problems or muscle loss (can be dangerous because it’s incorrectly used to increase muscle mass = liver/kidney/cardiovascular damage, hormone imbalance)
Proteins
the molecular tools of cells composed of monomers of amino acids
Structure of Proteins
Primary Structure: unique sequence of amino acids in proteins - determined by genes
Secondary Structure: regular repeated coiling/folding of polypeptides. Contributes to conformation.
Tertiary Structure: 3-D shape of a single polypeptide, formed by interactions between R groups/side chains.
Quaternary Structure: multiple folded polypeptides formed into one unit.
Primary VS Final: primary is the linear sequence with the genetic code that determines future folding. The final shape is 3-D, functional, and determines the protein's purpose.
DNA (type of nucleic acid)
stable, double-stranded, long helix that has deoxyribose sugar and thymine, used for storing genetic information.
RNA (type of nucleic acid)
temporary, single-stranded, short molecule that has ribose sugar and uracil, used for protein synthesis/regulation.
Why Lactose Intolerance Evolved Differently Throughout the World
evolved differently due to convergent evolution, where independent mutations arose in distinct human populations to enable adult milk consumption (different diet/evolutionary pressures)
[Roots]
Aqua
water (aqueous: a type of solution where water is the solvent)
[Roots]
Valent
strength (covalent bond: an attraction between atoms that share one or more pairs of outer-shell electrons)
[Roots]
Iso
equal (isotope: an element having the same number of protons and electrons but a different number of neutrons)
[Roots]
Neutr
neither (neutron: a subatomic particle with a neutral electrical charge)
[Roots]
Sacchar
sugar (disaccharide: two monosaccharides joined together)
[Roots]
Glyco
sweet
[Roots]
Gen
producing (glycogen: a polysaccharide sugar used to store energy)
[Roots]
Hydro
water
[Roots]
Lyse
break (hydrolysis: breaking chemical bonds by adding water)
[Roots]
Philic
loving (hydrophilic: water-loving property of a molecule)
[Roots]
Phobos
fearing (hydrophobic: water-repelling property of a molecule)
[Roots]
Sclero
hard (atherosclerosis: hardening of the arteries)
[Quiz Question]
Match the compounds with the type of chemical bonding (hydrogen bond, polar covalent bond, ionic bond, and non-polar covalent bond)
Sodium chloride: ______
Carbon to carbon bond in glucose molecule: _______
Bond between hydrogen and oxygen in water molecule: ______
Bonds between two strands of DNA: ________
Sodium chloride: ionic bond
Carbon to carbon bond in glucose molecule: non polar covalent bond
Bond between hydrogen and oxygen in water molecule: polar covalent bond
Bonds between two strands of DNA: hydrogen bond
[Quiz Question]
The Group 8A (noble elements) are inert/unreactive because _____
They are heavy
Do not interact with other elements
Have maximum number electrons in their outermost shells
Have eight protons and neutrons
Have maximum number electrons in their outermost shells
[Quiz Question]
The life sustaining properties of water is due to _______
Molecule is made up of hydrogen and oxygen
Asymmetric charge distribution of the molecule
It is liquid
It can exist in liquid, solid, and gaseous form
Asymmetric charge distribution of the moleucle
![<p>[Quiz Question]</p><ol><li><p>The number of water molecules: ______</p></li><li><p>Oxygen and hydrogen within each molecule are bonded by: _____</p></li><li><p>Different water molecules are bonded by: ______</p></li><li><p>The partial charge on the oxygen atom is: ________</p></li><li><p>The partial charge on the hydrogen atom is: _______</p></li></ol><p></p>](https://assets.knowt.com/user-attachments/1402abb8-3e7c-4cb8-b38f-959b817fa1ff.jpg)
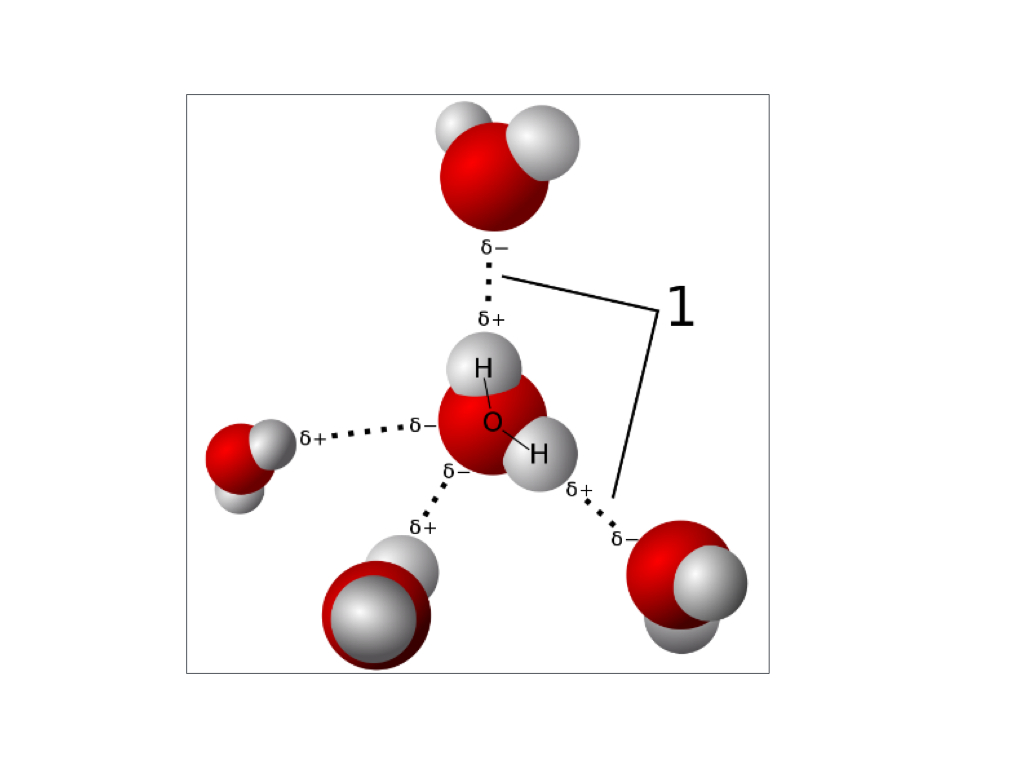
[Quiz Question]
The number of water molecules: ______
Oxygen and hydrogen within each molecule are bonded by: _____
Different water molecules are bonded by: ______
The partial charge on the oxygen atom is: ________
The partial charge on the hydrogen atom is: _______
The number of water molecules: five
Oxygen and hydrogen within each molecule are bonded by: polar covalent bond
Different water molecules are bonded by: hydrogen bonds
The partial charge on the oxygen atom is: delta negative
The partial charge on the hydrogen atom is: delta positive
[Quiz Question]
pH is the measurement of ________
Hydrogen ions in a solution
Hydroxyl ions in a solution
Carbonate in solution
Chloride in a solution
Hydrogen ions in a solution
[Quiz Question]
pH is important in living organisms because __________ (multiple answers)
It maintains cell structures
Living cells can survive under certain range of pH
Enzymes function within a narrow range of pH
Blood plasma and body fluids have fluids have pH of 6.8-7
1, 2, and 3 are correct answers.
![<p>[Quiz Question]</p><p>Identify the molecule in the photo:</p><ol><li><p>phospholipid</p></li><li><p>hexose</p></li><li><p>glycerol</p></li><li><p>amino acid</p></li></ol><p></p>](https://assets.knowt.com/user-attachments/892af1b0-1796-4364-a195-42c31fc7e4e0.png)
[Quiz Question]
Identify the molecule in the photo:
phospholipid
hexose
glycerol
amino acid
amino acid
Concept Map