Chemistry: Alcohols and esters
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
Acidity (what determines it?) + Compare the acidity of ethanol and water
The ability to donate proton
More Polar -OH bond is, stronger acid
Weaker acid:
Positive inductive effect, -OH in ethanol is less polarised
-OH(-) => conjugate base of water
C2H3O- : conjugate base of ethanol; O is more negative (+ Inductive effect) → more likely to re-bond with H+ → less stable, weaker
Summary: Ways to Produce? Reagents?
Produce (5 ways)
Alkenes: (2): hydration (Heat + H3PO4) NUOC (STEAM) A36, nuoc lanh oxidation with cold dilute ACIDIFIED KMnO4 → COLD DILUT → MILD O AGENT, = ISN’T FULLY BROKEN
Halogenoalkane, hydrolysis with H2O (aq) + nucleophilic substitution + Heat
Aldehydes + Ketones: reduction with LiAlH4, NaBH4; reduced to your origin & 2[H] * No condition
Carboxylic acid: reduction with LiAlH4, 4[H] * No condition
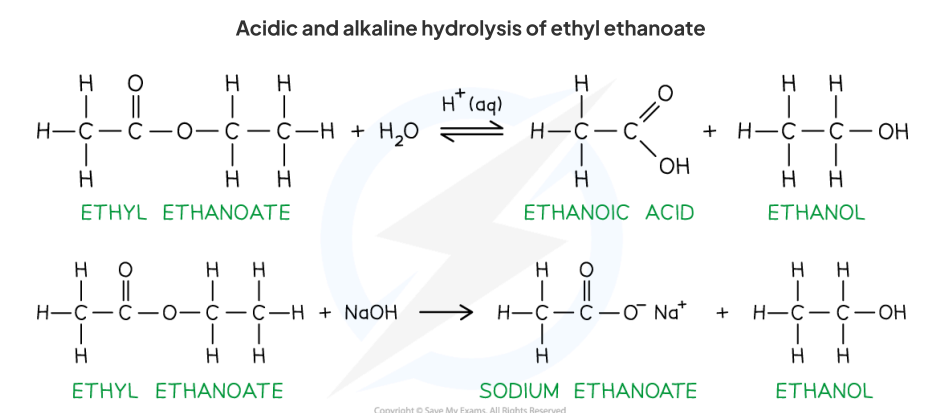
Ester: hydrolysis with dilute acid / alkali
Reactions (4)
Oxygen (Combustion): sufficient oxygen
Substitution with HX or KBr conc H2SO4 / H3PO4 (Basically 4 for Halogenoalkanes)
Na(s)
Dehydration reaction to alkene
Oxidation to Aldehydes / Ketones w/ Acidified K2Cr2O7
Condensation reaction
Two molecules are combined to form a larger molecules and at the same time eliminating a smaller molecule
Hydrolysis of Esters (easy) (Condensation reaction) → Producing
Add dilute Acid / Alkali + heat → condensation occured, alcohol forms!
easy: no need for concentrated
General form:
Add acid → like original: an alcohol + carboxylic acid : Acid ←> Alcohol
Add alkali → almost: alcohol (obvi) + … -oate (Eg: Sodium ethanoate)
what pass is passed
Combustion → reactions
→ CO2 + H2O
Na
General equation:
Alcohol + sodium → Sodium Alkoxide + H2 → Identifying reagents so don’t you dare to forget!!!
Aldehydes + Ketones
Acidified K2Cr2O7 (Mild OXIDATION THO!) / KMnO4
Primary alcohol: heat & oxidising agent → Aldehyde (distilled off) +H2O → Carboxylic acid (reflux)
Secondary alcohol: Ketone
Identify alcohols
Cr2O72- colour is green
basically change from orange → green if it’s either primary or secondary
Dehydration to alkene
Al2O3 + heat or A CONCENTRATED ACID
Esterification (YES)
Carboxylic acid + conc H2SO4 Catalyst
Deduce this + to react with wut? to form wut??
CH3CH(OH)
with aqueous alkaline I2
to form pale yellow precipitate CHI3 (Tri - iodomethane) + RCO2-
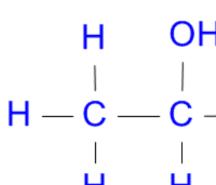
Substitution → Cl
PCl3 + heat, PCl5 → HCl (steamy white fumes) proves presence of alcohol, HCl → H2O, SOCl2

Bromine
HBr or reaction of NaBr + conc H2SO4 → HBr + NaBH4 (nas is for halogenoalkane….KBr + conc H2SO4)
or
2P + 3Br2 → 2PBr3
→ Halogenoalkane + P(OH)3
Iodine (normally)
2P + 3I2 → 2PI3
→ Halogenoalkane + P(OH)3