Experiement 2. Piperine Extraction
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
When today's reaction mixture is heated to a reflux, the temperature of the system is kept nearly constant at
~40 oC, boiling temperature of dichloromethane
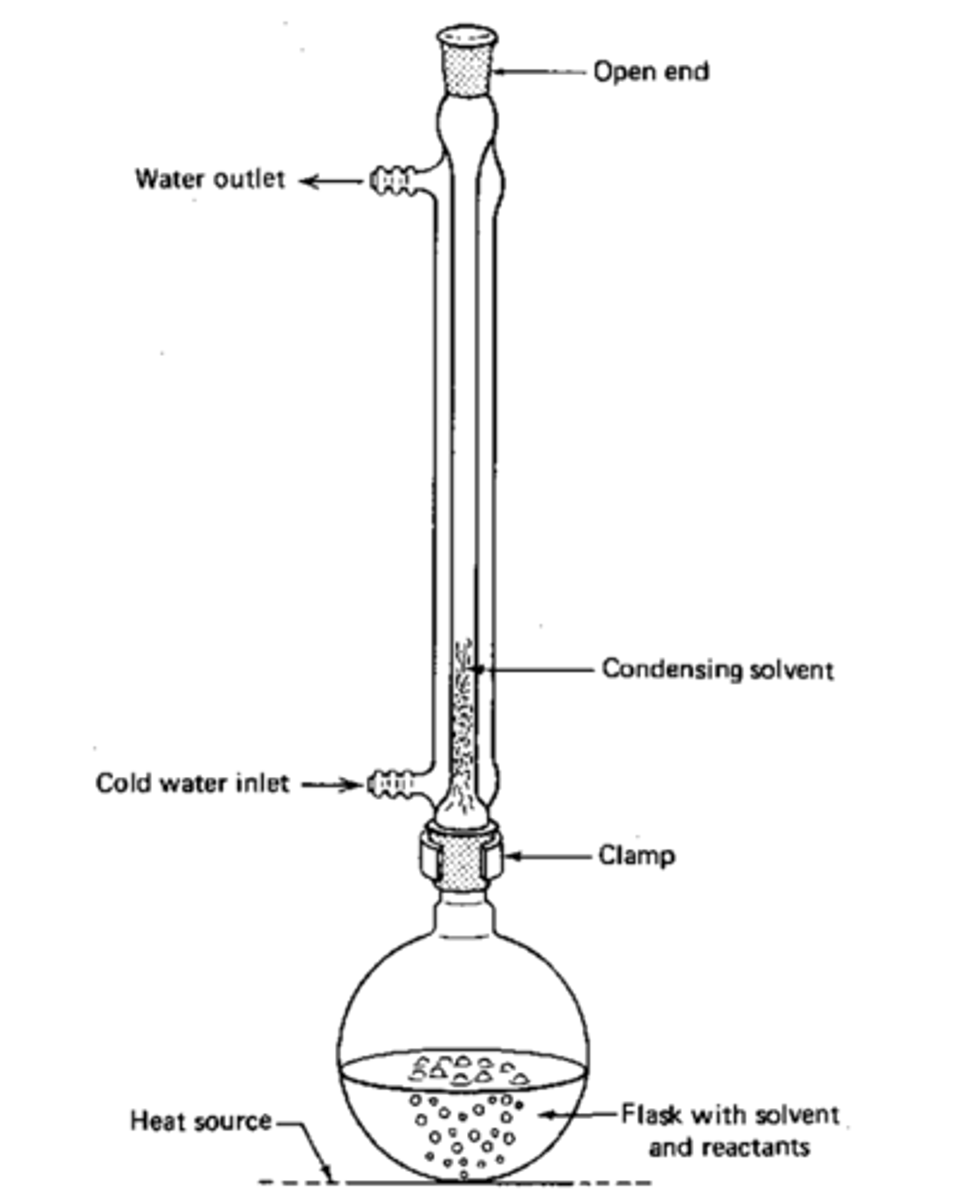
What role does reflux condenser play in the reflux set up?
water that runs through the condenser cools down solvent vapors so that they condense and return to the reaction flask
all applicable pieces of glassware and equipment that are needed for a typical vacuum filtration set up
buchner funnel
filter paper
rubber sleeve
filtering flask
Which method(s) will you use to determine purity of the isolated piperine
Melting Point Comparison
Dichloromethane is carcinogenic volatile solvent. What precautions should you take when working with DCM?
-work in ventilated area
-use glvoes and PPE
-clean spills immediately
Why is the extraction performed under reflux with a water-cooled condenser
To maintain a constant temperature and prevent solvent loss
After filtration, a drying agent (MgSO4 or Na2SO4) is added until it flows freely. Why is this step necessary before rotary evaporation?
To remove dissolved water that would contaminate the product and hinder recrystallization
In the ethanol-water recrystallization, why is water added to the hot ethanolic solution until precipitation appears
To decrease piperine solubility and induce crystallization in a mixed-solvent system
Why is the mixture hot-filtered through a cotton-plugged funnel before cooling?
To prevent premature crystallization in the funnel and remove charcoal/insolubles efficiently
Why is the filtrate first allowed to cool undisturbed to room temperature before placing it in an ice bath?
Slow cooling favors formation of purer, larger crystals; the ice bath completes crystallization
Why are crystals collected on a pre-chilled Büchner funnel and washed with ice-cold 1:1 ethanol-water?
To minimize product loss by reducing solubility while rinsing away impurities
Why does the procedure forbid using hot plates with exposed heating elements for this experiment?
Because ethyl acetate and ethanol are highly flammable; exposed elements are ignition sources
Why should the product be purified (recrystallized) before measuring its melting point?
Impurities depress and broaden melting ranges, obscuring comparison to the literature value
Why is percent recovery (mass of piperine obtained divided by initial pepper mass) reported instead of percent yield?
Because piperine preexists in the natural material; this is an extraction, not a synthesis with a fixed theoretical yield
Based on the structure of piperine, do you expect it to have better solubility in water or in dichloromethane?
In DCM, because polarity of the molecule matches polarity of DCM
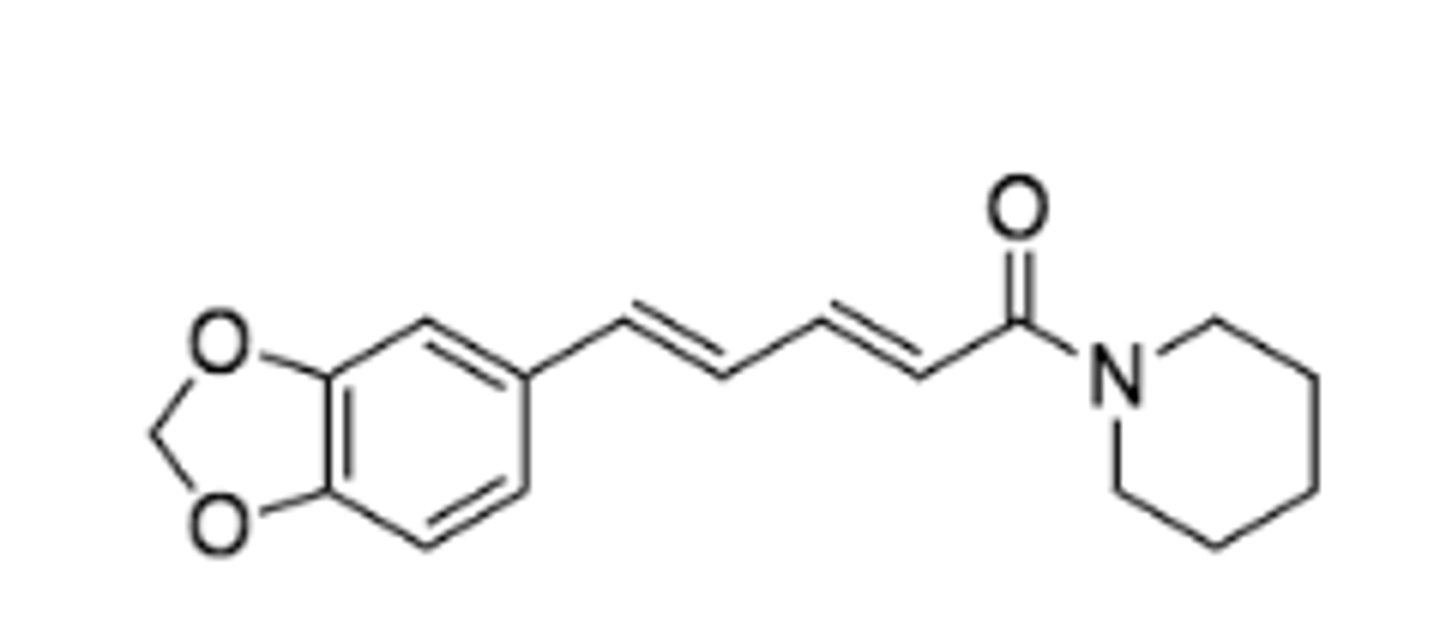
Given the structure of chavicine, what is the relationship between piperine and chavicine?
stereoisomers
If you were to react you piperine in the Bayer test we performed during experiment 1, what result will you expect?
the KMnO4 soultion will turn brown due to presence of free double bonds in piperine (note: aromatic rings do not react with KMnO4)
primary pungent principle of pepper is
piperine
strucutally what is apiperine
amide alkaloid
strucutre of piperine
Alkaloids are
orgnaic large compounds with N atom
-in plants that help with bitter taste and toxic to herbivores
Reflux is a method
heat reaction
mixture at constant temperature without losing solvents or reagents.
pure vs impure compunds vs melting temp
-Pure compounds melt
over a narrow range close to literature values;
-lower/broadened ranges suggest
impurities
Piperine is a
lachrymator- causes tears and eye irritation