MIDTERM 1
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
First Law of thermodynamics
Energy cannot be created nor destroyed, only transformed
ΔE = q + w
Second law of thermodynamics
entropy must be increasing over time to be spontaneous
(ΔSuniverse > 0 → spontaneous)
Third law of thermodynamics
The entropy of a perfectly ordered crystalline solid approaches zero as its temperature approaches absolute zero
exothermic signs
ΔH = - (favorable)
q = -
endothermic signs
ΔH = +
q = +
if something contracts…
w = +
surroundings do work on system
if something expands…
w = -
system is doing work
↑T
↑P, ↑V
KMT Assumptions
Volume of gas particles = 0
All collisions are perfectly elastic
No IMFs (bc huge distance between particles)
Motion is translational & proportional to temperature
If T is constant, how are urms & KE affected?
urms would only be affected by MW = heavier atom → slower speed
KE remains the same for all atoms
Boltzmann Distribution trends
As T ↑, the graph spreads out right
As MW ↑, the graph becomes skinner to the left
effusion
going through a pinhole
Lower MW = faster rate of effusion
subtract by 1 to find the percentage *
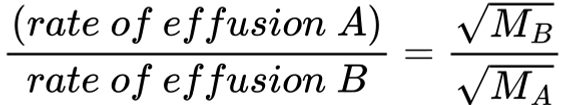
Path function
DEPENDS on the path taken from initial to final state (ex. heat, work)
State funtion
INDEPENDENT of the path taken (ex. internal energy)
CV (constant volume)
3/2R (for ideal gases)
ΔE = qV
More complex molecules (more bonds) = higher CV
w = 0
Why is CP > CV?
bc CP includes pressure-volume work
When is thermal equilibrium reached?
when heat transfer stops
Which state of matter has the highest CV and why?
Liquids bc they have all motion & strong IMFs
ideal gases - only translate
real gases = all motion & little IMFs
solids - vibrate & strong IMFs
How to find qrxn from qcal
qrxn = -qcal
CP (constant pressure)
5/2R (for ideal gases)
ΔE = qp + w
qp = ΔE + PΔV
where qp is a state function
qp = ΔH
w = -RTΔn
ΔH and ΔE are about the same when…
no gases are involved in the reaction
ΔV, ΔE, & ΔH when heating or producing a gas
ΔV = +
ΔH > ΔE
energy lost to work
ΔV, ΔE, & ΔH when cooling or losing gas in a reaction
ΔV = -
ΔH < ΔE
work done on system adds energy to system
How does stability/bonds relate to thermo?
more stable products = more exothermic
more sigma bonds in products → probably exothermic
bonds vs endo/exo
breaking bonds → endothermic
making bonds → exothermic
with BDE, it’s [bonds broken (reactants) - bonds formed (products)]
How to find temperature when given ΔH and ΔS
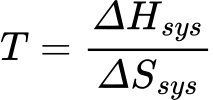
Born-Haber Cycle Steps
Sublimation(s → g)
BDE (break the bond)
Ionization (remove electron from cation)
Electron affinity (add electron to anion)
Lattice Energy
Add them all up to find the ΔHf
So Bob Is Evil Liar
isothermal
Constant T
favorable signs
ΔH = -
ΔS = +
spontaneous signs
ΔSuniverse = +
ΔG = -
What occurs at a phase change?
ΔT = 0
Both phases coexist
All heat is used to break IMFs
ΔSuniverse signs
ΔSuniverse = 0 → reversible
ΔSuniverse > 0 → NOT reversible
ΔSuniverse < 0 → can NOT occur
At slope of phase transition
use the equations that have change in temp (ex. ΔH = nCpΔT)
At constant of phase transition
use the transition values (ex. nΔHtrans)
enthalpy of fusion
s → l
how to compare the entropy of molecules
more bonds & more mass = more entropy
exergonic
ΔG = -
reaction wants to happen (favorable)
endergonic
ΔG = +
the reaction is NOT favorable BUT can still occur (if coupled w/ an exergonic reaction)
How to find Temp when not given
use PV = nRT → T = PV/nR
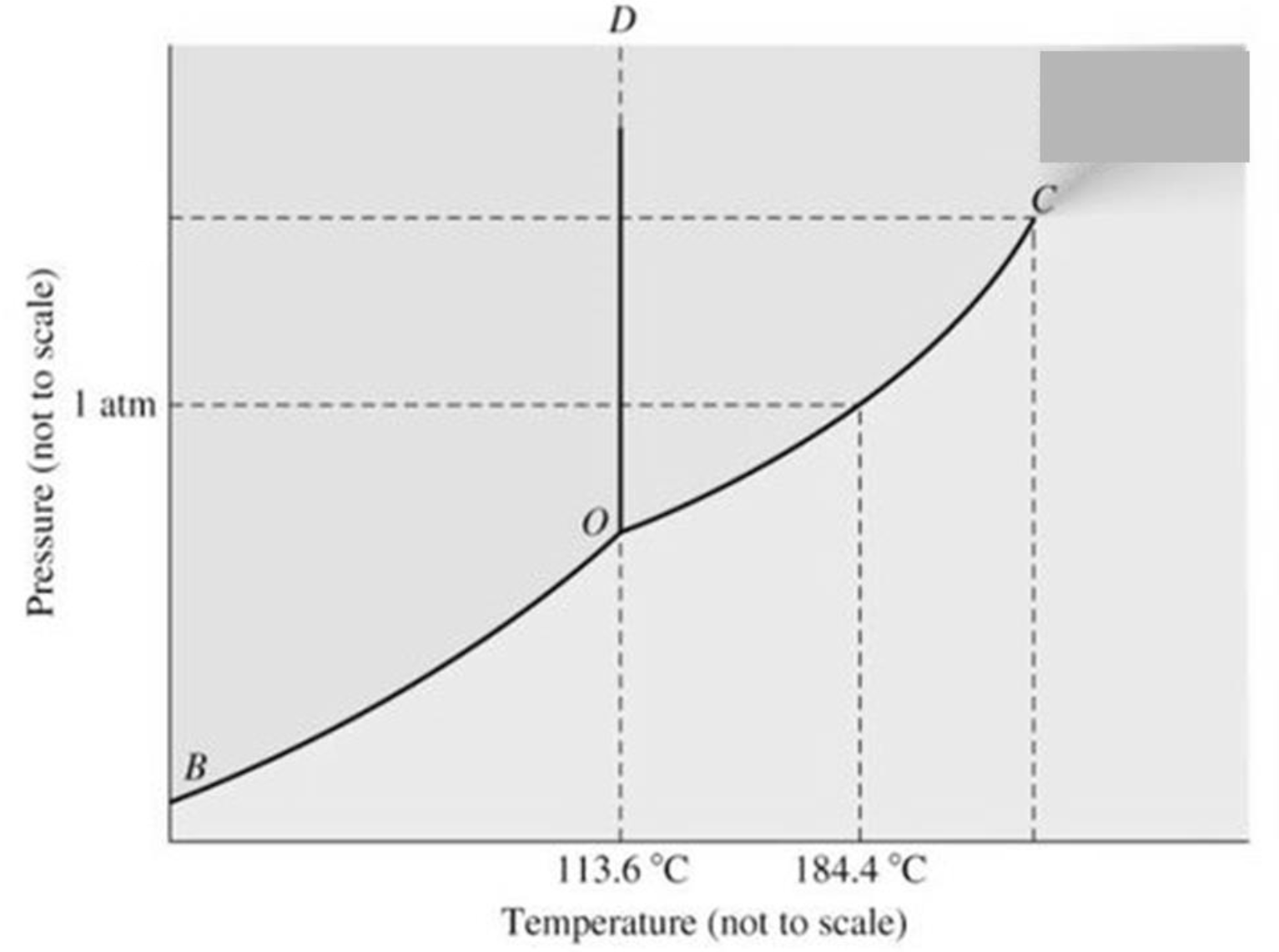
Label where vaporization, freezing, sublimation, melting, condensation, and deposition occur on the above diagram. Label the triple point and the point where a supercritical fluid is formed in the above diagram.
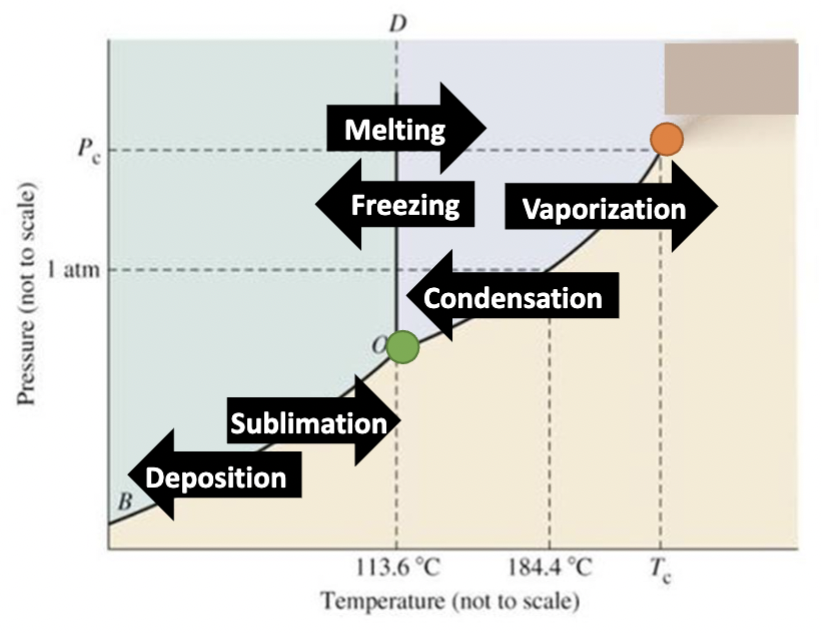
triple point
all three phases of matter (solid, liquid, and gas) coexist simultaneously
supercritical point
distinct boundary between its liquid and gas phases completely disappears, merging into a single continuous state
How to find S, G, or H from formation energies
products - reactants
How to H from BDE
bonds broken (reactants) - bonds formed (products)
state or path function: ΔH, ΔG, ΔS, ΔE
ΔH - state
ΔG - state
ΔS - state
ΔE - state
How does adding water to a bomb calorimeter affect heat capacity?
increases heat capacity
lower heat capacities =
higher changes in temperature