Extraction and Uses of Metals (2b)
1/22
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
Metal Ore
Rock that contains enough of a metal to make extraction profitable
Ways of Extracting Metals from Ores
electrolysis
blast furnace
reacting with a more reactive material
reduction: when ore is an oxide of the metal, since oxygen is being removed
Oxide Ores
ex: Iron → Haematite; Aluminium → Bauxite
extracted by reduction, since removal of oxygen
Unreactive Metals
don’t have to be extracted chemically since found as an uncombined element
because don’t react with other substances due to chemical instability
ex: Au and Pt
A metal can reduce another metal only if it is ___________ than the metal that is bonded to the oxygen
more reactive
Extraction of Metals and Reactivity Series
most reactive at the top
tendency to become oxidised is linked to position
higher up are less resistant to oxidation
position determines method of extraction
Metals extracted by Electrolysis
electrolysis of molten chloride or oxide
large amounts of electricity required → expensive process
Metals
K
Na
Ca
Mg
Al
Metals extracted by Reduction
extracted by heating with a reducing agent like C or CO in a blast furnace
cheap since C is cheap and can be used as a source of heat
Metals
Zn
Fe
Cu
Metals found as Pure Elements
Ag
Au
Extraction of Iron from Haematite
extracted in a large container called a blast furnace
modern blast furnaces produce approx. 10,000 tonnes of Iron per day
continuous process with raw materials added and removed all the time
due to cost and time associated with getting the furnace up to temperature
Extraction of Aluminium from Bauxite
Electrolysis
bauxite is purified to produce Al2O3
this is dissolved in molten cryolite to lower melting point
mixture is placed in an electrolysis cell made from steel lined with graphite
graphite lining acts as cathode, large graphite blocks act as anode
at cathode, Al ions get reduced, molten Al forms, gets siphoned off from time to time
at anode, oxidation, oxygen produced → C reacts with O2 to form CO2
anode wears away, has to be replaced
expensive process
Electrolysis Half Equations
Al3+ + 3e– → Al
2O2– → O2 + 4e–
C (s) + O2 (g) → CO2 (g)
Aluminium Uses
aircraft bodies (high strength-weight ratio)
saucepans (very good conductor of heat, unreactive)
overhead electrical cables (very good electrical conductor)
food cans (non-toxic, corrosion and acidic food-stuff resistant)
Copper Uses
electrical wiring (very good electrical conductor, ductile)
saucepans (very good electrical conductor, malleable)
water pipes (unreactive to water, non-toxic, malleable)
Steel Types
mild
high carbon
stainless
Mild Steel
Fe alloyed with 0.25% C
used in car bodies and wiring
soft and malleable
High Carbon
Fe alloyed with 0.5 - 1.4% C
used in car body panels and wiring
hard
Stainless Steel
Fe alloyed with 20% Cr and 10% Ni
used in cutlery, sinks and chemical plants
strong and corrosion-resistant
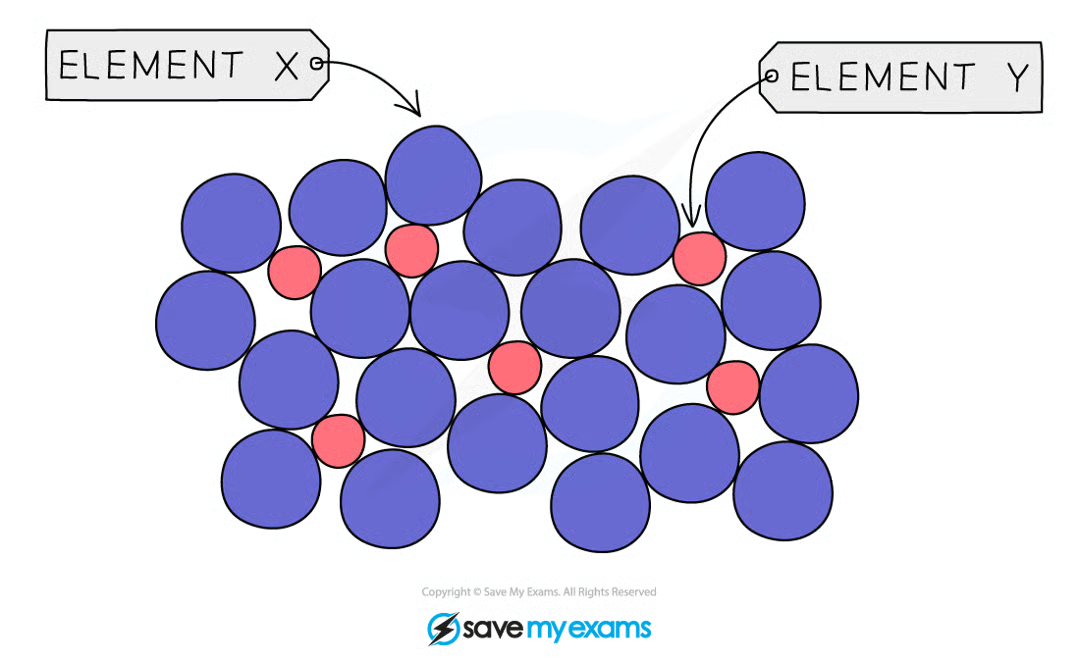
Alloys
Mixture of two or more metals with a non-metal like Carbon Ex: Steel
Alloy Properties
stronger and harder
resistant to corrosion
resistant to extreme temperatures
this makes them more useful than pure metals
Alloys are harder because:
contain different sized atoms
this distorts atoms’ regular arrangements
more difficult for atoms to slide over each other
Brass Composition
70% Cu, 30% Zn
Alloy Diagram