inputs and outputs of cellular respiration
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
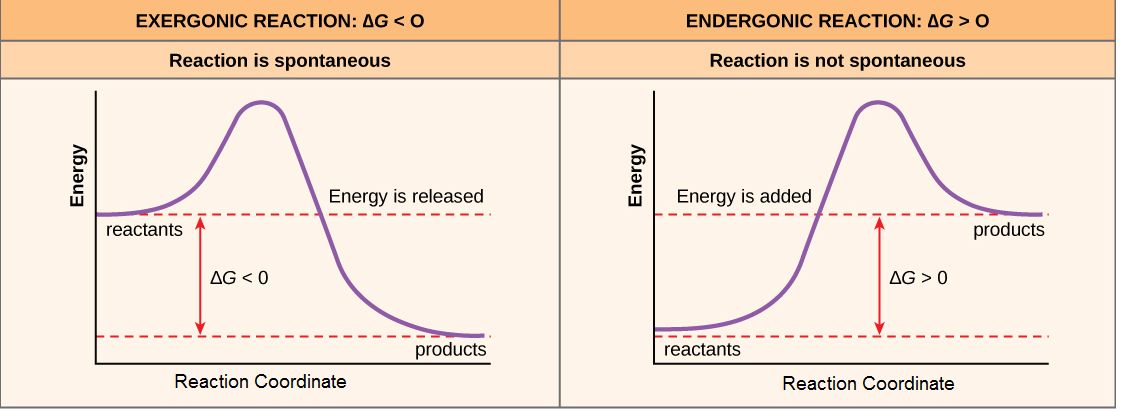
spontaneous reactions
Spontaneous reactions are chemical processes that occur without the need for external energy input. They can happen naturally under specific conditions, leading to a stable state.
nonspontaneous reactions
Chemical processes that require energy input to occur.
Inputs of Glycolosis
Glucose, 2 ATP, 4 ADP, 2 NAD^+
Outputs of glycolysis
2 pyruvate, 2 NADH, 2 ATP, 2 H2O
Inputs of pyruvate processing
2 Pyruvic acid, 2 Acetyl CoA, 2 NAD+
Outputs of pyruvate processing
2 acytyl CoA, 2 CO2, 2 NADH
Inputs of the citric acid cycle
2 acytyl CoA, 2 ADP, 6 NAD+, 2 FAD
Output of citric acid cycle
2 CoA, 4 CO2, 2 ATP, 6 NADH, 2 FADH2
Input to Electron Transport chain/ Chemosmosis
10 NADH, 2 FADH2, 6 O2, 34 ADP + Pi,
Output of Electron Transport Chain/Chemiosmosis
10 NAD+, 2 FAD, H2O, 34 ATP
spontaneous reactions
active zone- binds with effector molecules
non spontaneous reaction
allosteric zone- binds with substrate
Photosystem II and I location
Inside the thykaloid lumen
System that oxydizes water
Photosystem II
Photosystem that captures shorter wavelengths of light (Uses FADH)
Photosystem II
Photosystem I
Only gain electrongs from photosytem II. Uses longer light wavelengths. Reduced NADPH to NADP
What do photosystems I and II need to function
H2O
Chemiosmosis
The production of ATP using the diffusion of protons as it flows through ATP synthase