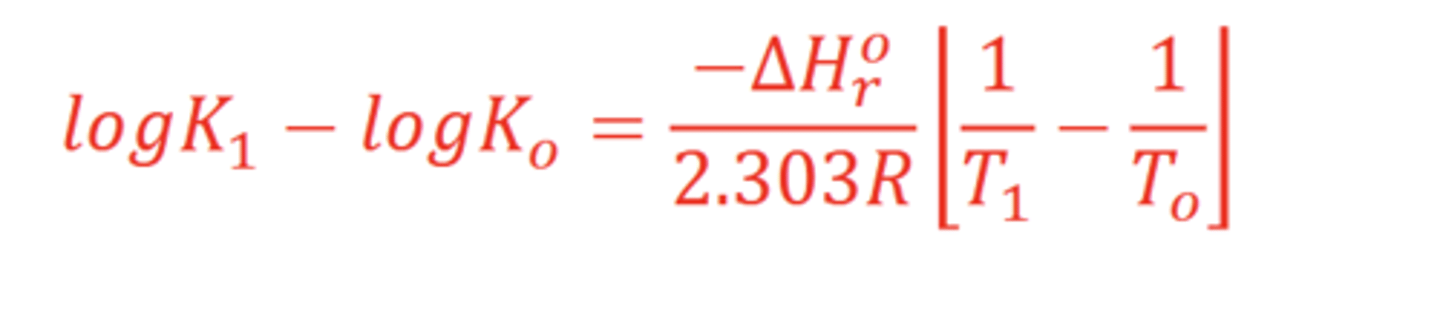EASC 2502 Module 6: Thermodynamics
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
What is thermodynamics?
Science of the transformation of energy
Define system
The part of the universe we wish to study
Define surrounding
All parts of the universe that are not part of the system
Define boundary
The surface that separates system form surrounding
Explain open system
Mass + energy exchange
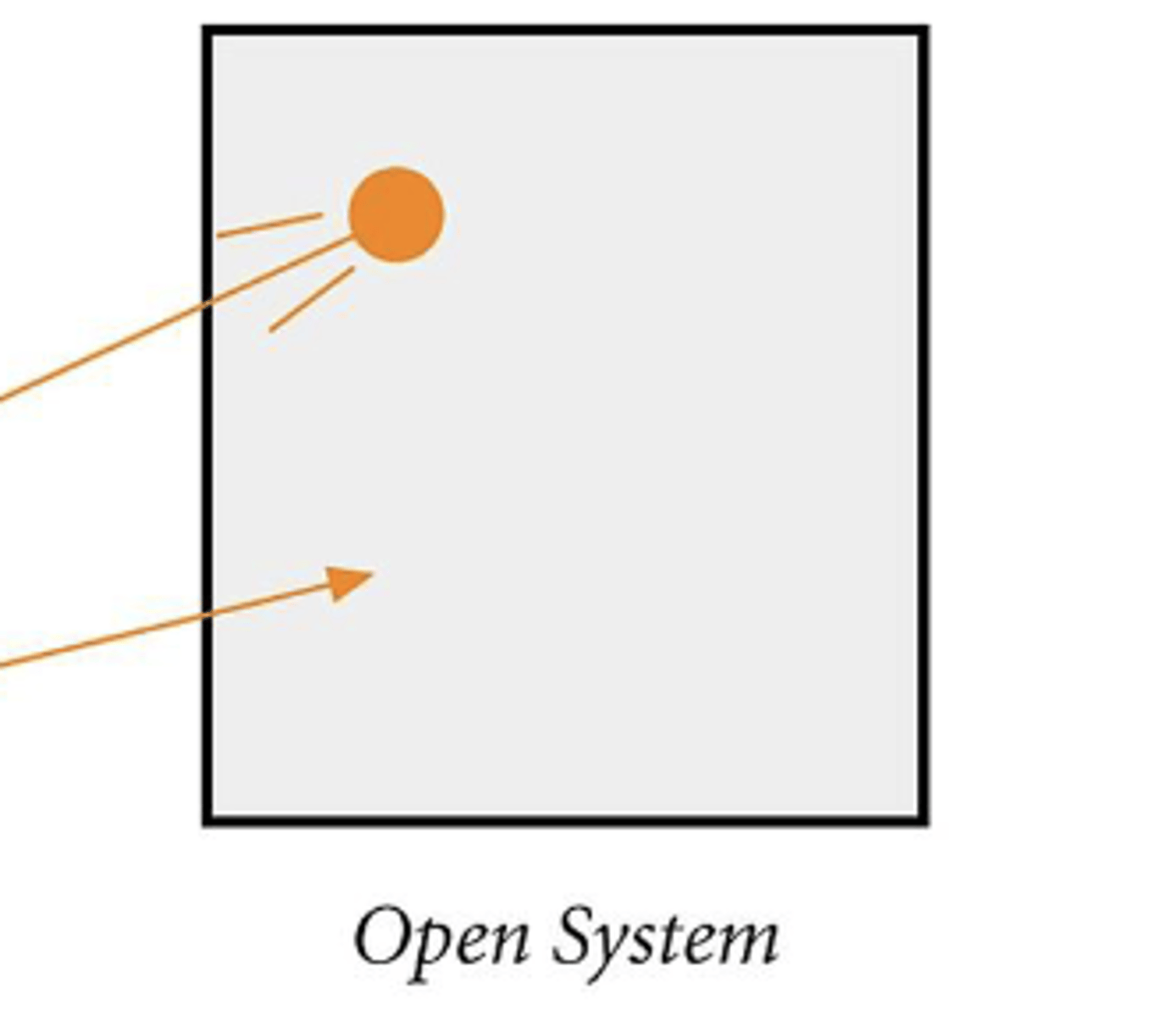
Explain closed system
Only energy exchange
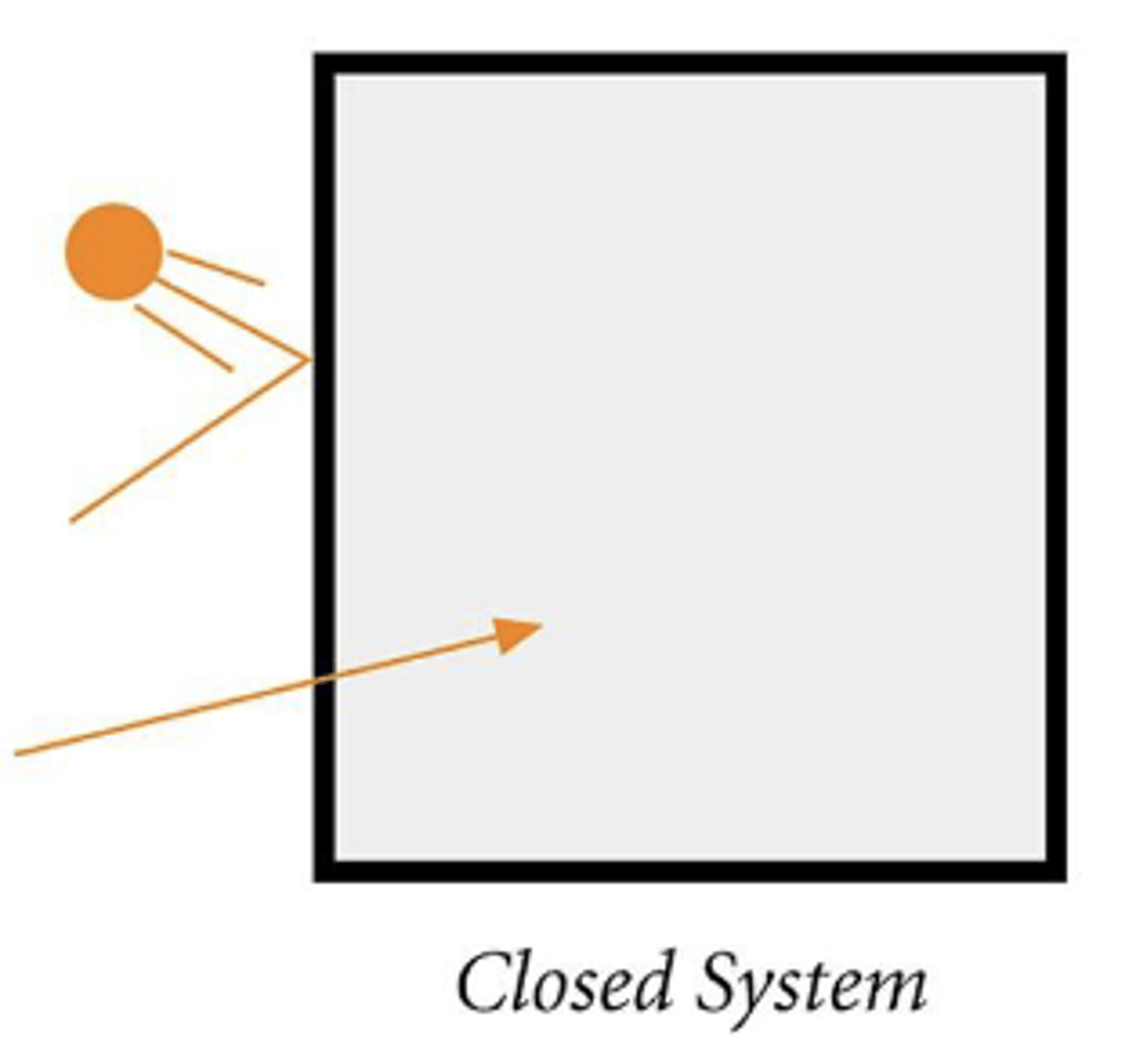
Explain isolated system
No mass or energy exchange
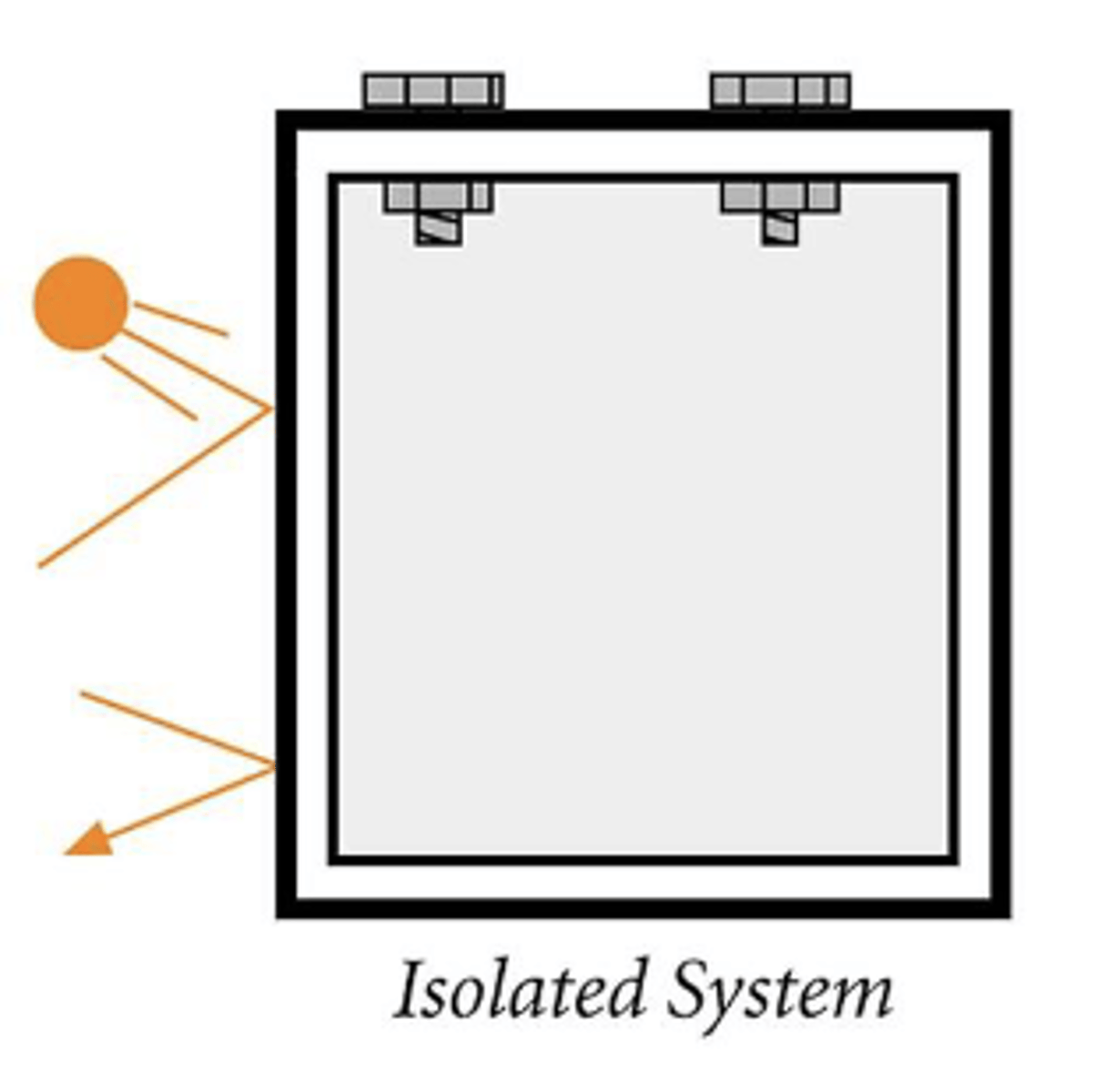
Define phase
A homogenous, physically distinct part of a system. Ex. olivine, or a gas phase or liquid phase
Define component
The chemical constituents needed to form the phases in the system. Ex. olivine has 2 components, MgO and SiO2.
In a magma chamber with partial crystallization, what are the phases and components?
Phases: 2 - liquid and solid
Components: the # of mineral species
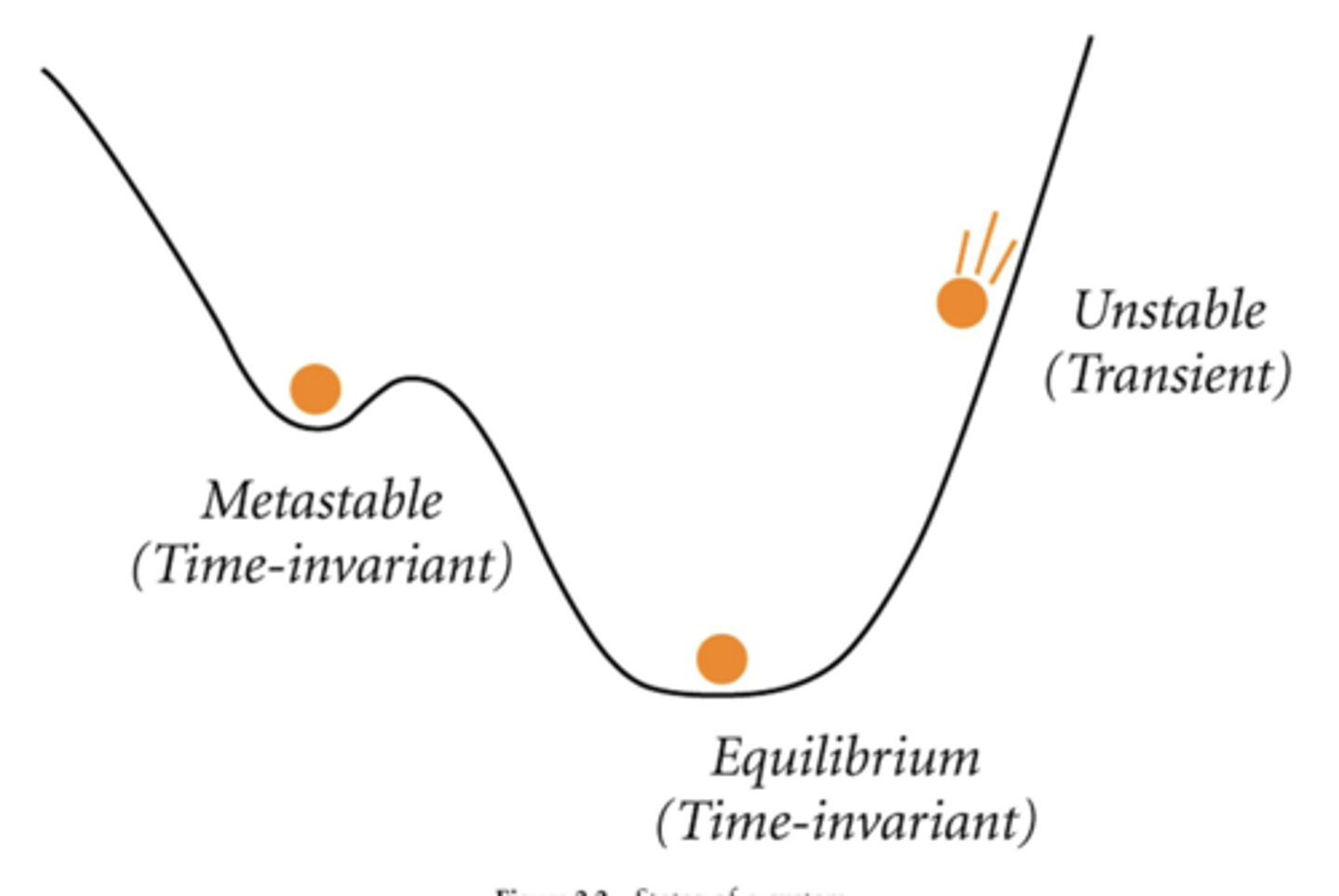
Define equilibrium
State where system does not change with time. Ex. A + B <-> C + D, reaction is constantly happening but overall system doesnt change
Is a diamond on a desk at equilibrium?
No, it is thermodynamically unstable, but the slow kinetics of the transformation reaction makes it metastable.
Explain STP of solids, gasses, and dissolved ions
Solids: pure materials
Gasses: pure gas behaving ideally.
Dissolved ions: ideal, 1 molal H2O solution
Define internal energy
The total energy in a system, U
When do reactions occur, with respect to Ui vs Uf?
Reactions occur to lower the energy, or when Uf < Ui, so ∆U < 0.
What is the 1st law of thermodynamics?
Conservation of energy
Explain the formula ∆𝑈 = 𝑈𝑓 − 𝑈𝑜 = 𝑄 − 𝑊
The change in energy in a system equals the difference of the heat (Q) added to the system and the work (W) done by the system
What is work?
Displacement of an object by a force
View the formula for ∆𝑈 with work done by volume change
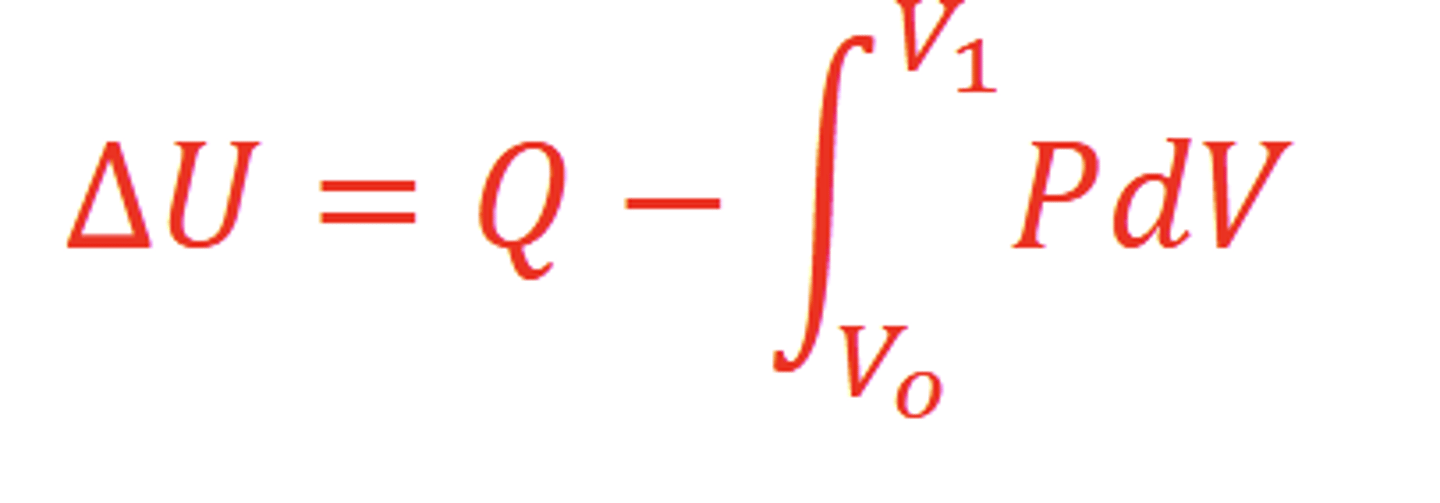
What is enthalpy (H)?
The heat of a system at constant pressure
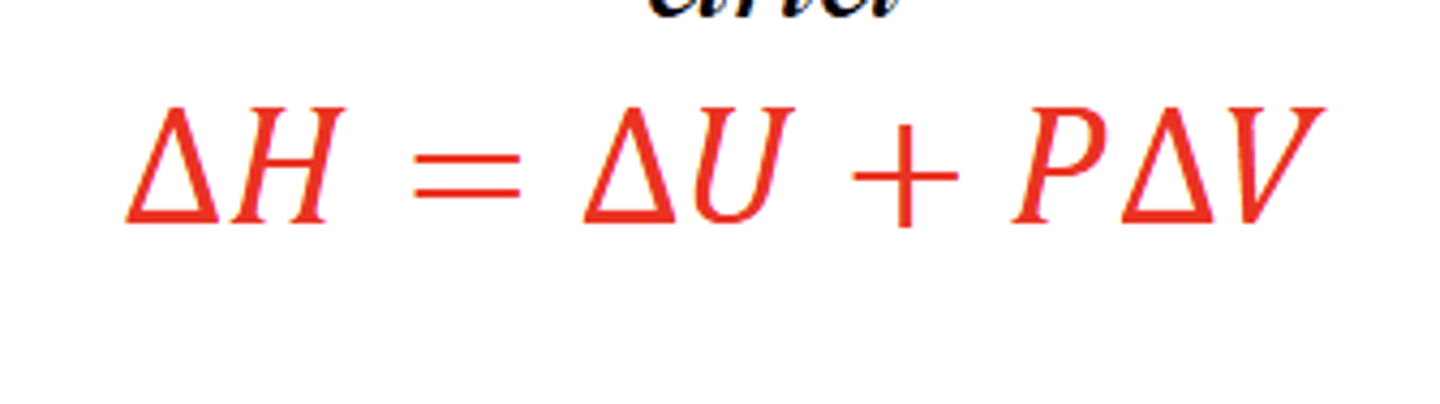
What is ∆Hf?
Change in enthalpy when 1 mole of pure substance is formed from its constituents in STP
Define standard state
The most stable form of a pure substance at STP
What is ∆Hr > 0?
Endothermic
What is ∆Hr < 0?
Exothermic
In exothermic reactions, products have _____ enthalpy than reactants and the reaction is ______________
Less, spontaneous
If you add heat to a system, will endothermic reactions be more or less favorable?
More
Can enthalpy (∆H) by itself predict if a reaction is spontaneous?
No
What is the 2nd law of thermodynamics?
Entropy.
Energy spontaneously goes from high to low concentration of energy
What is entropy (S)?
The energy dispersed per unit temperature in any process.
Is the energy in entropy available to do work?
No
What is Gibbs free energy?
Combo of H and S to determine spontaneity. Spontaneous reactions decrease Gibbs
Explain Gibbs for spontaneous and nonspontaneous.
G<0 = spont
G>0 = non
The dissolution of salt is endothermic but why does it occur spontaneously?
Because ∆Gr < 0
View reaction quotient formula
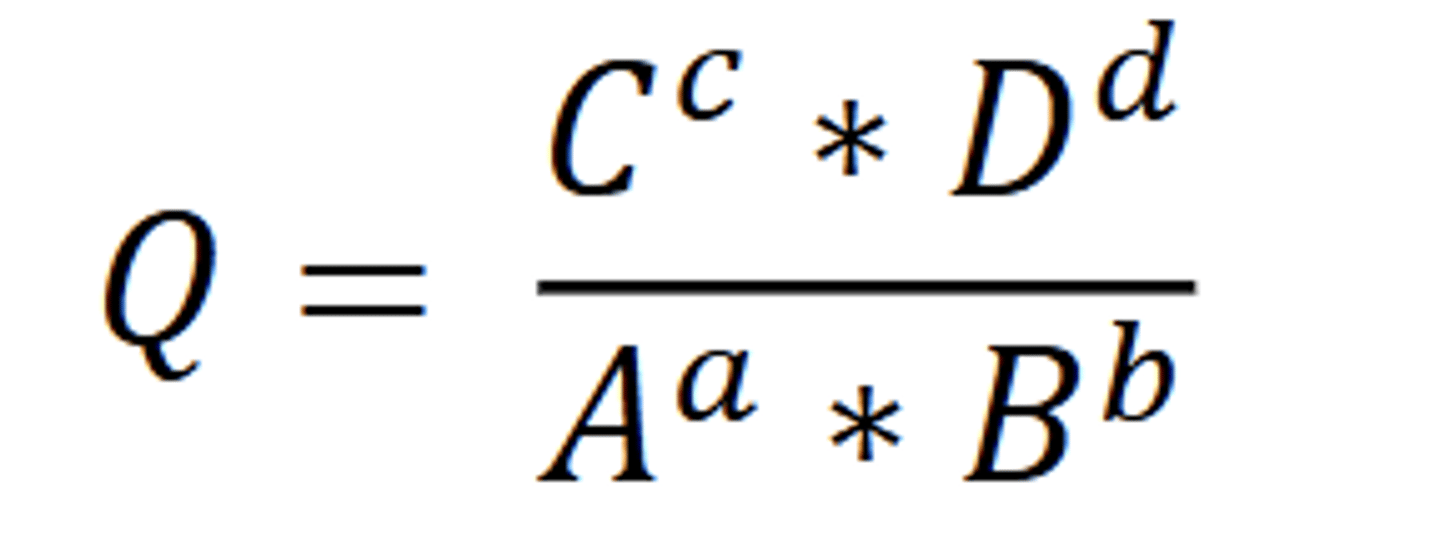
What is equilibrium constant K and how does it relate to Q?
K is Q at equilibrium
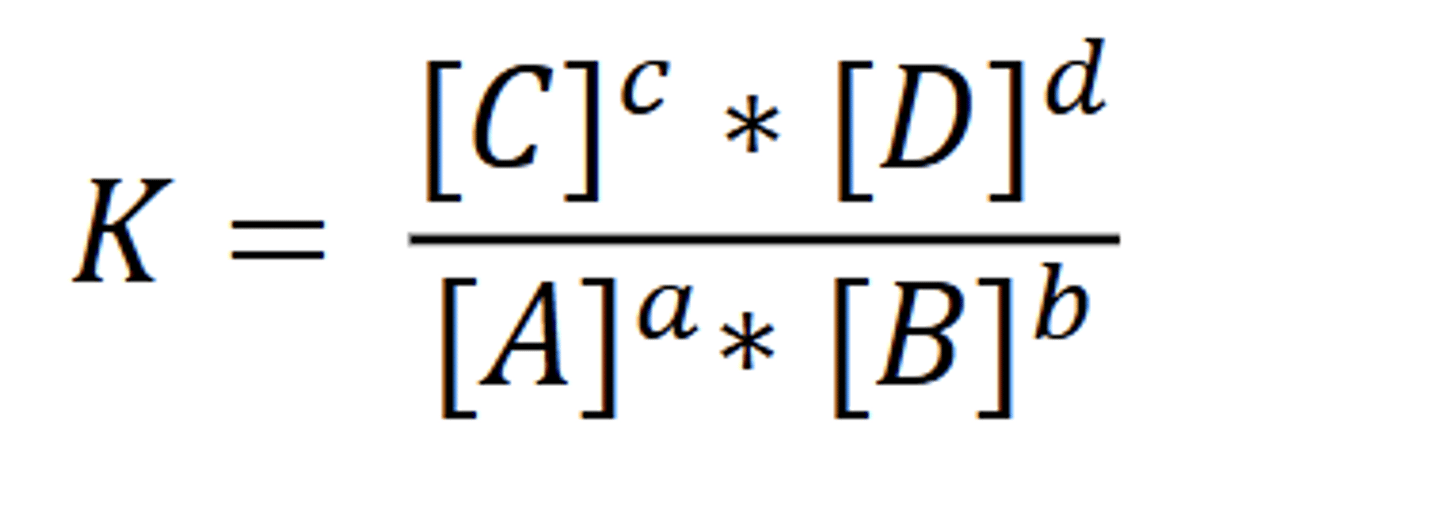
What happens if K >> 1?
What happens if K << 1?
What happens if K = 1?
Forward reaction, products favored
Reverse reaction, reactants favored
Equilibrium
What happens to K if reaction reverses?
1/K
What happens to K if you combine 2 reactions?
K = K1 * K2
What is the van't Hoff equation for?
Reactions at non-standard T and P