Biology 210 Final
1/67
Earn XP
Description and Tags
Chapters 1-16
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
68 Terms
Information Flow of Bio System
DNA, RNA, Protein
Properties of Life
Evolution, Order, Sensitivity, Reproduction/Growth/Development, Adaptation, Homeostasis, Energy Processing
Evolution
process of gradual change in a population over time
Order
organisms are highly organized and coordinated structures
Sensitivity/Response to stimuli
ability to respond to positive/negative stimuli
Reproduction, Growth and Development
creation of new cells, increase in size or stage due to DNA
Adaptation
organisms change to fit their environment
Regulation/Homeostasis
maintaining stable conditions
Energy Processing
sourcing energy for metabolic activities
3 Domains of Life
Bacteria, Archaea, Eukarya
Bacteria
Prokaryotes (lack intracellular organelles)
Archaea
Prokaryotes that are able to live in extreme conditions
Eukarya
eukaryotes (cells with a membrane, a nucleus, and organelles)
Cell hierarchy (smallest to largest)
atom, molecule, macromolecule, organelle, cell, tissue, organ, organ system, organism, population/community, ecosystem, biosphere
Major Elements of Life
oxygen, carbon, hydrogen, nitrogen
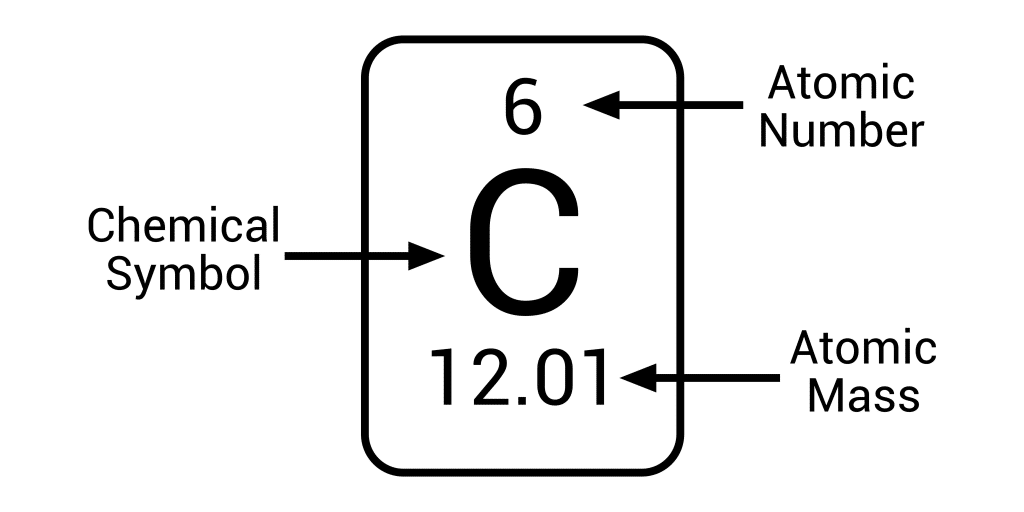
Nuclear Symbol Notation
Atomic # = number of protons
Mass # = Protons + neutrons
Octet Rule
Atoms are more stable with 8 valence electrons in outermost cell
Location of Protons and Neutrons
nucleus
Location of Electrons
orbitals
Covalent Bond
Sharing of electrons
Polar (different electronegativity) and Nonpolar (similar electronegativity)
Ionic Bond
Donation of electron to another atom
Hydrogen Bond
Hydrogen (partial positive) bonds to N, O, or F
found in DNA
Van Der Waals Interactions
weakest bond, depends on electron density flunctuations
Water Chemical Properties
Good Solvent due to polarity
High heat capacity and heat of vaporization
Neutral pH
Carbon
backbone of macromolecules
Saturated (single bond)
Unsaturated (double/triple bond)
Geometric Isomer
atom arrangement around C=C bond
Trans arrangement
linear backbone (packed tightly)
Cis arrangement
bent backbone (packed loosely)
Enantiomers
L-amino (protein) and D-glucose (photosynthesis)
Dehydration Synthesis
Monomer + Monomer = Polymer + H2O
Lose water to combine monomers
Hydrolysis
Polymer + H2O = Monomer + Monomer
split water to form monomers
Monomer of Nucleic Acid
Nucleotides (sugar, phosphate and base)
Purines
A & G (double ring base)
Pyrimidines
T, U, & C (single ring base)
DNA
genetic material of life
Double helix
directionality (5’ to 3’ or 3’ to 5’)
RNA (ribonucleic acid)
Involved in protein synthesis
U instead of T
Complementary Base Pairing Practice:
5’ AAGGTTCCATCG 3’
3’ TTCCAAGGTAGC 5’
Types of RNA
In protein synthesis
mRNA, tRNA, rRNA
Gene regulation
microRNA
Proteins
Molecules of work
digestive, transport, structural, hormones, defense, contractile, storage
Amino Acids
Protein monomer
Central carbon, amino group, hydrogen, and carboxyl group)
20 amino acids w/ side chain R
Dehydration synthesis: amino acid = proteins w/ peptide bond
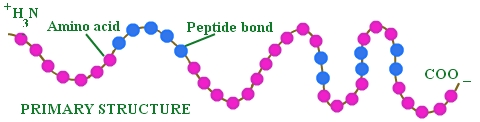
Primary Protein Structure
Insulin
2 polypeptide chains and 3 disulfide bonds
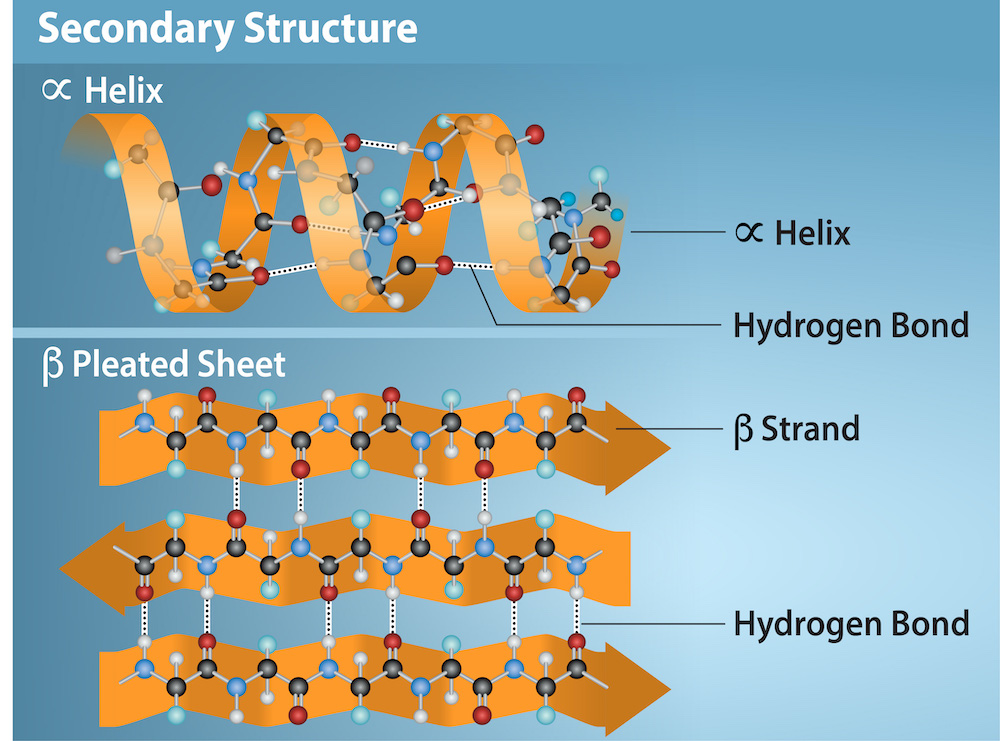
Secondary Protein Structure
Folding
Hydrogen Bonds
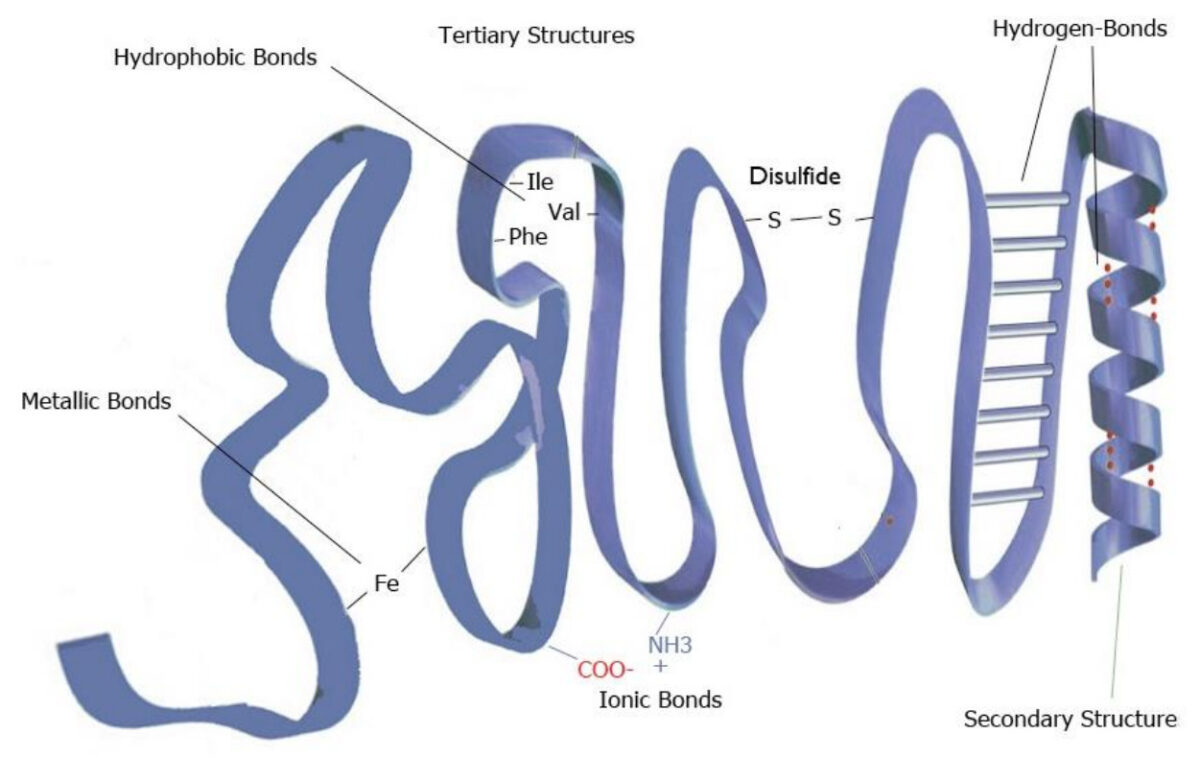
Tertiary Protein Structure`
Hydrogen bond & ionic bond
Hydrophobic
Quaternary Protein Structure
Polypeptide subunit interaction
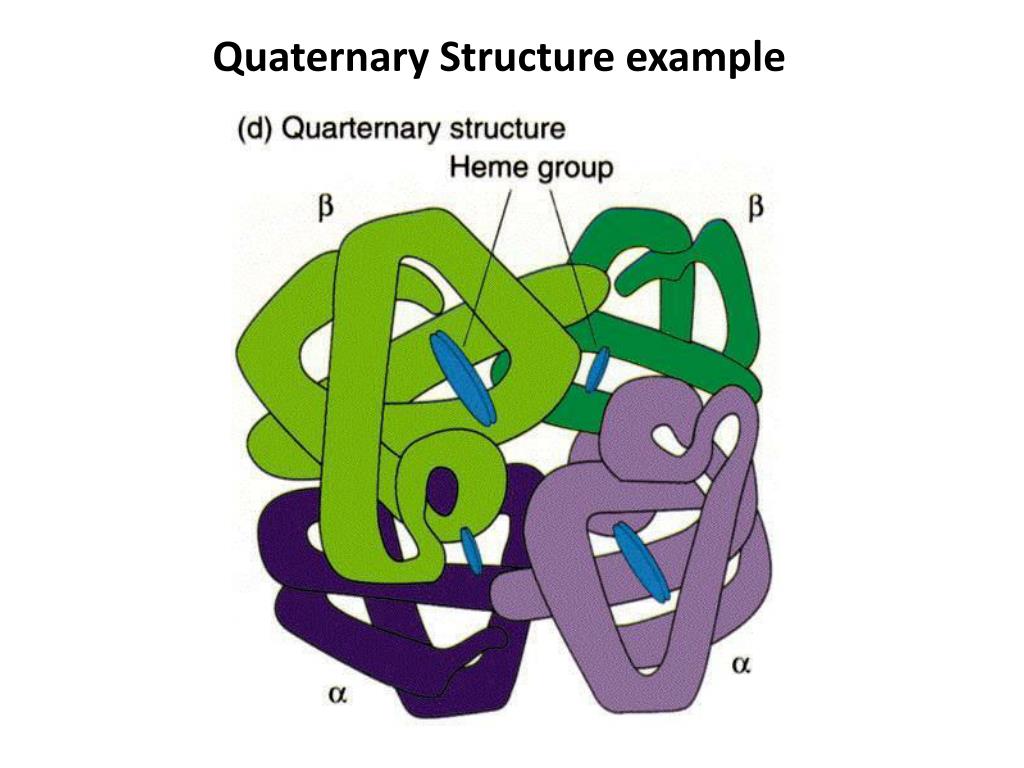
Protein denaturation
Unfolding of protein structure
heat, pH, solvents or agitation
Carbohydrates
Energy storage and structural support
Formula:
(CH2O)N
N = # carbon atoms
Monosaccharides
building blocks of carbohydrates
Linear or rings
glucose, fructose, galactose, ribose, and deoxyribose
Disaccharides
formed by a glycosidic bond (strong covalent) between monosaccharides
Maltose, lactose and sucrose
Polysaccharides
Multiple monosaccharides bonded
Starch (plant storage)
Cellulose (most abundant in plant cell walls)
Glycogen (animal storage)
Chitin (contains nitrogen and is found in exoskeletons)
Lipids
Fats & Oils (glycerol + fatty acid)
Waxes (long fatty acid chain + long alcohol chain)
Phospholipids (in cell membrane)
Steroids (4 carbon ring + tail)
Saturated fatty Acid
Carbon single bond (C-C)
tightly packed
Solid @ room temp
Unsaturated Fatty Acid
Carbon double bond (C=C)
Loosely packed
Liquid @ room temp
Essential fatty acid
NOT made by the body
Omega 3 & Omega 6
Cholesterol
Precursor to hormones & vitamin D
Essential to the phospholipid bilayer
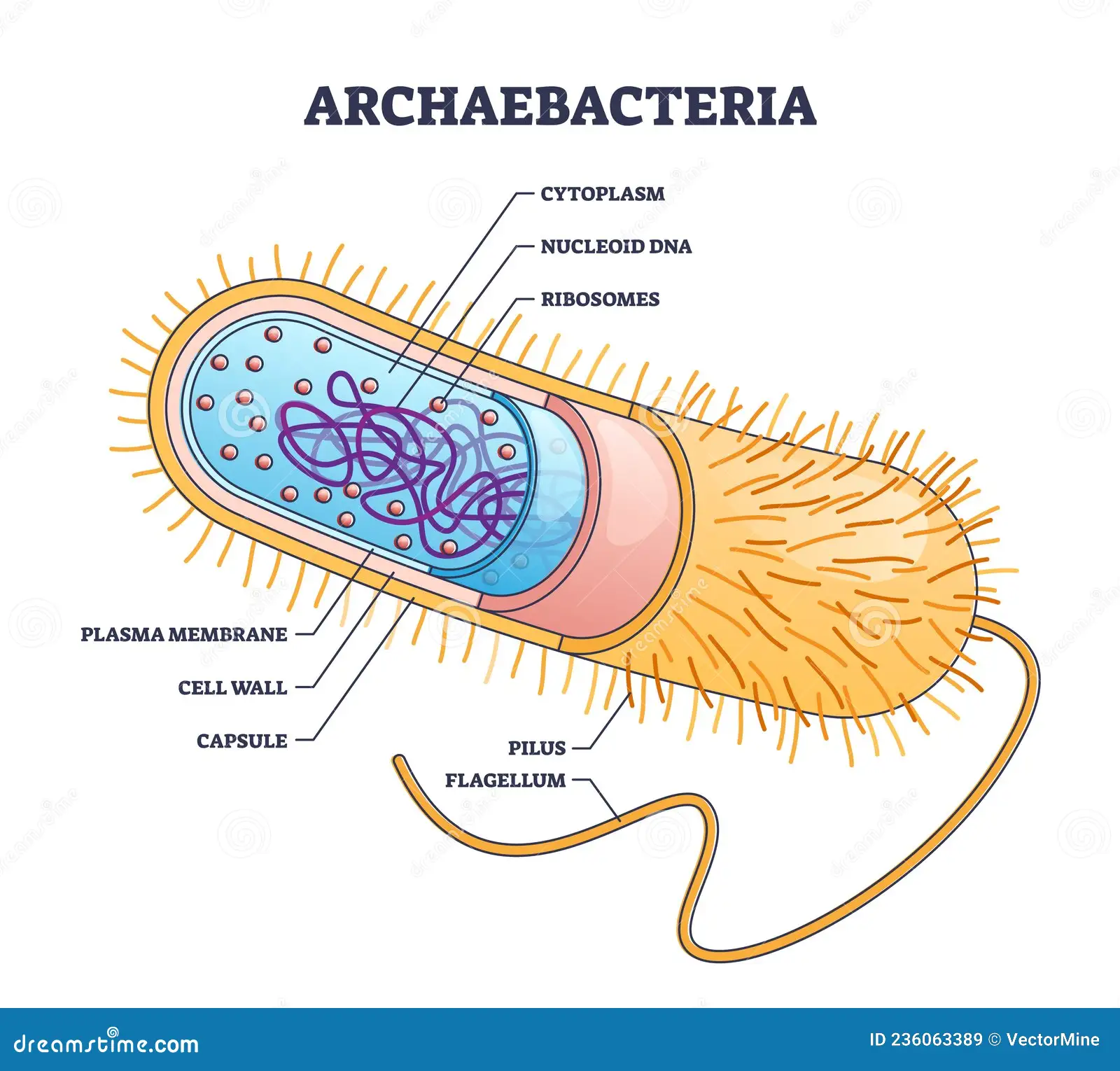
Bacteria/Archaea Cell Structure
Flagellum
Pili
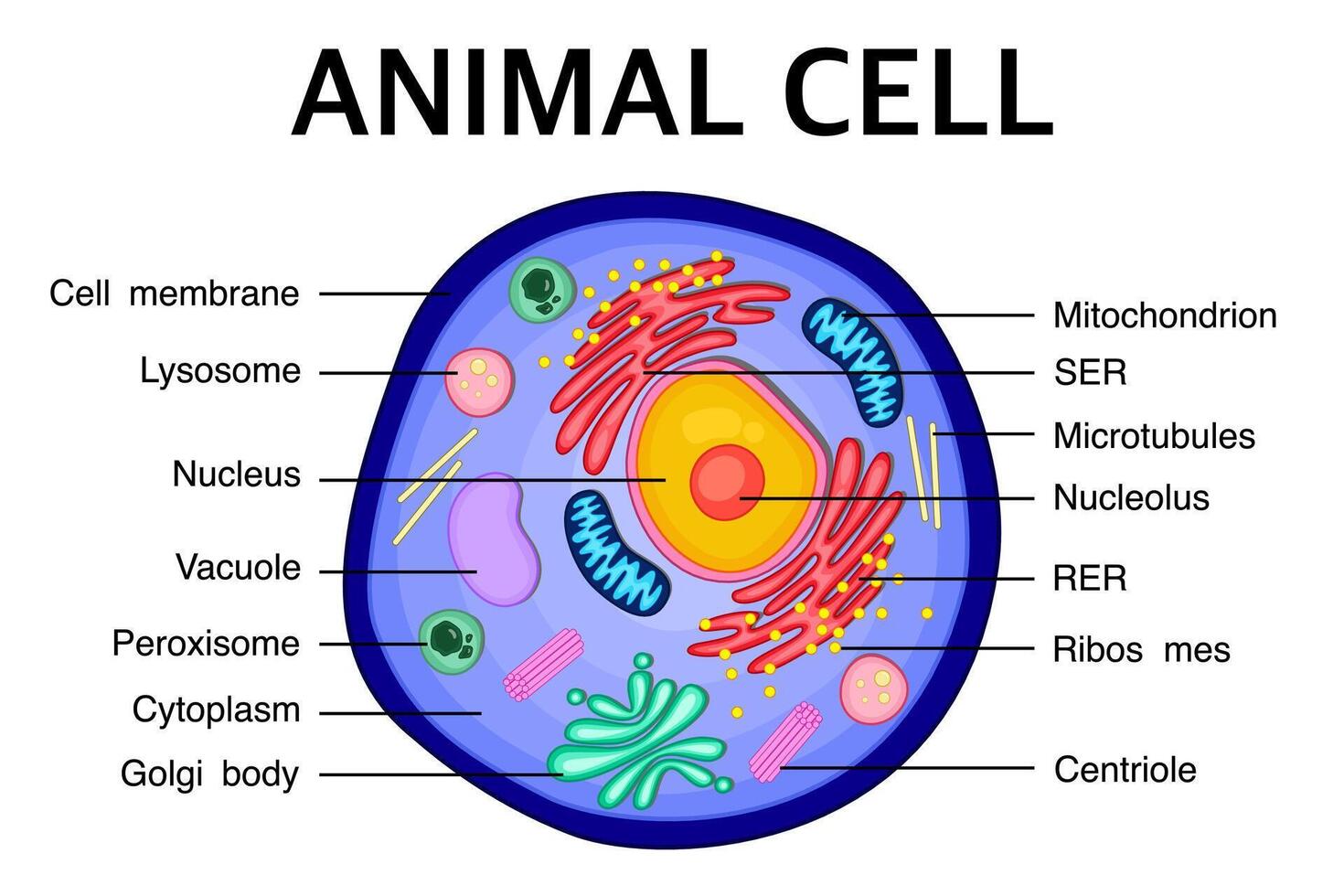
Animal Cell Structure
Lysosome
Centrosome
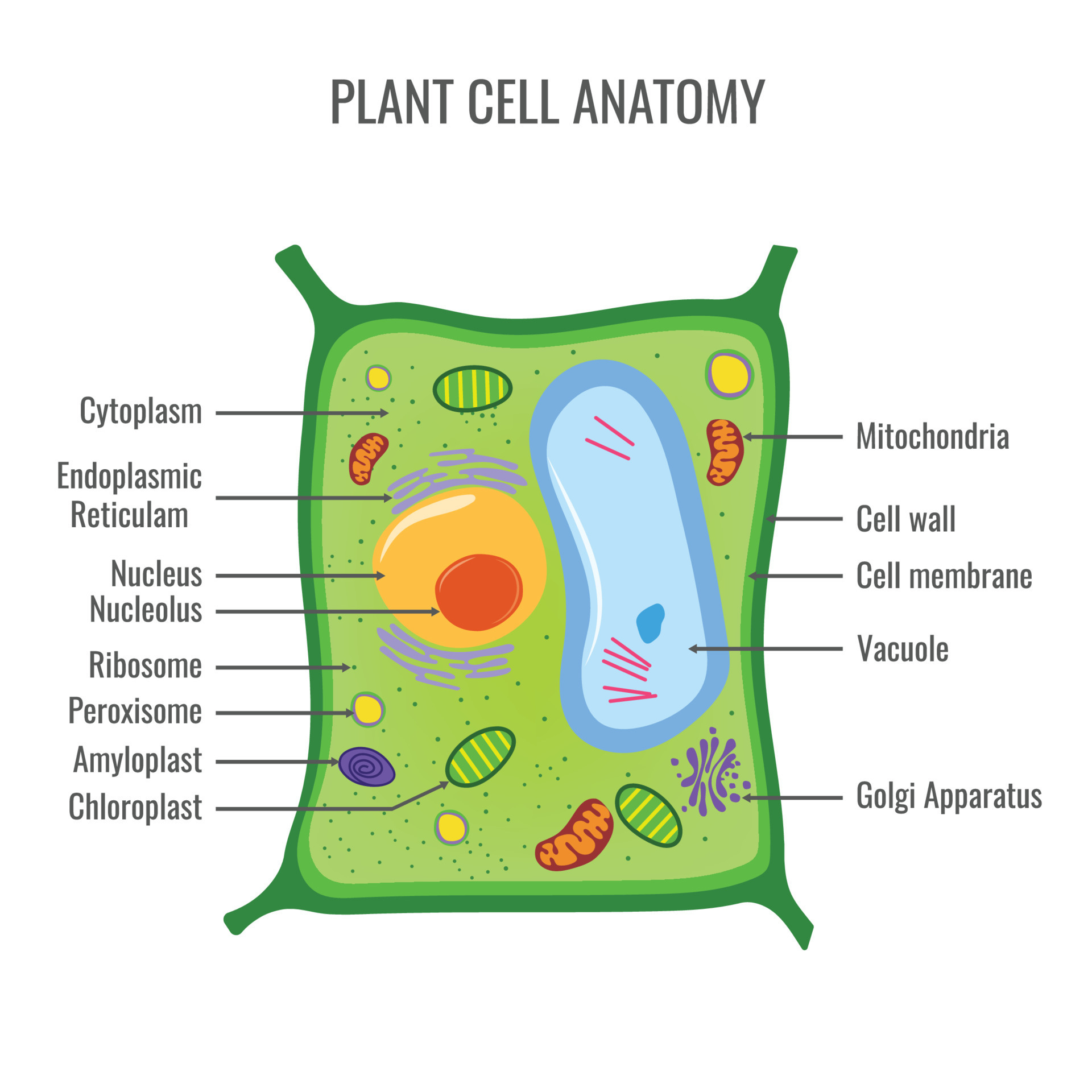
Plant Cell Structure
Cell wall
Central Vacuole
Chloroplast
Plasmodesmata
Plasma Membrane
Phospholipid Bilayer
Folds increase cell absorption
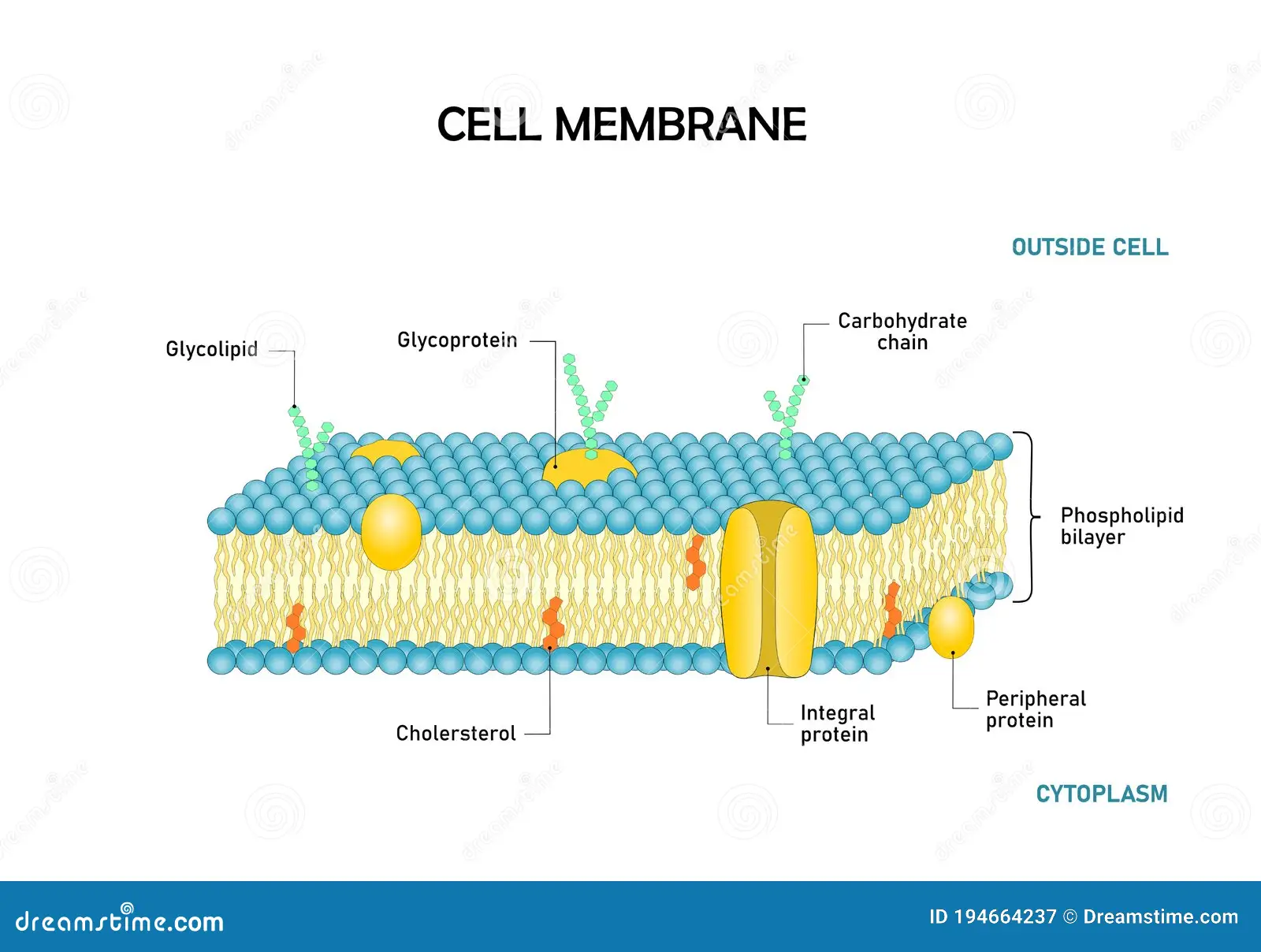
Cytoplasm
Cytosol (fluid of the cell) + organelles
Ribosome
Site of protein synthesis (found in ER)
Nucleus, Nucleoplasm, & Nuclear Envelope
Contain DNA
Nucleolus - site of ribosome biogenesis
Chromosomes
Made of DNA = condensed chromatin
Chromatin (unwound protein-chromosomes)
Centrosome
9 microtubule triplets
Help with cell division
Lysosome
Acidic organelle that breaks down macromolecules (garbage disposal)
Peroxisome
Detoxification
Oxidize fatty acids and amino acids
Produce and break down hydrogen peroxide
Mitochondria
Site of ATP Synthesis
Has own DNA and ribosomes
Chloroplast
only in plants
contain chlorophyll pigments for photosynthesis
has own DNA and ribosomes