Proteins
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
sulfur used in amino acids
formation of disulfide bonds
covalent links between two cysteine amino acids
crucial for stability in tertiary and quaternary structure
primary structure
the number and sequence of amino acids in a polypeptide, determined by the base sequence of the gene that codes for the polypeptide
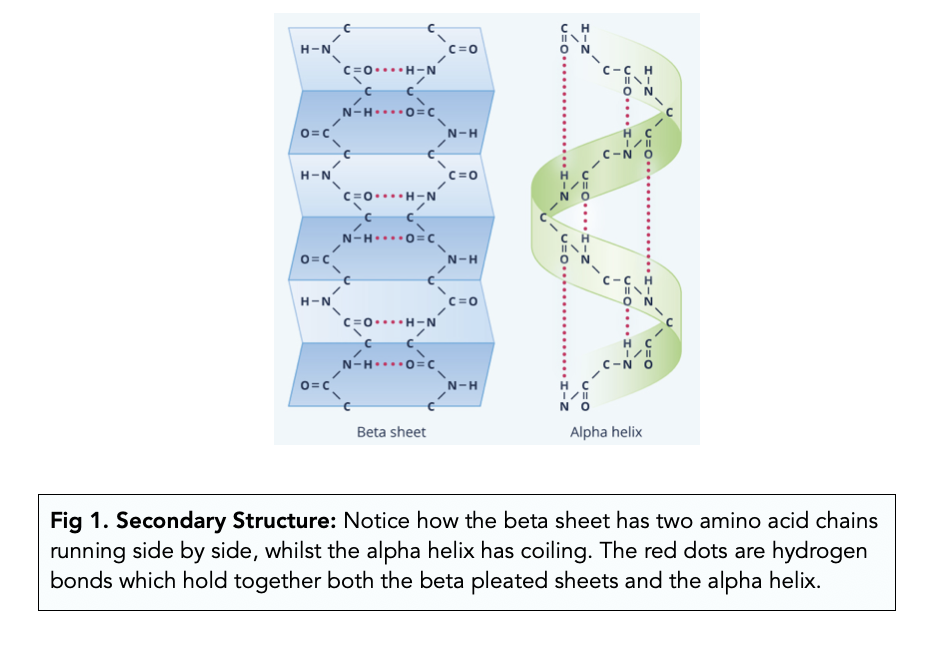
secondary structure
held together by hydrogen bonds between oxygen of carbonyl groups (C=O) and hydrogen atoms of an amine group in the polypeptide backbone
alpha helix
beta pleated sheet
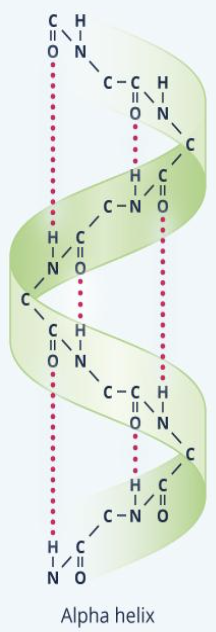
alpha helix
right-handed helix in which hydrogen bonds can form adjacent turns of the helix
beta pleated sheet
amino acid chains connected laterally by at least two or three backbone hydrogen bonds, forming a generally twisted, pleated sheet
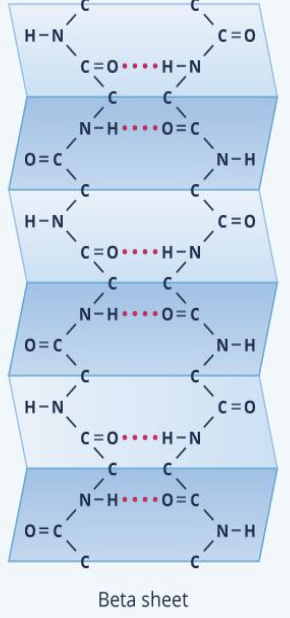
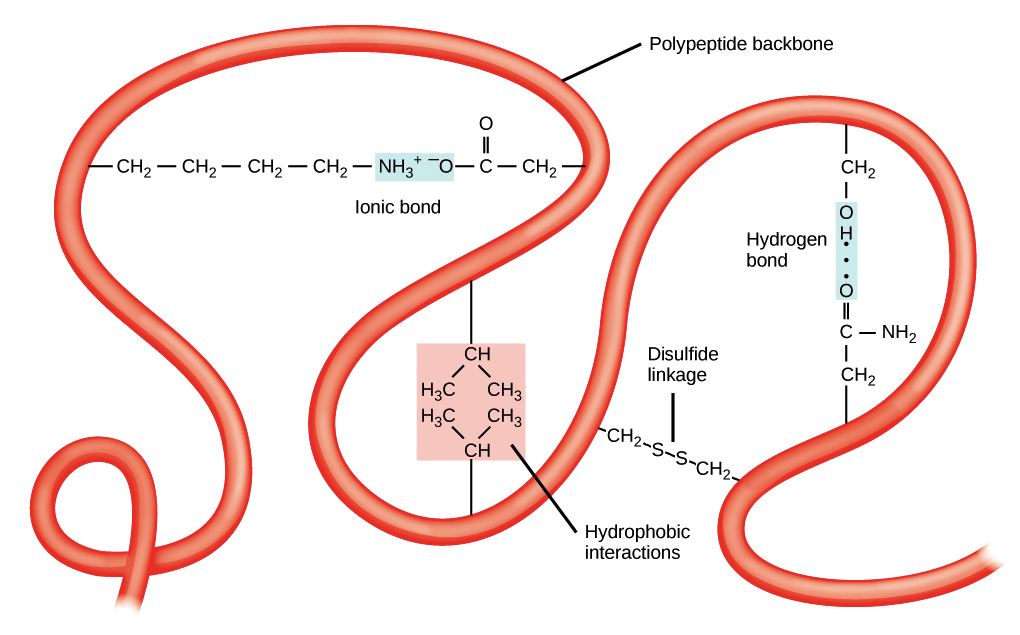
tertiary structure
folding of a single protein/polypeptide chain into a 3D structure with a hydrophobic core, giving proteins their functional properties (ex. active sites on enzymes), stabilized by:
interactions between R-groups of amino acids
ionic bonds
hydrogen bonds
hydrophobic bonds
disulfide bridges
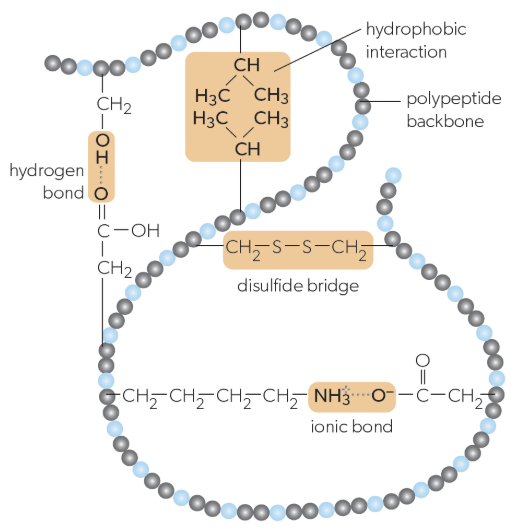
ionic bonds in tertiary structure
can form between positively and negatively charged R-groups
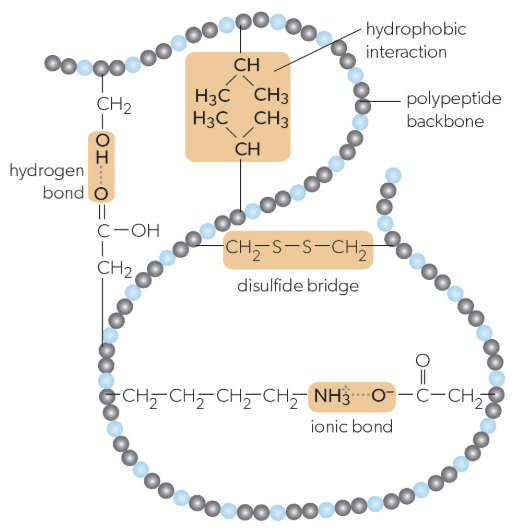
hydrogen bonds in tertiary structure
can form between some polar r groups
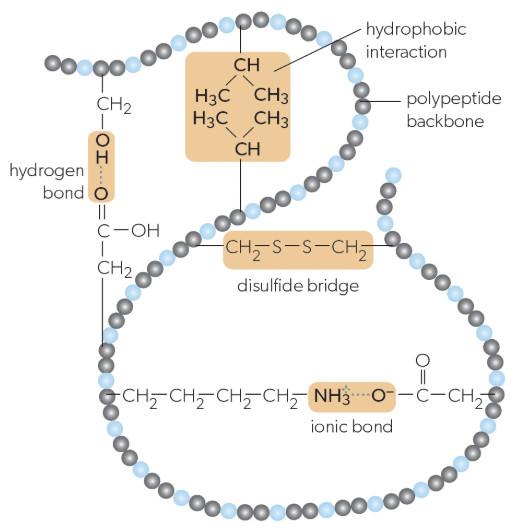
hydrophobic bonds in tertiary structure
weak chemical interactions that can form between non-polar R-groups
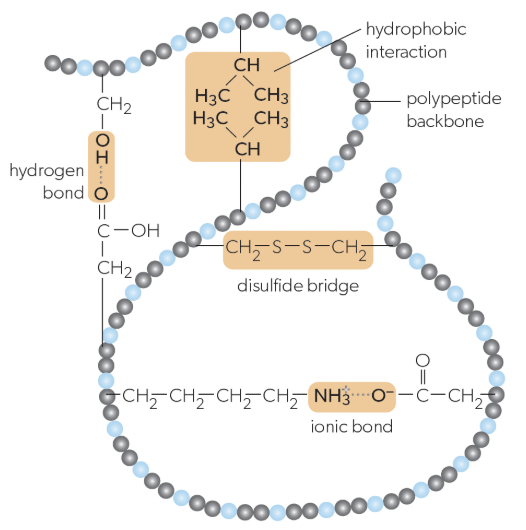
disulfide bridges in tertiary structure
strong bonds can form between pairs of cysteine
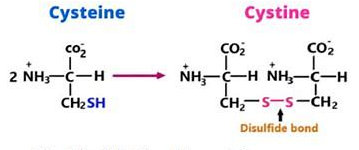
examples of tertiary structure
channel proteins and integral proteins
channel proteins
inside lined by polar amino acids
held in a transmembrane position by hydrophobic amino acids
allow hydrophilic molecules like water or ions to pass through with ease
integral proteins
polar amino acids on the outside extending into the extracellular fluid, associating with polar water molecules
non-polar amino acids anchor the protein into the membrane, associating with hydrophobic parts of the membrane
quaternary structure of proteins
how different polypeptides are arranged together (contains more than one polypeptide) by the same types of interactions contributing to tertiary structure (hydrogen bonding, London dispersion forces)
conjugated proteins
one or more non-polypeptide subunits in addition to their polypeptides
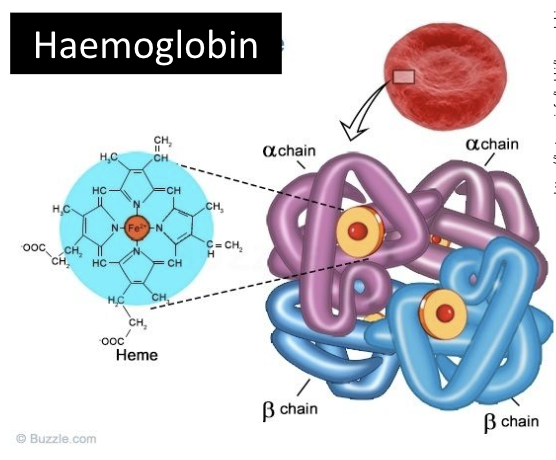
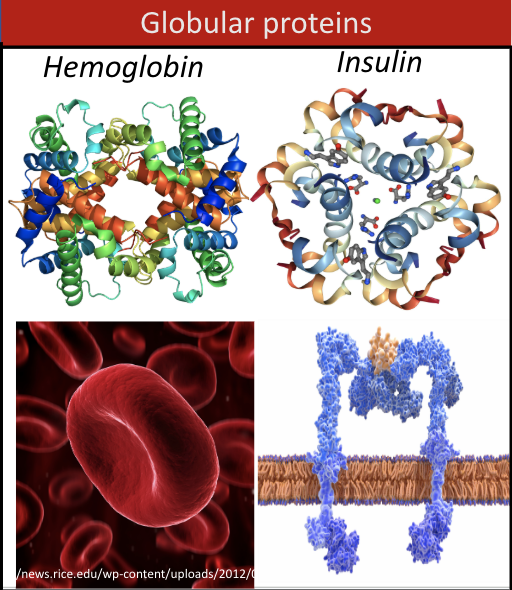
hemoglobin as an example of a conjugated protein
oxygen carrying protein consisting of four polypeptides
each subunit contains one iron atom/heme (prosthetic group) and can carry one molecule of oxygen
non-conjugated proteins
structure composed only of polypeptides
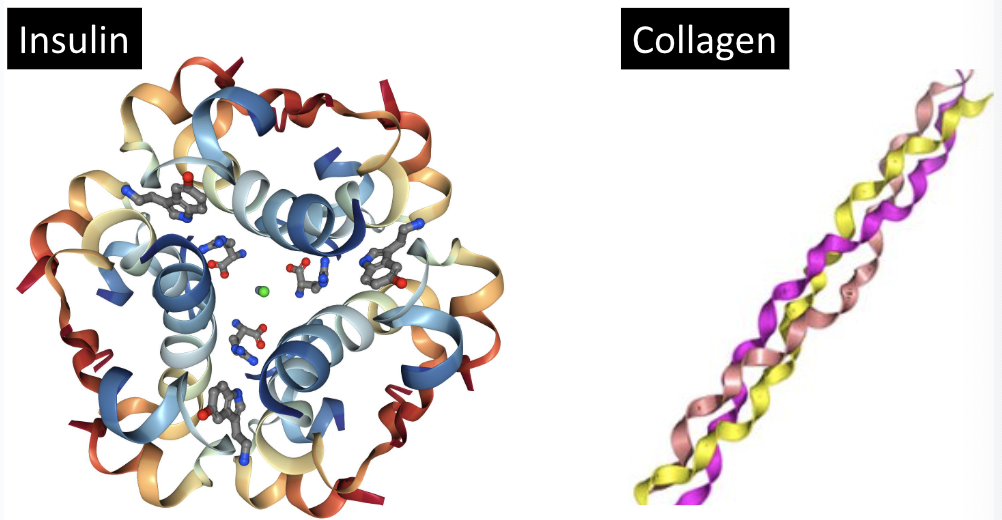
ex. insulin, collagen
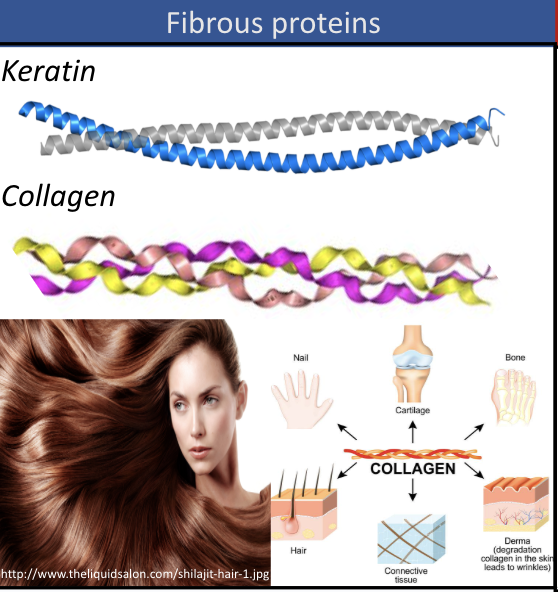
fibrous proteins
tube-like, structural protein (ex. keratin, collagen)
globular proteins
round, traveling, or transmembrane proteins (ex. hemoglobin, insulin)