ELECTRODE POTENTIALS
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
What is a cell?
made up of 2 half-cells
connected with a salt bridge (KNO3)
metal electrodes and a solution of a compound containing the metal
Why does a voltage formed (in a zinc, copper cell)?
Zinc has more of a tendency to oxidise to the Zn2+ ion and release electrons
more electrons will build up
a potential difference is created
Why do you use a high resistance voltmeter?
stops the current from flowing in the circuit, meaning its possible to measure the maximum possible potential difference
What is a salt bridge?
used to connect up the circuit (free moving ions conduct the charge)
made from a piece of filter paper soaked in potassium nitrate solution
salt should be unreactive with the electrodes and electrode solutions
What happens if current is allowed to flow?
occurs if the voltmeter is removed and replaced with a bulb
reactions will occur separately at each electrode
voltage will fall to zero as the reactants are used up
What happens at the positive electrode?
reduction
What happens at the negative electrode?
oxidation
What’re the rules for electrochemical cell representation?
solid vertical line represents different phases
double line represents the salt bridge
more positive half cell is written on the right
oxidised form is put next to the double line
What is the electrochemical cell standard representation for Zn and Cu?
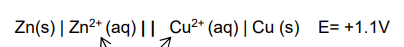
What is used if a system doesn’t include a metal?
a platinum electrode, which provides a conducting surface for electron transfer
What’re properties of a platinum electrode which means they can be used?
unreactive
conducts electricity
What is the standard hydrogen electrode?
potentials of electrodes are measured by comparing their potential to the standard hydrogen electrode potential
potential of 0V
What is the hydrogen electrode equilibrium?
H2(g) —> 2H+ (aq) + 2e-
What is the cell representation of the standard hydrogen electrode?
Pt| H2(g) | H+(aq)
What’re the conditions of the standard hydrogen electrode?
hydrogen gas at a pressure of 100kPa
solution containing hydrogen ions at 1.0mol dm^-3 (from HCl)
temperature of 298K
platinum electrode
Why are secondary standards used?
the standard hydrogen electrode is difficult to use so different standards are used which are easier
they’re calibrated against the standard hydrogen eelctrode
What is the standard electrode potential?
the potential difference measured when an electrode system is connected to the hydrogen electrode system and standard conditions apply
What’re standard conditions of a cell?
all ion solutions at 1 moldm ^-3
temperature of 298K
gasses at 100kPa
no current flowing
What happens to the more positive cell in a cell equation?
it will always go forwards
What does a more positive E mean?
more likely to be reduced and act as oxidising agents
What does a more negative E mean?
more likely to be oxidised and act as reducing agents
What would be the exam style answer to ‘use electrode data to explain why fluorine reacts with water. Write an equation for the reaction that occurs?
As E F2/F- > E O2/H2O, F2 will oxidise H2O to O2
2F2(g) + 2H2O (l) —> 4F- (aq) + 4H+ (aq)
What happens to the Ecell if the current is allowed to flow?
the cell reaction will occur and the Ecell will fall to zero as the reaction proceeds and the reactant concentrations drop
What does a more positive Ecell mean?
more likely the reaction is going to occur
What happens to the Ecell when the concentration of reactants increases?
increases E cell
What happens to Ecell if you increase the temperature on an exothermic cell?
causes a decrease in Ecell as the equilibrium reaction would shift backwards
What does a positive Ecell indicate?
the reaction might occur
if the reaction has a high activation energy the reaction will not occur
What are the different types of cells?
non-rechargeable
rechargeable
fuel cells
What makes a cell non-rechargeable?
when the reactions that occur within them are non-reversible
What makes a cell rechargeable?
forward reaction occurs on discharge giving out charge
charging causes the reaction to reverse
What’re fuel cells?
use the energy from the reaction of a fuel with oxygen to create a voltage
What is the equations at the electrodes for the hydrogen fuel cell in alkaline conditions?
negative : H2 + 2OH- → 2e- + 2H2O
positive : 4e- + 2H2O + O2 → 4OH-
overall : 2H2 + O2 → 2H2O
Why do fuel cells maintain a constant voltage overtime?
they’re continuously fed with fresh O2 and H2, maintaining constant concentration of reactants
What conditions are used in hydrogen fuel cells?
higher temperatures which increase the rate however causes Ecell to fall as the reaction is exothermic
higher pressure will counteract this
What’re advantages of fuel cells over petrol or diesel powered vehicles?
less pollution and CO2
greater efficiency
What’re limitations of hydrogen fuel cells?
expensive
hard to store and transport hydrogen safely
limited lifetime
high production costs
use of toxic chemicals in production
How can hydrogen be stored in fuel cells?
liquid under pressure
absorbed on the surface of a solid material
absorbed within a solid material
What’re advantages to ethanol fuel cells?
ethanol can be made from renewable sources in a carbon neutral way
raw materials to produce ethanol by fermentation are abundant
ethanol is less explosive and easier to store than hydrogen
new petrol stations would not be required
What’re the half cell and full equation to represent a litium ion cell?
negative = Li —> Li+ + e-
positive = e- + Li+ + CoO2 —> Li+[CoO2]-
overall= Li + Li+ + CoO2 —> Li+ + Li+[CoO2]-