Fundamental Chemistry
1/98
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
99 Terms
Elements with different ion charges
Iron → Fe²⁺, Fe³⁺
Copper → Cu⁺, Cu²⁺
Cobalt → Co²⁺, Co³⁺
Chromium → Cr²⁺, Cr³⁺, Cr⁶⁺
Hydrogen
H (atomic number 1)
Helium
He (atomic number 2)
Lithium
Li (atomic number 3)
Beryllium
Be (atomic number 4)
Boron
B (atomic number 5)
Carbon
C (atomic number 6)
Nitrogen
N (atomic number 7)
Oxygen
O (atomic number 8)
Fluorine
F (atomic number 9)
Neon
Ne (atomic number 10)
Sodium
Na (atomic number 11)
Magnesium
Mg (atomic number 12)
Aluminum
Al (atomic number 13)
Silicon
Si (atomic number 14)
Phosphorus
P (atomic number 15)
Sulfur
S (atomic number 16)
Chlorine
Cl (atomic number 17)
Argon
Ar (atomic number 18)
Potassium
K (atomic number 19)
Calcium
Ca (atomic number 20)
Scandium
Sc (atomic number 21)
Titanium
Ti (atomic number 22)
Vanadium
V (atomic number 23)
Chromium
Cr (atomic number 24)
Manganese Mn (atomic number 25)
Iron
Fe (atomic number 26)
Cobalt
Co (atomic number 27)
Nickel
Ni (atomic number 28)
Copper
Cu (atomic number 29)
Zinc
Zn (atomic number 30)
Acetate
C₂H₃O₂⁻
Nitrite
NO₂⁻
Carbonate
CO₃²⁻
Sulfate
SO₄²⁻
Phosphate
PO₄³⁻
Permanganate
MnO₄⁻
Hypochlorite
ClO⁻
Dichromate
Cr₂O₇²⁻
Hydrogen carbonate (bicarbonate)
HCO₃⁻
Formate
HCO₂⁻
Sulfite
SO₃²⁻
Nitrate
NO₃⁻
Peroxide
O₂²⁻
Hydrogen sulfate (Bisulfate)
HSO₄⁻
Perchlorate
ClO₄⁻
Ammonium
NH₄⁺
Cyanide
CN⁻
Hydroxide
OH⁻
What is matter
anything that has mass and occupies space
what are the 2 main types of matter
pure substances and mixtures
what is a pure substance
a matter with a fixed (constant) composition and definite properties. elements and compounds
what is an element
a substance made of only one type of atom
what is a compound
a substance made of two or more different types of atoms, chemically bonded in a fixed ratio
what is a mixture
matter with variable composition made by physically combining substances
what are the two types of mixtures
homogeneous and heterogeneous mixtures
what is a homogeneous mixture
a mixture with a uniform composition throughout, in a single phase and cannot be separated easily. Often called solutions
what is a heterogeneous mixture
a mixture with a non-uniform composition, has different states which are not uniformly distributed.
what are the main states of matter
solid, liquid, gas
what is a phase change
a physical change where matter changes between states
what are fluids
substances that can flow and occupy the space they are in. Liquids and gases.
what are the 2 types of changes that matter an undergo
physical changes and chemical changes
physical change
a change in form or state that does not alter chemical composition
chemical change
a change where substances are transformed into new chemical species through a reaction
structure of an atom
a central nucleus of protons (positive) and neutrons (neutral) surrounded an electrons (negative). most of the mass is in the nucleus, however the electron cloud is larger than the nucleus, but most of it is empty space (vacuum)
what is an ion
an atom or molecule with a net electric charge
what is a cation
positively charged ion (lost electrons)
anion
a negatively charges ion (gained electron)
what is an isotope
atoms of the same element that have the same number of protons but different number of neutrons, resulting in different mass numbers. Can be radioactive → have an unstable nucleus and undergoes spontaneous decay
what is a molecule
a collection of two of more atoms chemically bonded together
what are reactants
starting substances in a chemical reaction
what happens during a chemical reaction
chemical bonds are broken and/or formed
what are products
new substances formed from a chemical reaction
Law of conservation of mass
mass is neither created nor destroyed in a chemical reaction
Law of definite proportions
a compound always has the same elements in the same ratio by mass
Atomic Number
symbol Z. the number of protons in the nucleus, identifies the chemical element.
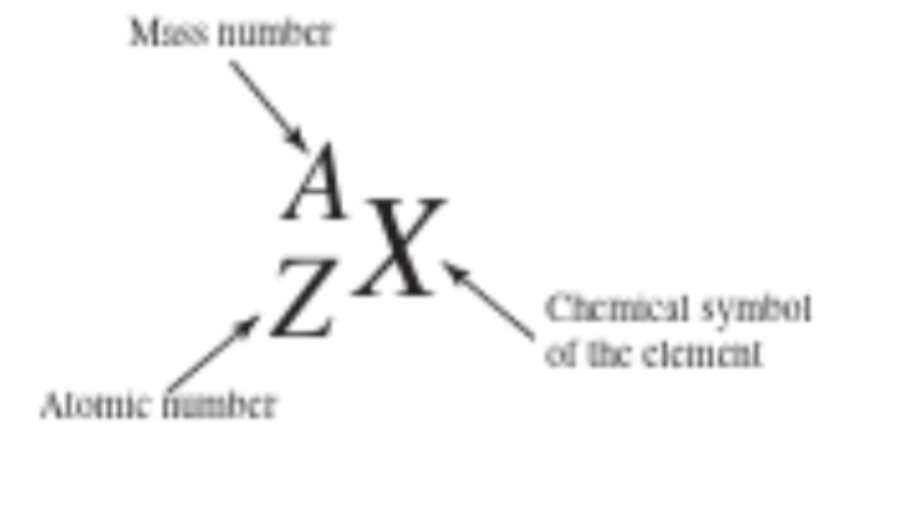
Mass number
Symbol A. The total number of protons + neutrons
the atomic mass unit
It is 1/12 the mass of a carbon-12 atom. u = 1.66054 × 10⁻²⁷ kg
what is the atomic mass of an element (relative atomic mass)
the weighted average mass of naturally occurring isotopes of an element.
What is light
a form of energy and electromagnetic radiation, travels as waves
characteristics of waves
wavelength(λ) the distance between two peaks of a wave. in metres
frequency(v): number of waves passing a point per second (measures in Hz or s⁻¹
What are the two key equations for light and waves?
change in energy = hv (Planck’s constant? x frequency
change in energy = hc / λ (Planck’s constant x speed of light / wavelength)

speed of light
symbol c. 2.999 × 10⁸ m/s.
planck’s constant
symbol h = 6.626x10^-34
what is a continuous spectrum.
A spectrum that contains all wavelengths of light with no gaps (a full range of colours)
What is a line spectrum?
A spectrum that contains only specific wavelengths (lines), not a continuous range.
what is an emission spectrum
a type of line spectrum. Light emitted by excited atoms as electrons fall to lower energy levels. every element has a pattern of colours unique for that element.
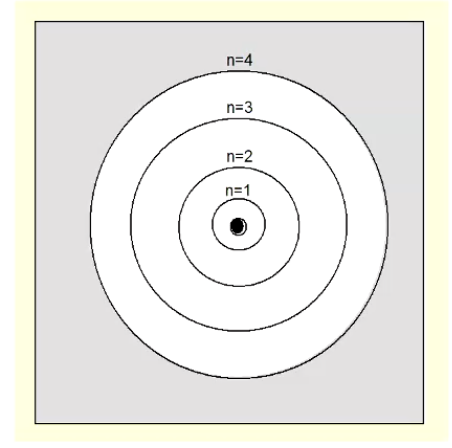
Bohr Model of Atom
electrons move in fixed circular orbits around the nucleus
each orbit with integer n
higher n = higher energy and larger orbit
each orbit can only hold 2n² electrons
when electrons move between orbits, light gets emitted/absorbed by atoms
Bohr energy equation for hydrogen like atoms (one electron)
Eₙ = −2.179 × 10⁻¹⁸ J × (1 / n²)
Quantum Mechanical Model
probability of finding electrons in regions around the nucleus
model has shells, subshells and orbitals
orbital: place that can hold 2 electrons
subshell: group of orbitals, hold 1 or 3 or 5 or 7 orbitals
shell: collection of subshells
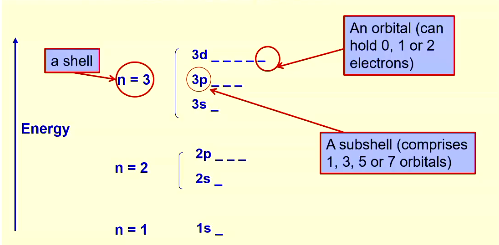
what is an orbital
region of space where up to 2 electrons are likely to be found. a probability distribution.
how does there quantum model differ from Bohr’s model
electrons do not move in orbits, but exist in probability clouds/orbits
what are shells labelled as
n = 1,2,3…
what are the subshell types?
s = 1 orbital, 2 electrons
p = 3 orbitals, 6 electrons
d = 5 orbitals, 10 electrons
f = 7 orbitals, 14 electrons
g = 9 orbitals, 18 electrons
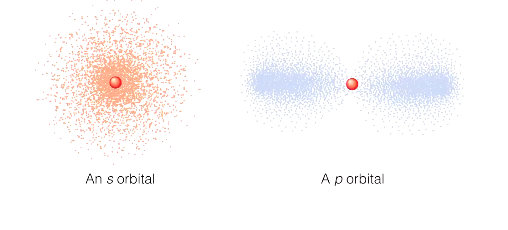
probability cloud.
orbitals have different shapes, electrons are most likely to be found in some regions than others.
many dots = electron likely to be
no dots = electron unlikely to be
Bohr vs Quantum
Bohr: line emission spectrum for hydrogen like atoms = one electron systems, simpler energy calculation
Quantum: more than one electron, describes probability clouds and not fixed orbits. Describes electron configuration with detail. Mathematically complex and hard to visualise.
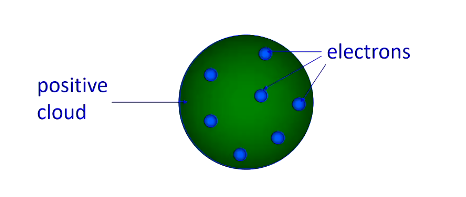
Thompson/Plum Pudding Model
Electrons embedded in a positively charged sphere. Did not include a nucleus. Could not explain why alpha particles passed through gold foil with minimal deflection (thus positive charge must be concentrated not diffused)
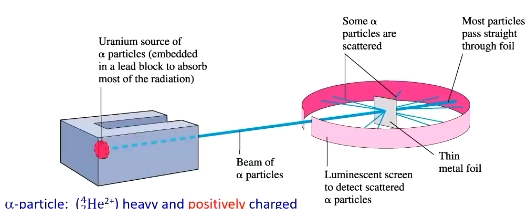
Rutherford’s gold foil experiment
Used alpha (very positive) particles to pass through a gold foil
most particles passed straight through, small number deflected
conclusion: most atom is empty space with a small positively charged central nucleus
Naming covalent compounds
first element keeps its full name
suffix -ide
use prefixes to show number of atoms