Analytical Chemistry
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
CAPECCE
Choosing a method
Acquiring the sample
Preparing the sample
Eliminating interferences
Calibration
Calculating the result
Evaluation of result
What are the 7 steps in Quantitative Analysis?
Reagent Grade
It is a type of chemical that conform to the minimum standards of purity and used whenevr possible in analytical work.
Primary-Standard Grade
It is a type of chemical that is used to standardize solutions.
SHALS
Stable
High purity
Absence of water
Large molar mass
Soluble in medium
What are the properties of a primary-standard grade?
Special Purpose Reagent Grade
It is a type of chemical that is used for specific instrument (e.g. LC-MS—grade)
Technical Grade Chemicals
It is a type of chemical that has no official standard for purity.
Practical Grade Chemicals
It is a type of chemical that is used for synthesis.
6 months
It is the maximum holding time for acid-preserved samples that will be subjected to determination of chemicals.
Sulfuric acid, nitric acid, and/or perchloric acid
In wet ashing, what are the strong acids and oxidizing agents used to break down and remove the organic matrix of the sample to leave the metals only.
3:1 mixture of HCl and HNO3
Aqua Regia is a combination of and in what ratio?
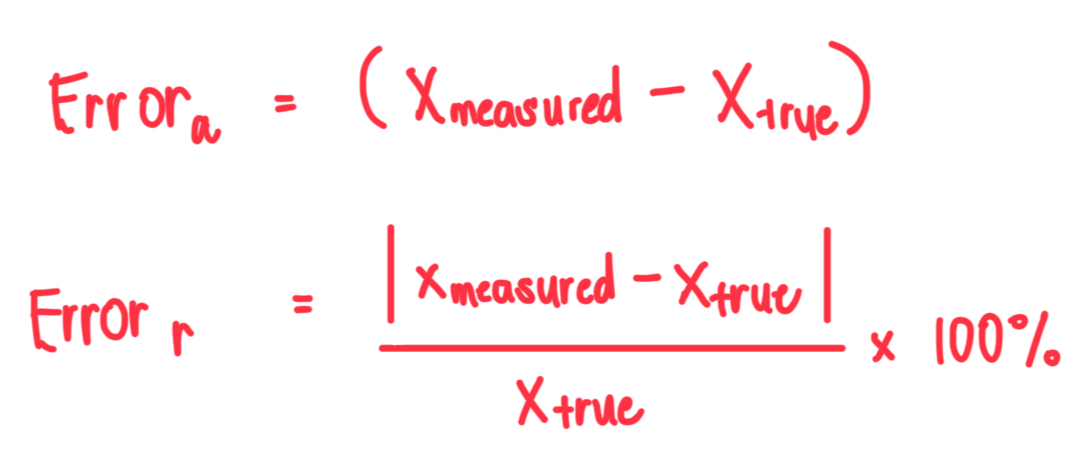
What is the difference between absolute and relative error?
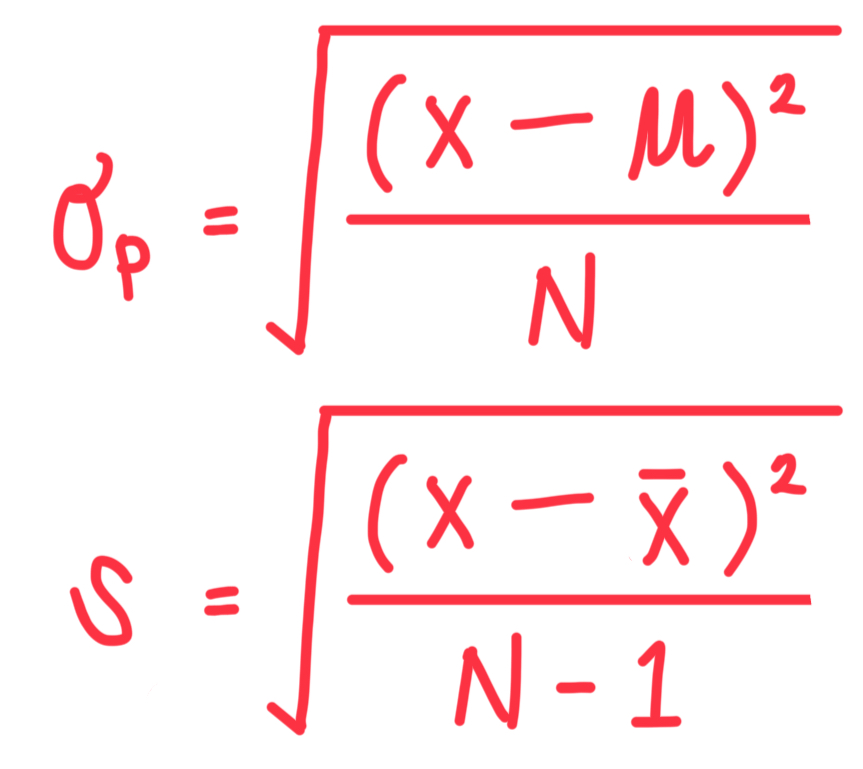
What is the formula of standard deviation for (a) population and (b) sample?
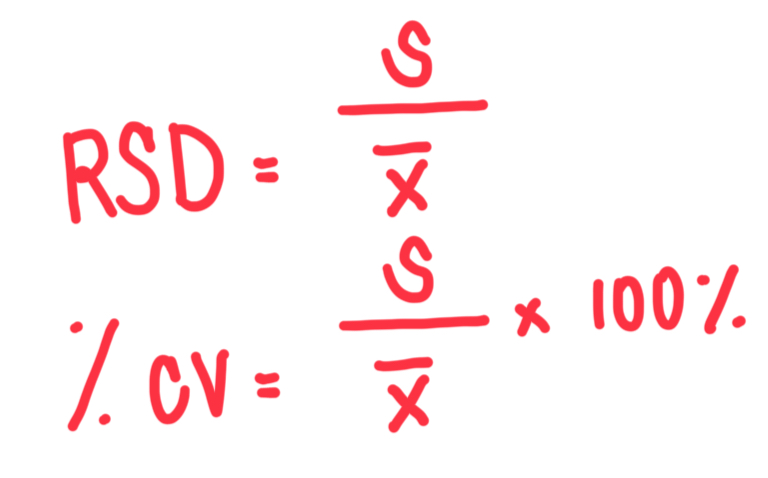
Formula for relative standard deviation (RSD) and coefficient of variation?
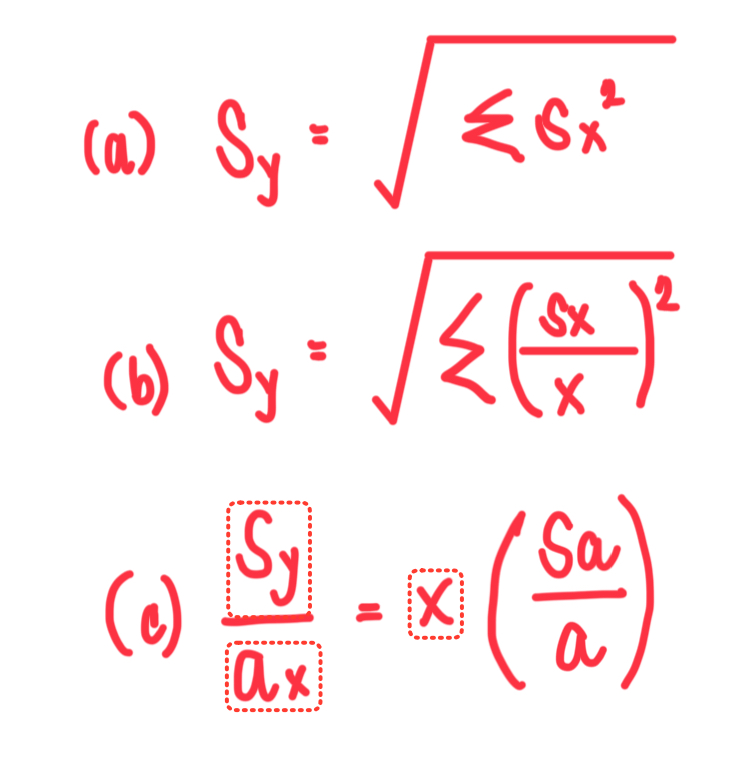
N
What is the formula for uncertainty in (a) sum/difference, (b) product/quotient, and (c) exponential calculations
Instrument blank
It is a type of blank used to establish the baseline of the instrument. It is done by determining the signal without sample added.
Solvent Blank
It is a type of blank used to measure the signal of the solvent.
Method blank
It is a type of blank used to detect contamination from method.
Z-Test
It is a statistical test used when population standard deviation is known and when sample size is large.
T-Test
It is a statistical test used when population standard deviation is unknown and when sample size is small.
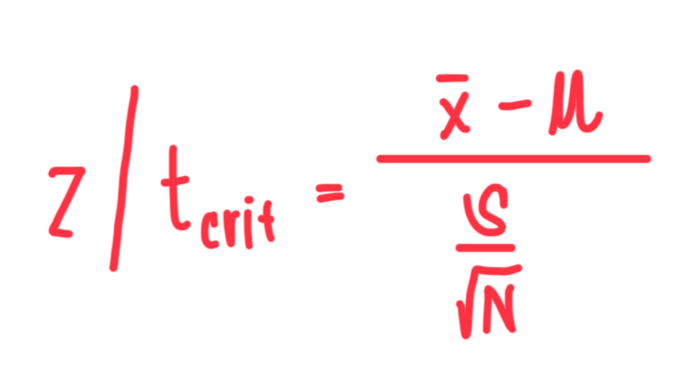
Formula for z and t-tests statistical value
Reject
When z/t > z/t (critical), do we accept or reject null hypothesis?
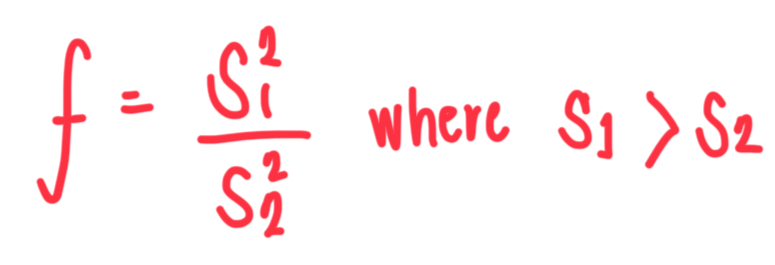
What is the formula for f value in f-test?
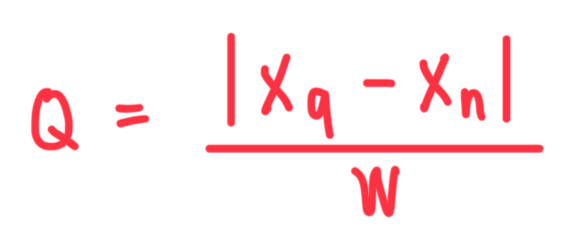
What is the formula for Q in Q Test for outlier?
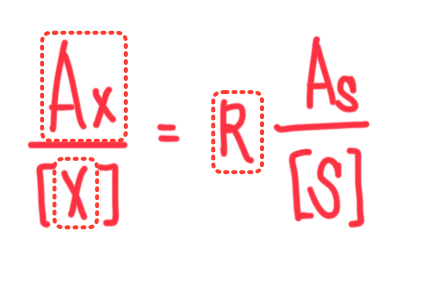
Formula for Internal Calibration
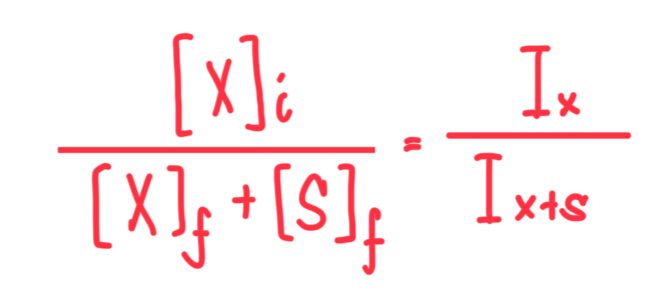
Formula for Single-Point Standard Deviation
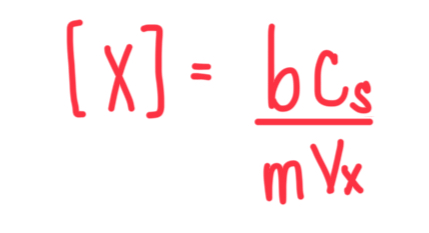
Formula for Multiple-Point Standard Deviation
Limit of Detection
It is the smallest concentration from zero that can be reported with a certain level of confidence.
Limit of Quantitation
It is the smallest concentration of a sample with a distinguishable response, and acceptable accuracy and precision.
LOD=3σ/m while LOQ=10σ/m
What is the formula for LOD and LOQ?
The equilibrium lies to the right
What does K>1 denote in chemical equilibrium?
Kp=Kc(RT)^n
What equation shows the relationship of Kp and Kc?
Products
When Q<K, the equilibrium favors ___.
Reactants
When Q>K, the equilibrium favors ___.
Lower
Based on Le Chatelier’s principle, when pressure increases, the equilibrium will shift towards ____ number of moles
Activity
It is the effective concentration of a substance in a system where it behaves like it is available to react.
Weak acid/base and its salt
A buffer solution is a combination of a _______ and _______.
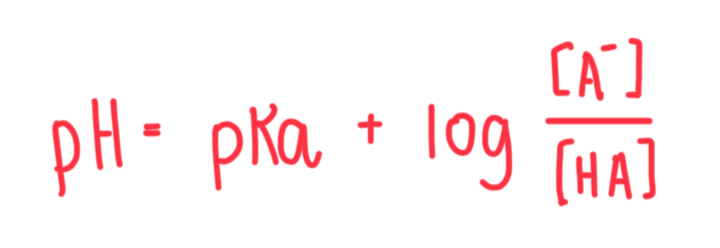
Formula for Henderson-Hasselbalch equation
Ionic strength tells how much of ions are present while activity tells how much an ion behaves like it is ready to react.
What is the difference between ionic strength and activity?
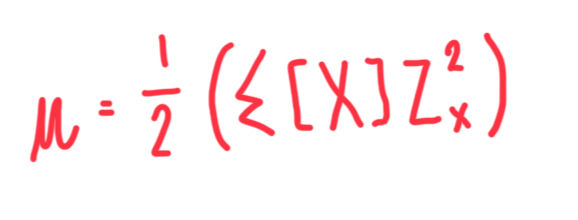
Formula of ionic strength
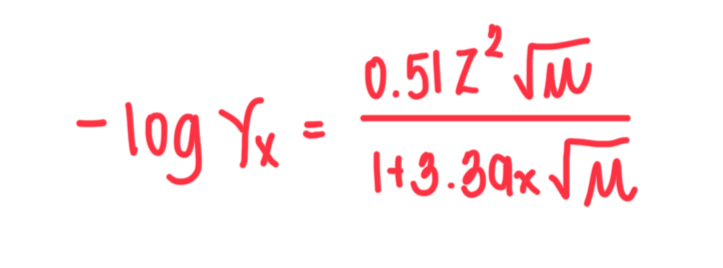
Formula of Debye-Huckel Equation
Na-Yellow (New York)
K-Purple (HKP)
Rb-Pink (RuBini)
What are the color of flames of the ff: (a) Na, (b) K, and (c) Rb?
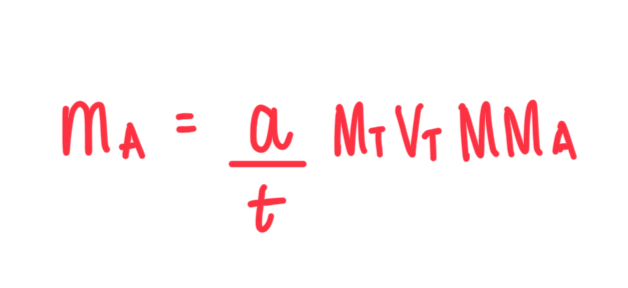
What is the shortcut formula for calculating the mass of analyte in titration?