Equilibrium constant Kp for homogeneous systems (3.1.10)
1/3
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
4 Terms
How is Kp deduced?
Kp is deduced from the equation for a reversible reaction occurring in gas phase. It is defined as:
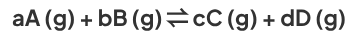
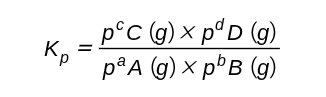
Where a, b, c and d and the respective number of moles of each species
Where p(x) is the partial pressure for each respective species
How do you calculate partial pressure?
1) Work out the moles at dynamic equilibrium of each species
2) Find the mole fraction of each species by dividing its moles over the total number of moles
3) Multiply each species' mole fraction by the total pressure
If a total pressure isn’t given it will probably cancel out - unitless Kp
When does the Kp of a reaction change?
When there is a change in temperature.
If the forward reaction is exothermic, increasing the temperature will decreases Kp
If the forward reaction is endothermic, increasing the temperature will increase Kp
How many significant figures should be used when there is a varying number of significant figures in the question?
Your answer should be quoted to the lowest number of significant figures in the question. This is because it can only be reported to the limits of the least accurate measure