Biotech II Lecture 11 - Protein Therapeutics
1/74
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
75 Terms
what are therapeutic proteins
proteins based on or inspired by naturally occurring proteins in the human body
types of therapeutic proteins
antibodies, enzymes, cytokines and hormones and more
why are therapeutic proteins important
size and complexity gives them advantage over small molecules when targeting extracellular or membrane associated proteins, modulating immune responses or replacing dysfunctional/missing proteins
why do therapeutic proteins often provide better specificity, reduced off-target effects and more natural regulatory effects
due to their structural similarity to endogenous proteins
proteins which are engineered in the laboratory for clinical uses
therapeutic proteins
an approach in treating diseases using proteins and peptides
protein therapeutics
delivers protein to the body in specific amounts to treat diseases and is similar to gene therapy
protein therapy
first recombinant protein therapeutic
insulin
timeline of protein therapeutic development
1920 - isolation of insulin
1980 - FDA approval of insulin and antibody muromonab
1990 - FDA approval of pegylated drug, enzyme and Fc fusion
2000 - FDA approval of ADC and lipidated protein
2010 - FDA approval of gycoengineered bispecific albumin fusion proteins
2020 - FDA approval over 100 antibody drugs
most widely used type of biologic
monoclonal antibodies (mAbs)
what do mAbs bind and how do they bind them
bind to a single target, antigen, and with high specificity
how are mAbs created
cloning a single type of immune cell so that all the antibodies produced are identical
what does mAbs high specificity allow them to do
bind only to disease related targets such as cancer cell markers or inflammatory proteins
due to their precision, mAbs can treat…
a variety of conditions, including cancers, autoimmune disorders and infections
what is herceptin (trastuzamab) used for and what does it do
HER2-positive breast cancer and it targets cancel cells with the HER2 protein, sparing normal cells
why are therapeutic enzymes often used
to replace enzymes that are missing or defective in patients with genetic disorders
what do therapeutic enzymes do
catalyze specific biochemical reactions, to aid digestion in patients with pancreatic insufficiency, target tumor cells by converting prodrugs to active drugs
why can enzymes be used to carry out precise therapeutic functions
they are highly specific
long-term enzyme replacement therapy (ERT) in patients with Pompe disease.
myozyme® (alglucosidase alfa)
what is pompe disease
glycogen storage disease type II causes by acid alpha glucosidase (GAA) deficiency
GAA
enzyme in lysosome that degrades glycogen
Fc fusion proteins consist of what
biologically active protein domain fused to the Fc region of an antibody
how do Fc fusion proteins extend the half-life of antibodies in the bloodstream
Fc region binds to receptors that recycle antibodies rather than degrade them
different types of Fc fusion proteins
monovalent hGh-Fc (just one hGH on top)
monovalent Fc-hGh (just one hGH on bottom)
dinvalent hGh-Fc (two on top)
dinvalent Fc-hGh (two on bottom)
dont think its that deep but u rmr if hgH comes first it’s on the top
enbrel® (etanercept)
a recombinant fusion protein, combining two soluble TNF receptor segments with the Fc portion of IgG1, used to treat rheumatoid arthritis
blocks RA inflammation and lasts longer in the body
acts as a soluble decoy receptor
TNF
tumor necrosis factor, a cytokine that promotes inflammation, joint destruction and stiffness
what are antibody-drug conjugates (ADCs) and how do they work
combine the targeting ability of mAbs w the potency of cytotoxic drugs
antibody part brings drug to specific cell type (cancer cell) and the drug/payload is released upon binding
enables targets delivery of chemotherapeutics, minimizing damage to healthy tissues
ADCetris® (brentuximab vedotin)
for hodgkin lymphoma; ADC that delivers toxic drug to cancer cells for targeted killing
these are essential for stopping bleeding and forming blood clots
coagulation proteins: factor VIII and factor IX
how are patients with blood factor deficiencies treated
receive replacement therapies made from donated plasma or recombinant proteins
fusion proteins of these factors last longer in the bloodstream
eloctate ® (efmoroctocog alfa, recombinant factore VIII protein)
treats hemophilia A
PDD FVIII (plasma derived factor 8) linked to IgG1 fc region
Fc interacts with FcRN, providing protection against lysosomal degradation
interferon vs interluekin
both are cytokines
interferons have antiviral activity and interleukins regulate immune cell communication and activation
what are therapeutic cytokines used for
conditions like multiple sclerosis, chronic viral infections and cancer
avonex (interferon beta- 1a)
modulations immune activity to slow nerve damage, used for multiple sclerosis
(Fear von bc he slows ur nerves)
what are subunit vaccines and which patients is it safer for
use purifies pieces of pathogen to stimulate an immune response without introducing live pathogens (hep b, hpv, covid)
safer for immunocompromised individuals
gardasil 9TM
protein subunit vaccine for HPV prevention, uses viral protein to train immune system without risk of infection
major class of chemical messengers in the body that can be lab made to restore normal function when out of balance
hormones
lantus
replaces natural insulin to regulate blood sugar in diabetes
well established protein modification strategies
site-specific mutagenesis, antibody-drug conjugates, PEGylation, Fc fusion, glycoenginerring, lipidation, albumin fusion or binding, computational
emerging chemical strategies on protein therapeutics
supercharging, attachment of non-PEG polymers, fusion to other partners, unnatural amino acids
desirable characteristics of protein therapeutics
stability, desired pk & pd, intracellular delivery
site specific mutagenesis
by changing amino acids in the protein sequence, make recombinant proteins have better pk profile, more stable, less immunogenic or better at binding to target
ex. betaseron® (interferon beta-1b), Lantus® (insulin glargine)
betaseron®
human interferon beta, cys 17 substitutes w serine, 165 residues, produced in e coli no carbs
albumin fusion/binding
therapeutic proteins can be directly fused to albumin or engineered to bind it non-covalenty
ex. albutrepenonacog alfa (idelvion®), caplacizumab
what does albumin fusion do to the drug
prolong the circulation time of drug, extend half life w reduced kidney filtration, protect from in vivo protease degradation, improve stability and solubility
what is PEG and what is pegylation
polyethlene glycol is a water soluble, biologically inert, non-immunogenic, synthetic polymer; pegylation is the chemical attachment of peg chains to proteins
what does pegylation do to the protein
increases size, hides them from immune sys, slows renal circulation → extends circulation time
improved pk and stability, reduced immunogenicity
adverse effects and disadvantage of pegylation
hypersensitivity reaction (anaphylaxis), anti-PEG antibodies, metabolite accumulation in the kidney impairing normal kidney function
not cost-effective
neulasta® (pegfilgrastin)
PEGylated form of the recombinant human granulocyte colony-stimulating factor (GCSF), stimulates production of wbcs/neutrophils to fight infection
used in cancer patients with chemother
antibody drug conjugate advantages
minimize systemic toxicity, targeted delivery
kadcyla® (ado-trastuzumabemtansine (T-DM1) with thioether linker)
antibody drug conjugate
adc moa
binding to cognate antigen
internalization of adc-antigen complex
lysosomal degradation of the antibody portion
release of payload/drug in cytoplasm
interaction with target (dna, microtubules, topoisomerase 1)
taken up by neighboring cells
Fc fusion protein
therapeutic protein can be fused to the Fc region of an antibody to extend its half-life and improve its stability, fc region interacts w recycking receptor and avoids degradation
Fc fusion protein advantages
extended half life
lower immunogenecity
improved stability
enhanced pk/pd
some can recruit immune cells to target cancer or other pathogens
fc fusion protein examples
etanercept (enbrel®)
aflibercept (eylea®)
dulaglutide (trulicity®)
glycoengineering
changing sugar moieties on proteins to influence immune system’s response or how long the protein stays active
glycoengineering advantages
improve protein folding, enhance receptor binding, reduce immunogenicity, important for antibodies and glycosylated proteins
improved pk and activity
enhanced antibody function
reduced side effects
diverse production methods
erythropoietin vs darbepoetin alfa
darbepoetin alpha has a longer half- life than rhEPO, given once in one or two weeks bc it is hyperglycosylated
lipidation
adding lipid (fatty acid) to a protein to help it stick to albumin → longer blood circulation, delays clearance and helps maintain steady dug levels
commonly used in insulin modification and hormone therapies
lipidation advantages
reversible, extended release, intracellular delivery
insulin detemir (levemir®) modification
has an added fatty acid chain (lipidation)
emerging and advanced strategies
xtenylation, multi-specific proteins (bi-/tri- specific), conditional activation (prodrug proteins), self assembling protein nanostructures, glycan masking and steering, engineered scaffolds (non-antibody formats)
XTEN
unstructured, hydrophilic and biodegradable synthetic polypeptide, has excellent water solubility and high chemical stability
how is xten synthesized
using recombinant DNA technology (pegylation is chemical modification)
what are the 6 amino acids in xten that are used to extend half-life and improve stability/solubility of therapeutic peptides and proteins
GGPAST
glycine, glutamine, proline, alanine, serine, threonine,
which amino acids are avoided when designing protein therapeutics to extend half-life and why
Hydrophobic amino acids: due to protein aggregation
Amide-containing amino acids: unstable upon long-term storage
Positively charged side chains: may cause binding to membranes
Cysteine residues: may become oxidized or cross-linked
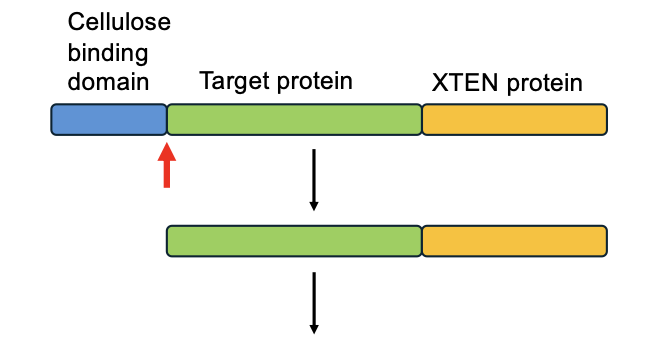
where is the encoding DNA of xten fused and where is the cellulose binding domain fused
dna is bound to 3’ end of target protein
cellulose binding domain is fused to therapeutic protein gene at 5’ end
how to remove the cellulose-binding domain from the target protein
digest it with a protease
how is xtenylation different from pegylation
its genetically encoded, uses long unstructured polypeptides, and is biodegrable (can be rapidly degraded upon internalization and reduces tissue accum/toxicity)
tunable, genetically encode its production and is more homogenous than products
xtenylation
how does xtenylation extend a proteins half-life
increases its size and reduces kidney clearance
xten proteins advantages
• Excellent water solubility
• Precise genetic encoding and expression of its sequence
• Unparalleled control over sequence fidelity and molecular weight
• Highly homogenous product
• XTEN fusion proteins are highly soluble,
properly folded in E. coli
• Non-immunogenic
• Biodegradable
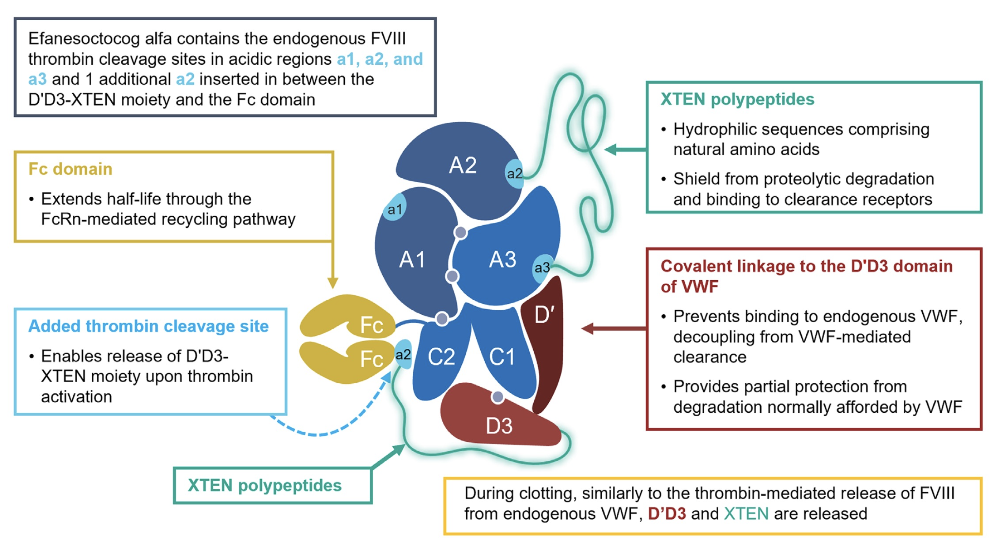
Efanesoctocog alfa (Altuviiio®)
protein therapeutic advantages
Highly specific
Less adverse effects
Protein replacement therapy
Clinical approval time could be faster than small molecule drugs.
protein therapeutic disadvantages
Solubility
Distribution
Stability
Route of drug delivery (iv, sc, im)
Physiologically active post translational modification
Cost and storage