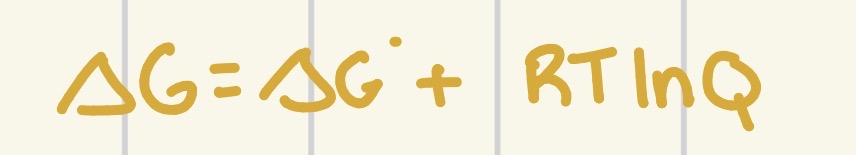Chem quiz 6
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
What is a reactant forward reaction?
When there are more reactants than products at equilibrium
What is a product forward reaction?
When there are more product than residents at equilibrium
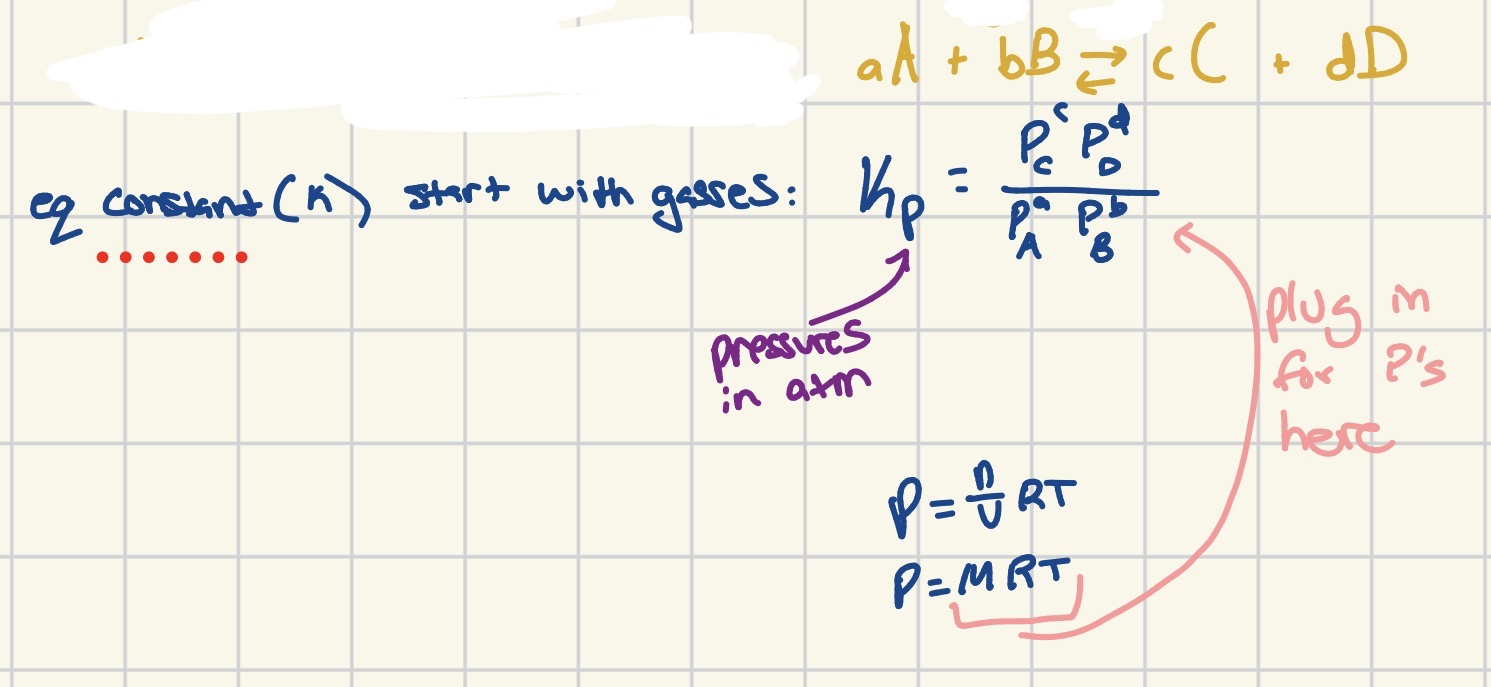
How do you calculate the equilibrium constant (Kp) for gases?
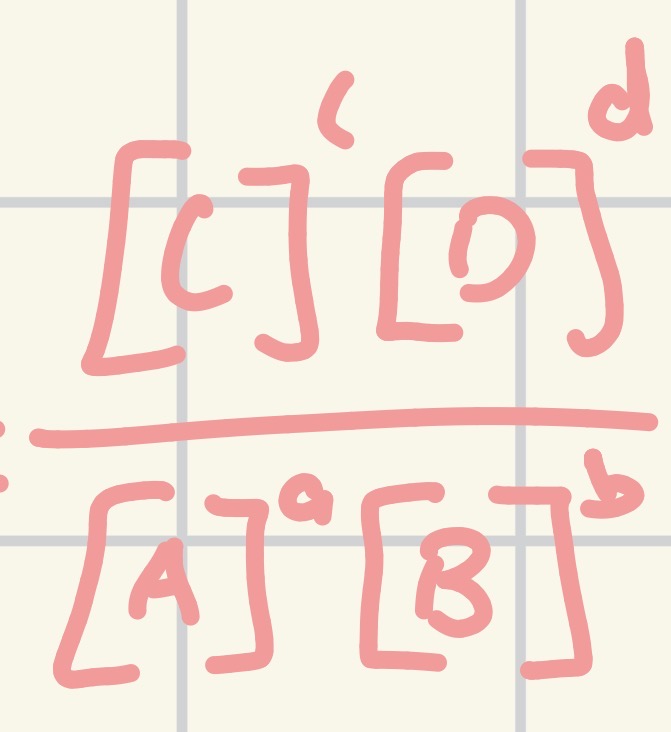
How do you calculate the equilibrium constant (Kc) for concentrations?
What does it mean if k< 1?
The reaction is reactant favored, if K < 10^-3 the reaction appears to be only reactants
What does it mean if k > 1?
The reaction is product favored, if K > 1000 the reaction appears to be only products
How do you write kp for an equation with substances or then than a gas?
Write 1 instead of the pressure for the liquid/solid

Fill out this table

What is the reaction quotient (q)?
It uses the same formulas as Kp or Kc and tells you where he reaction is at in relation to equilibrium
What does it mean if q=k?
We're at equilibrium
What does it mean if q < k?
You have more reactants currently and need to make more products to get to equilibrium
What does it mean if q > k?
You have more products currently and need to make more reactants to get to equilibrium
What formula do we use to relate delta G and delta G knot?