Insulin, Oral Hypoglycemics and Glucagon
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
How does insulin work in the body?
Insulin is released when usable energy (glucose) is present and signals the body to store that energy as glycogen and lipids.
What are the three chains of insulin? What peptide needs to be be cleaved for proinsulin to become active?
The three chains are chains A, B, and C. The C peptide needs to be cleaved for proinsulin to be active.
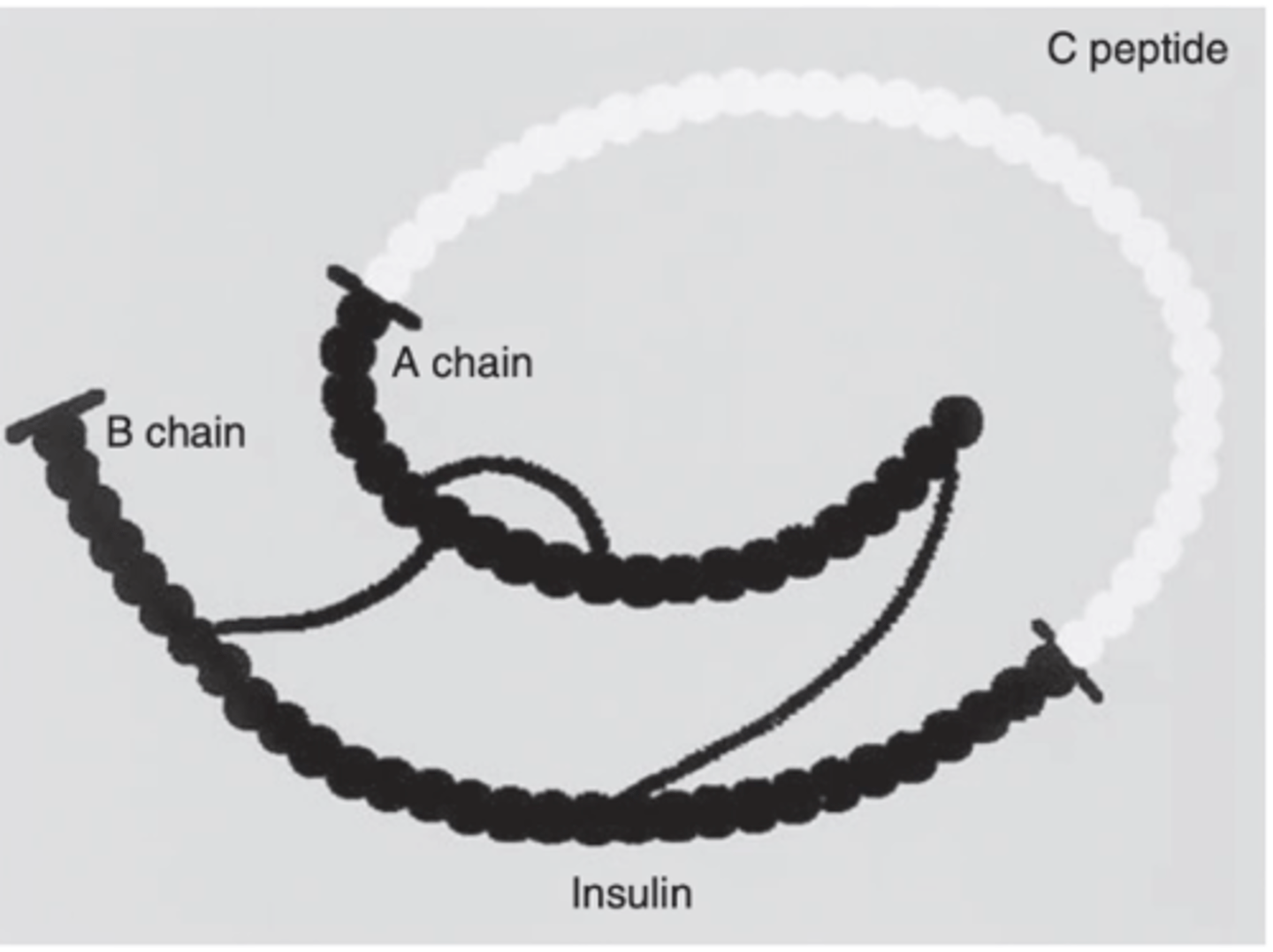
Why is the half life of insulin fairly short?
The A chain and B chain are held together by disulfide bonds, these bonds are unstable and easily hydrolyzed.
where is insulin made?
beta cells of pancreas
How is insulin secreted from pancreatic beta cells?
Glucose transported by GLUT-2 transporter increases ATP, which closes potassium channels and depolarizes the cell, leading to opening of calcium channels; the rise in calcium then triggers insulin release by exocytosis.
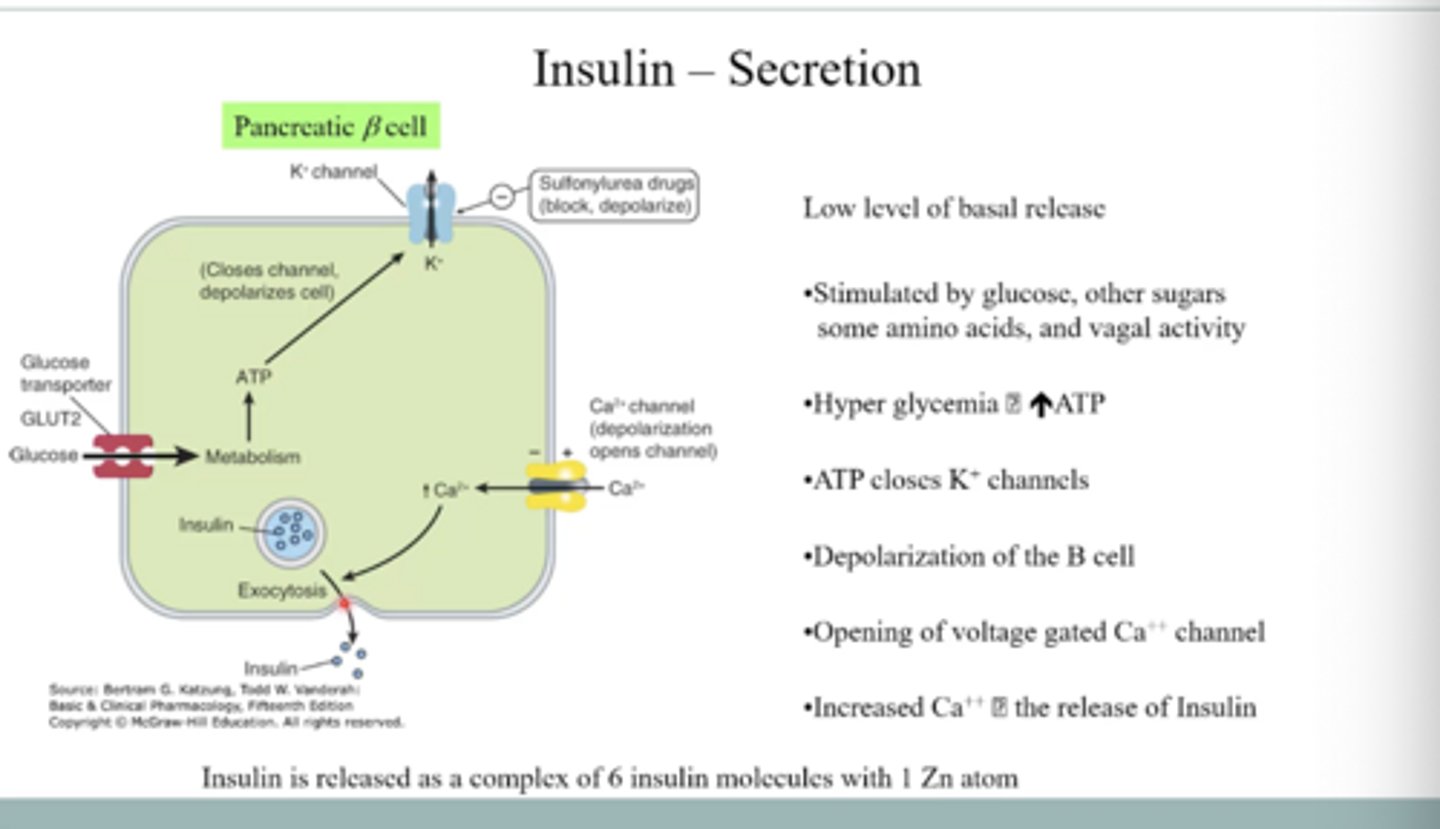
What is the structure of insulin when it is secreted? What must happen to the insulin complex in order for it to be effective?
When insulin is secreted, it forms a complex of six insulin molecules bound to one zinc atom. The insulin complex must dissociate from the zinc atom to be effective.
what three tissue types have the most insulin receptors
liver, muscle, adipose
the insulin receptor is intrinsic or extrinsic in activity
intrinsic
Describe the location of the GLUT-2 versus GLUT-4 transports.
GLUT-2: is always on the plasma membrane (in beta cells)
GLUT-4: is inside the the cells, when insulin is activated the transports will move to plasma membrane (in adipose, liver and muscle)
How do pancreatic cells respond when glucose levels are high?
When glucose levels are high, beta cells of the pancreas increase insulin release
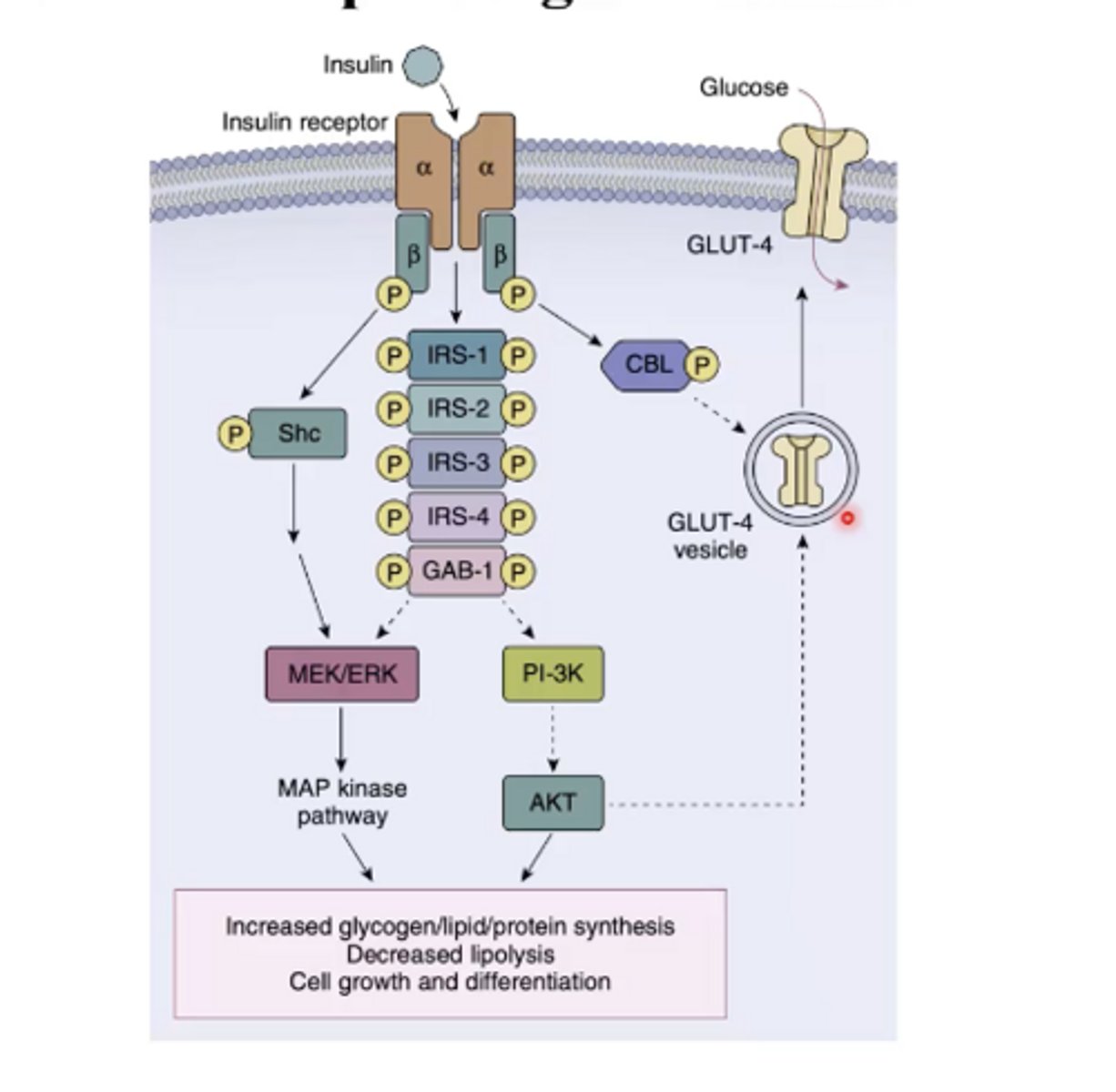
What are the main effects of insulin receptor signal transduction?
It increases glycogen, lipid, and protein synthesis, decreases lipolysis, and promotes cell growth and differentiation.
Why are blood glucose levels important in relation to insulin?
Blood glucose levels serve as a surrogate for insulin action.
How do changes in blood glucose levels affect insulin and energy use?
Increased glucose increases insulin, leading to uptake and storage of energy; decreased glucose decreases insulin, leading to use of stored energy and production of glucose.

What is the most important physiological role of insulin?
Increasing glycogen synthesis and promoting glucose uptake from the blood.

Is diabetes characterized by hyper or hypoglycemia?
What are the major classifications of diabetes?
hyperglycemia
The are for types of diabetes Type I- IV
type I diabetes is also known as?
Insulin dependent diabetes mellitus (the name tells you what the problem is)
describe type I diabetes and how it is treated
Patients do not make insulin because their beta cells have a disfunction. Patients have to administer their own insulin
Type II diabetes is also known as?
Non-Insulin Dependent Diabetes Mellitus
Describe Type II diabetes, what are 3 strategies to treatment Type II diabetes
pancreas is secreting enough insulin, but body is unable to use it to move glucose into cells. The strategies, are to secrete more insulin, restore sensitivity to insulin, reduce blood glucose from diet
describe Type III diabetes
diabetes linked to pancreatic disease, hormonal changes, drugs, or genetics
Type IV diabetes is also known as?
Gestational Diabetes/ diabetes during pregnancy
What are the benefits of good glycemic control?
What are the downsides of glycemic control?
Benefits: It minimizes blood glucose swings, lowers glycated hemoglobin, and reduces eye, kidney, and nerve complications.
Downsides: Risk of hypoglycemia and weight gain
what are the four different insulin acting types
short, regular, intermediate, ultralong
how is the short acting/rapid insulin modified differently from regular insulin?
Insulin molecules that do not complex with the zinc at all, they are free ready to dissociate. Once injected they are ready to bind to a receptor.
how is the slow acting insulin different from regular?
it is more tightly bound to insulin and slowly dissociates rather so the onset of action is slower and insulin stays in the body longer
What are the main insulin delivery systems?
Subcutaneous injections (syringes and pen injectors) and continuous subcutaneous insulin infusion devices (insulin pumps).
What are the main complications of insulin therapy?
Hypoglycemia, weight gain, immune reactions (such as insulin allergy or resistance), and lipodystrophy at the injection site.
are the various insulin preparations different in their pharmacodynamics or pharmacokinetics?
pharmacokinetics
What are three therapeutic goals of drugs used to treat diabetes?
To increase insulin release, increase insulin sensitivity, and decrease blood glucose levels.
what drug category might be used to increase insulin release (in tx type II diabetes)
insulin secretagogues
what are 2 specific drug types that are insulin secretagogues? which one is prefered?
sulfonylureas, meglitinides; meglitinides (faster onset action)
How do sulfonylureas increase insulin release?
They bind to receptors on pancreatic beta cells and close ATP-sensitive potassium channels, causing depolarization, and lead to calcium influx, and insulin release.
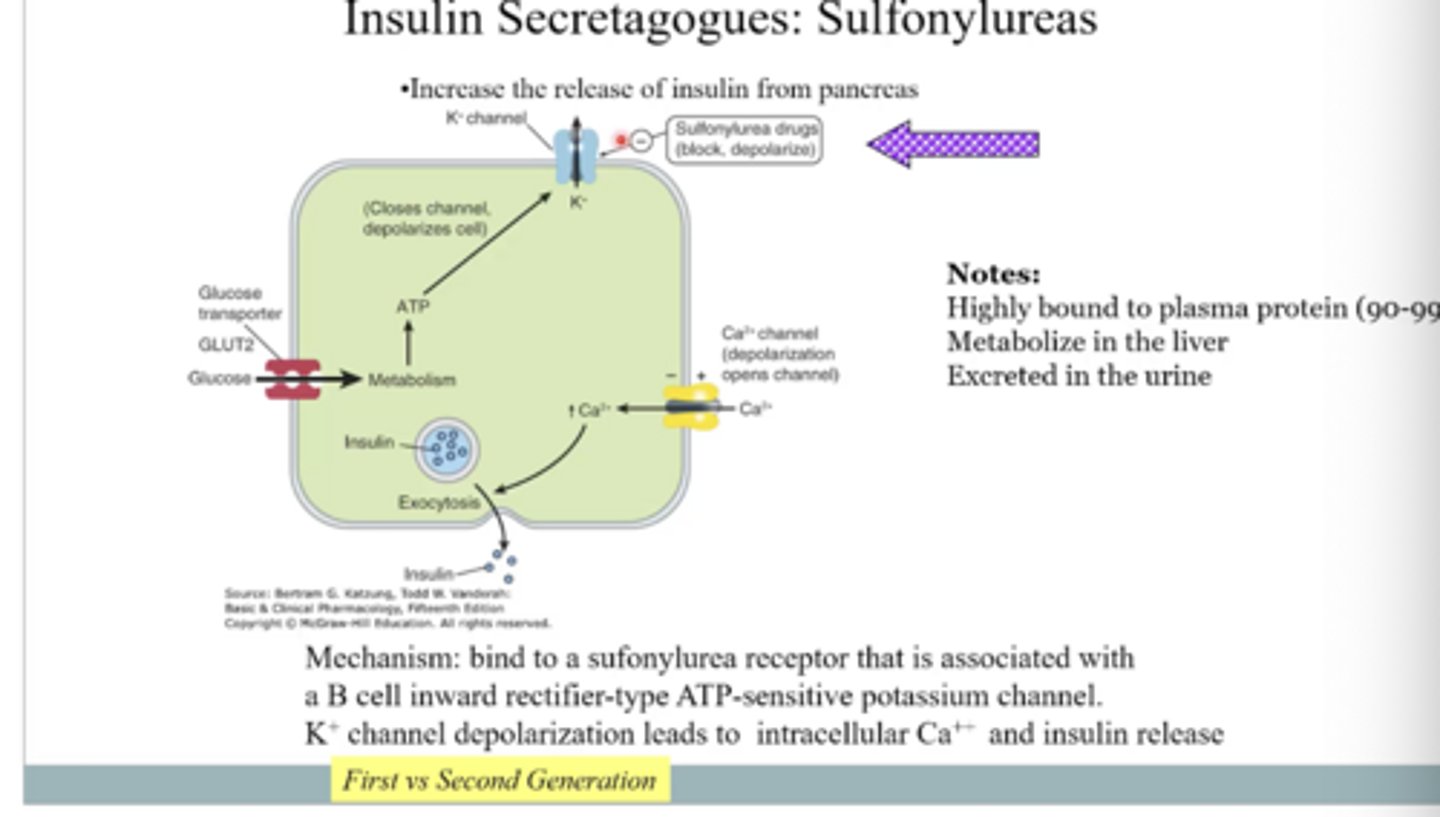
drug interactions with sulfonylureas
antifungals and oral anticoagulants
what is a drug example of a meglitinides?
What are the key features of meglitinides like repaglinide?
Repaglinide ; They regulate potassium channels to increase insulin release, share a binding site with sulfonylureas, and have a rapid onset of action with peak effect within about one hour.
How does metformin work as an insulin sensitizer?
It increases glucose uptake in tissues, activates AMPK, decreases hepatic gluconeogenesis, slows glucose absorption from the GI tract, and lowers glucagon levels.
what drug category might be used to increase insulin sensitivity (in tx type II diabetes)
insulin sensitizers
what are 2 specific drug types that are insulin sensitizers
biguanides, thoazolidinediones
what is a specific drug example of thoazolidinediones? Explain their MOA.
Pioglitazone ; They improve insulin sensitivity by activating PPAR-gamma, leading to changes in gene transcription involved in glucose and lipid metabolism, with both acute and chronic effects.
what drug category might be used to decrease blood glucose (in tx type II diabetes)
enzymatic inhibitors
what is specific drug type that is an enzymatic inhibitor (of glucose)
alpha-glucosidase inhibitors
How do alpha-glucosidase inhibitors like acarbose work and what is a common side effect?
They competitively inhibit intestinal alpha-glucosidase enzymes to slow carbohydrate digestion and glucose absorption after meals, and commonly cause gastrointestinal distress.
what is a drug example of alpha-glucosidase inhibitors
acarbose
What do GLP-1 agonists do to regulate blood glucose?
They stimulate insulin release and inhibit glucagon release to decrease blood glucose
what is a drug example of a GLP-1 agonist
exenatide
what enzyme inhibits/breaks down GLP-1?
DPP-4
what drug is a DPP-4 inhibitor
sitagliptin (allows for increased GLP-1 effects)
drugs that target the sodium-glucose co-transporter (SGLT2 inhibitors) prevent?
reuptake of glucose in the kidney
what is a drug that is a SGLT2 inhibitor
canagliflozin
hat does glucagon do to energy in the body?
Glucagon converts stored energy into usable energy by promoting glucose production.
Which cells of the pancreas secrete glucagon?
A (alpha) cells of the pancreas
What is pramlintide and how is it used?
Pramlintide is an amylin analog given by injection at mealtime.
How do SGLT2 inhibitors like canagliflozin lower blood glucose?
They block sodium-glucose co-transporters in the kidney, preventing glucose reabsorption and increasing glucose excretion in the urine, which lowers blood glucose levels.
glucagon signals through what receptor types
GPCR
What is the half-life of glucagon?
3-6 minutes
clinical uses of glucagon
tx of hypoglycemia, diagnosis of endocrine disorders, Beta-blocker poisoning, radiology of the bowel