O4: Mechanistic Organic Chemistry
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
what does more negative free gibbs E mean and what causes it?
more negative free Gibbs mean the reaction is more favoured to the products, this could when:
the enthalpy (delta H) is negative (due to the product’s bonds are stronger than the reactant’s, or release of strain)
Enthalpy is high when forming bonds and low when breaking them
Or positive entropy (delta S) (more freedom ) this is observed in the reaction formula eg. 1A + 2B → 3C + 2D. 3 → 5, products are prefered in here
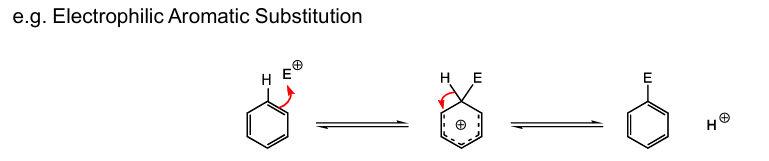
What is the difference between a protic and an aprotic solvent , give eg. of each from more polar to least and how does polarity impact the solvents and its relation to the dielectric constant
Protic solvents: contain O–H or N–H bonds and solvate positive charges and negative charges,
e.g. solvents such as H2O or CH3OH (methanol), Ethanol (EtOH)
Aprotic solvents do not contain O–H or N–H bonds and will only solvate positive charges,
e.g. solvents such as DMSO [(CH3 )2SO], acetonitrile (NCMe) and acetone [(CH3)2CO], DCM
The more polar the protic or aprotic solvents the higher the dielectric constant( ability to solvate charges)the better they are at solvating
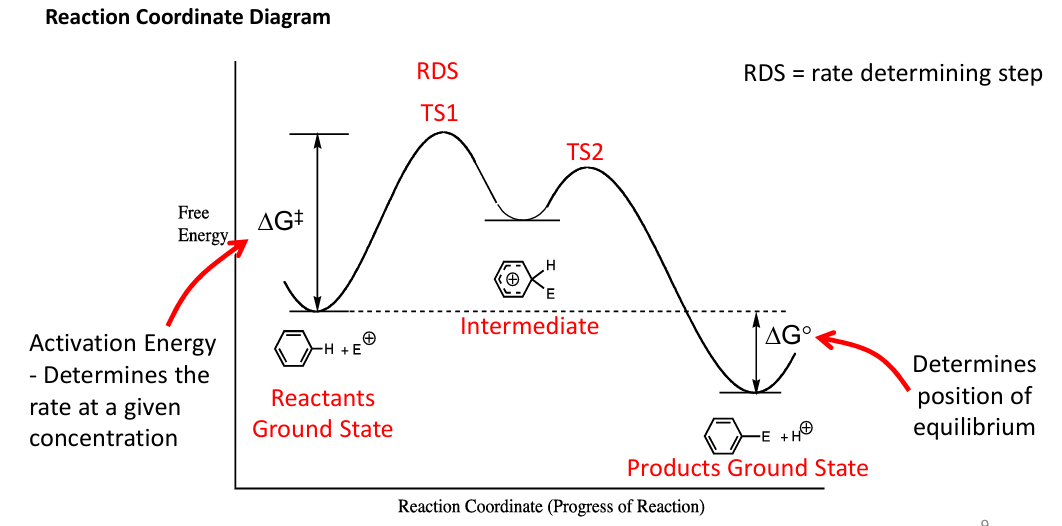
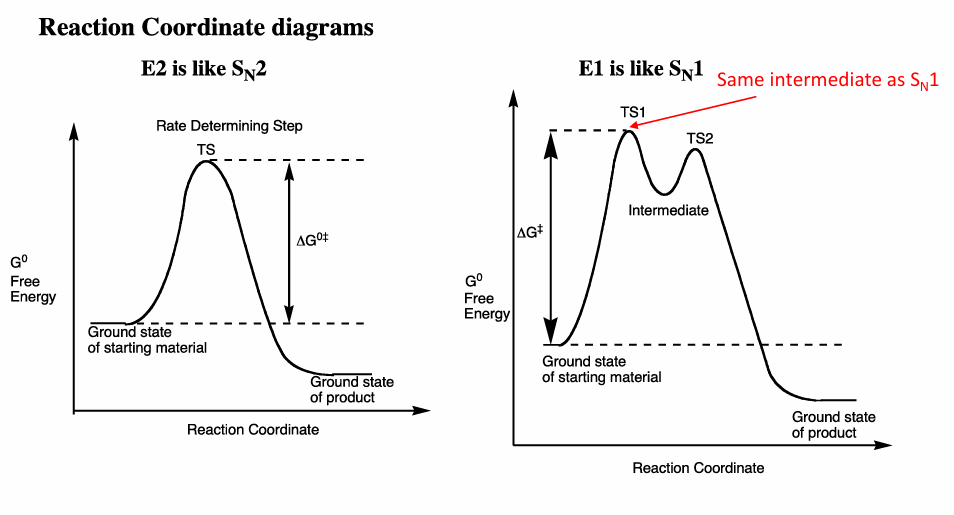
Draw a reaction coordinate diagram of this reaction and label it, and draw the intermediate
pKa relation to gd LG and gd Nu-
the more positive the pKa the more basic, the more basic the more Nu- a molecule is
The more negative the pKa the more acidic, the more acidic the more E+ something is
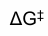
what is this, how to make it smaller and its effect on rxn
state arrhenius equation
Also what is the difference btwn Sn1 and Sn2 do an example and which one has an intermediate? draw a reaction coordinate diagram
this is the molar free energy of activation i.e activation energy
, the difference between reactants and TS, more negative Ea means more favoured rxn to lower it u got to bring reactants up or lower TS or both.
Sn2 does not have an intermediate it’s bimolecular only has TS, Sn1 has an intermediate
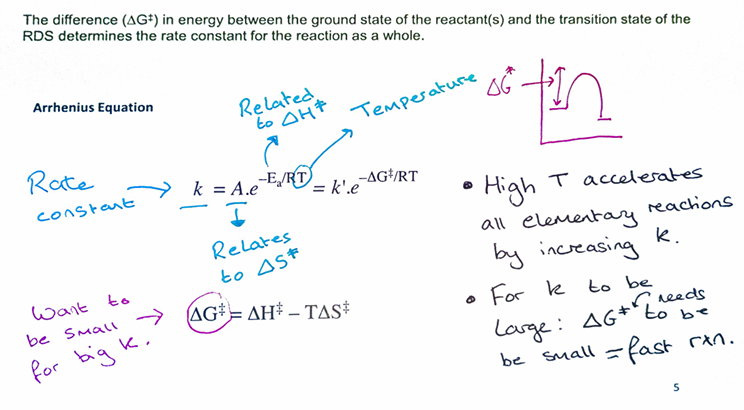
What Sn2 rxn will proceed faster , give 3 things and why? state all that u know about Sn2
Sn2 will prefer more acidic LG, the more you go down a group the more E+ the LG the lower the pka, so from fastest I>Br>Cl to slowest
prefers less bulky so prefers to react with primary > 2>>3
does not really do sp2 C=C either
will prefer to react with NaOMe rather than EtOH coz NaOMe is a harder Nu-
Sn2 will always result in a change of stereochemistry (new Group will be in dashes point to the back or opposite to LG in general)
How does a Polar aprotic solvents help with an Sn2 reaction, give an eg. of it and draw and explain how it would for eg. solvate KNu
Polar aprotic solvents work by dissolving the cation solely which makes the anion highly Nu- ready to donate to the E+ and making aprotic solvents very gd for Sn2 reactions eg. of that is is DMSO
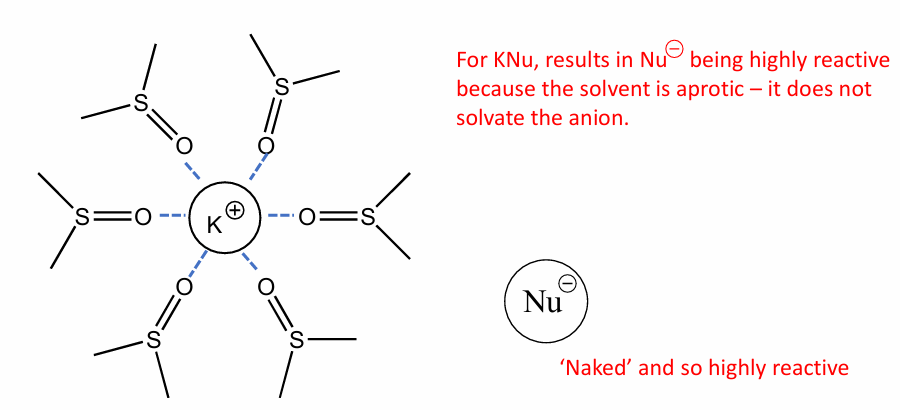
Give an eg of a non- polar aprotic solvent and how it would solvate KNu
which is faster for Sn2 protic or aprotic and why? how do protics dissolve then? which are faster polar or non polar aprotics and why?
THF solvent
Protics dissolve both cations and anions and dissolves harder Nu- (most basic) the best , eg. OH> Cl >Br >I. Makes Sn2 slower
Polar Aprotics are faster because charge seperation is stabilised by polar aprotics
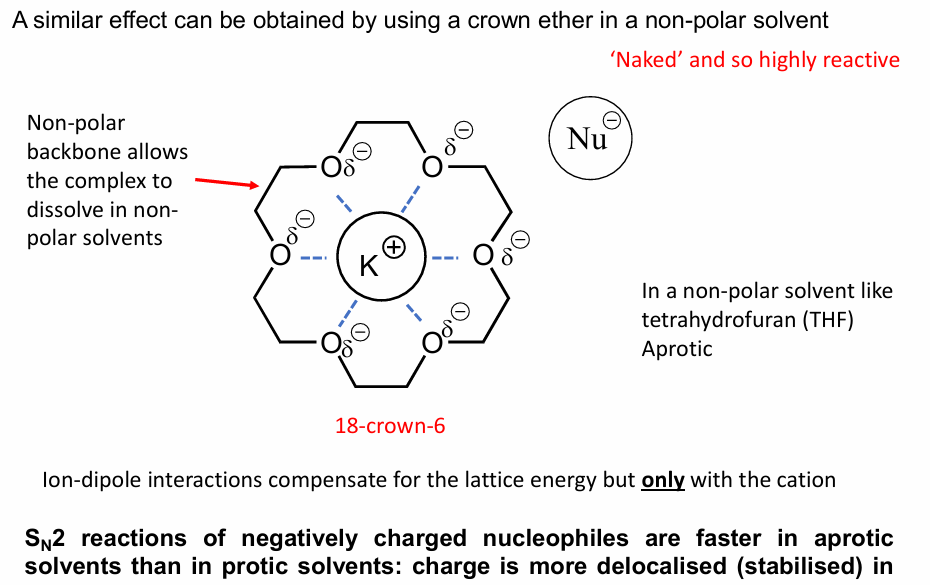
we know that polar dissolves in polar and non-polar dissolves in non-polar in we want them to go an Sn2 rxn, how would we allow this to happen? draw the structure and name it and how it will work
We need to use a phase transfer Catalyst, eg. Bu4N+
Bu4N+ will drag the aqueous anion into the organic layer striping it, making it highly negatively charged for Sn2 rxn
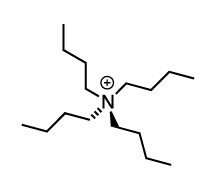
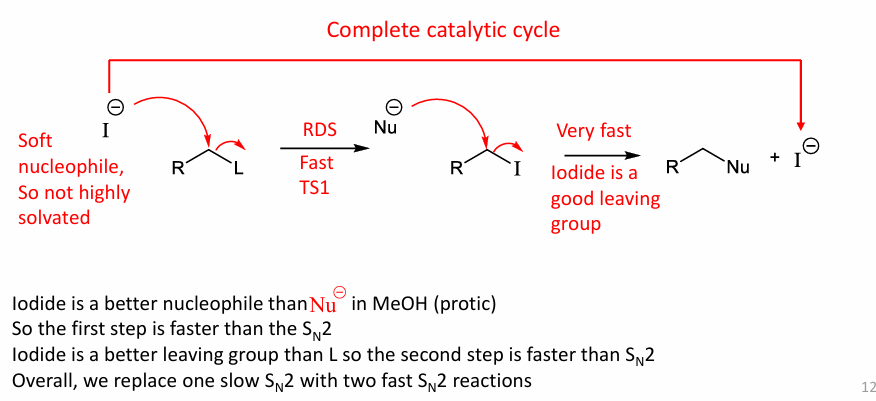
Talk about protic Sn2 rxn?
eg. of protic solvent, that are used, prefered Nu- and faster rxn
draw mechanism of protic Sn2 RCH2L rxn
Protic solvents such as MeOH prefer reacting with a soft Nu- (more acidic down a group), coz they solvate both charges.
it can go a single Sn2 but it is slow so it goes 2 fast Sn2, intead 1 slow one.
coz iodine will be a better LG than L
Protic prefer weak Nu-
Aprotic prefers strong Nu- (eg.MeOH, CN-)
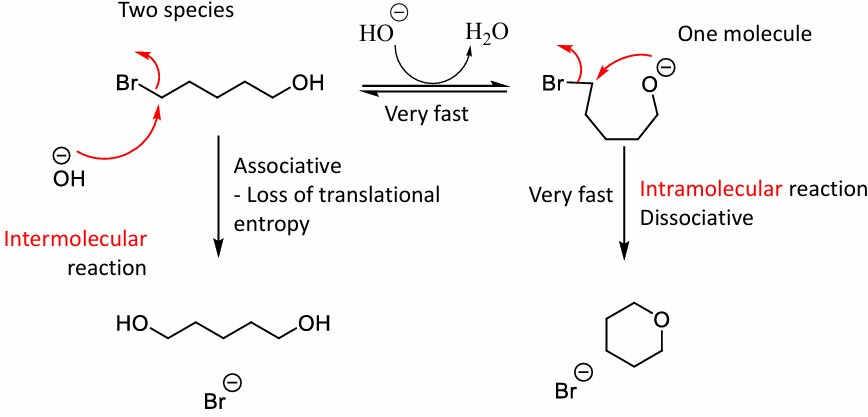
What rxn are favoured inter or intra, and the order
Intramolecular cyclysations are favoured they are faster
Most favoured. 5>6>3>4>7. least
Name the rxn it will endergo, with explaining the terms used
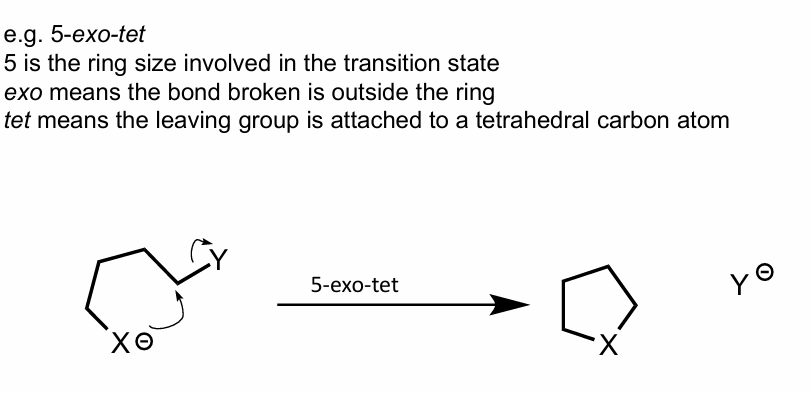
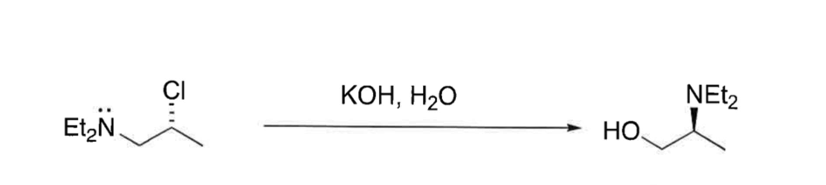
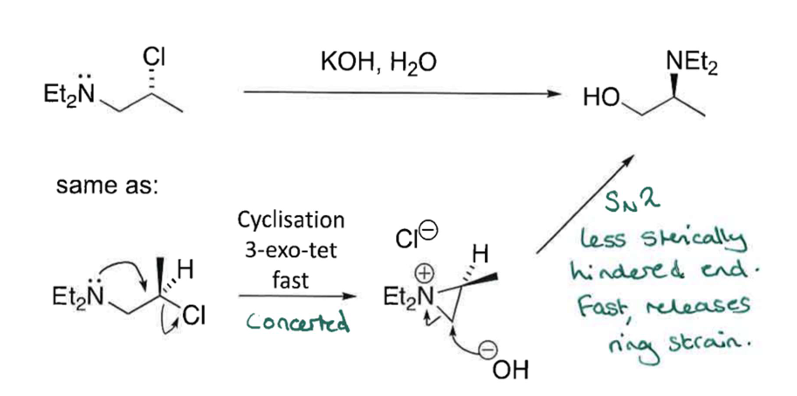
Show mechanism
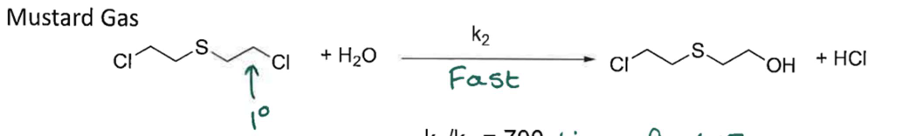
Show Mech
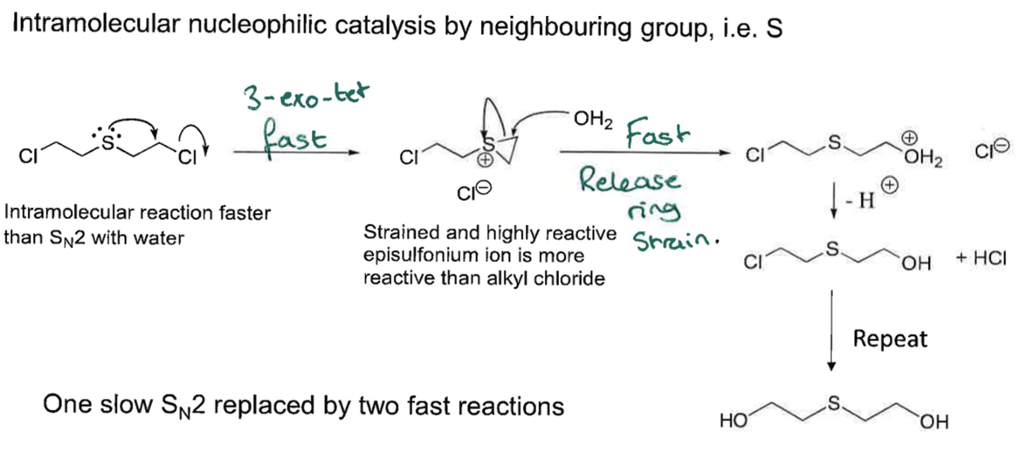
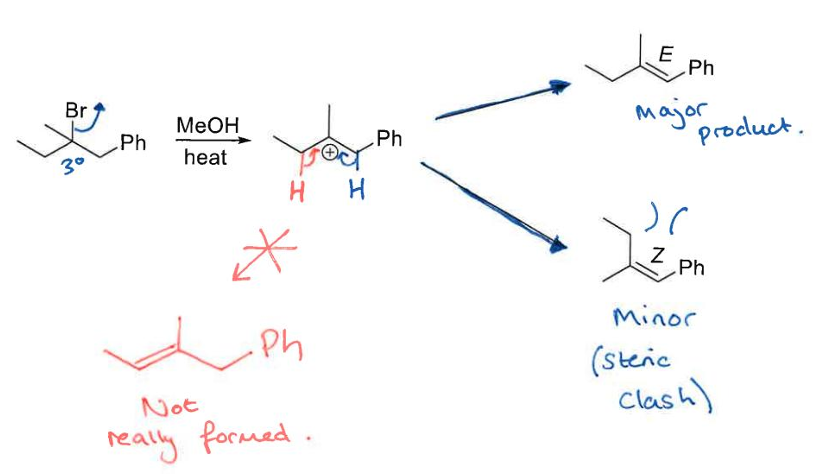
Talk about Sn1:
what is it ? its RDS? what increases the RDS? what fastens the reaction ? mechanism? and what solvents does it prefer?
Unimolecular has 2 TS and an intermediate, 1st step (LG leaves) is the RDS, the better the LG (more acidic) and the higher the rate of the rxn.
Reacts better with soft Nu- (more acidic), and prefers Protic polar solvents eg. MeOH
coz when LG leaves, the molecule becomes planar and Nu- can attack from any side giving racemic mix
also the better release of strain the more favourable the rxn
reacting with tertirary alkyl halides
Primary alkayl halides do not undergo Sn1
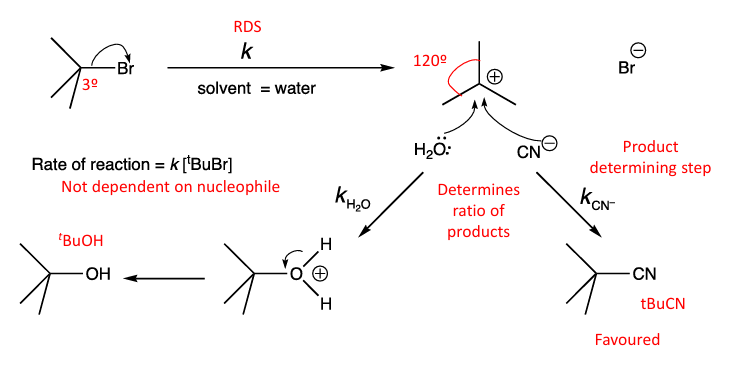
what are the exceptions and why would they go Sn1 when not 3 alkyl halides
If the Nu- is not strong (basic) enough they will go Sn1
Benzylics will not go Sn1 if the group attached are EWG, eg. NO2, halogens
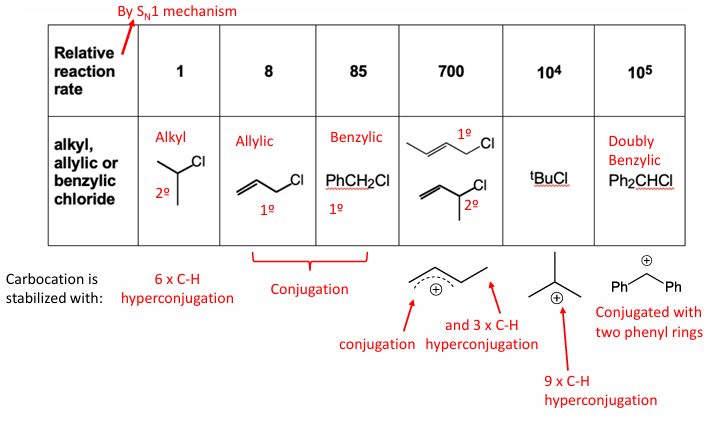
We know that E1 competes with Sn1
How can we favour Sn2,
and how can we favour Sn1 if Sn2 is not possible
how to minimise Elemination
how to maximise elemination
E1 competes with Sn1 rxn

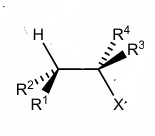
Show E2 of this
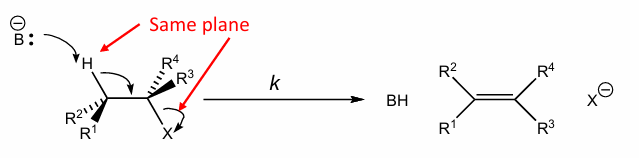
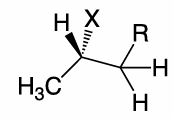
use Newman projection to draw the E2 of this molecule , and what product conformation does it favour
It favours E(trans) alkenes products
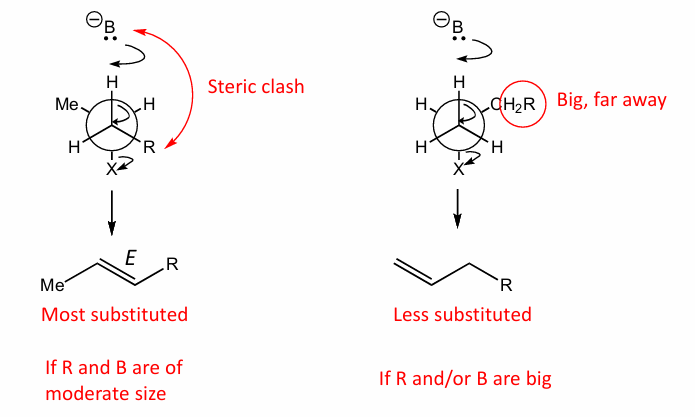
How to favour E2 over E1

How to favour E2 over Sn2

Draw coordinate diagrams for Sn1, E1, Sn2 and E2
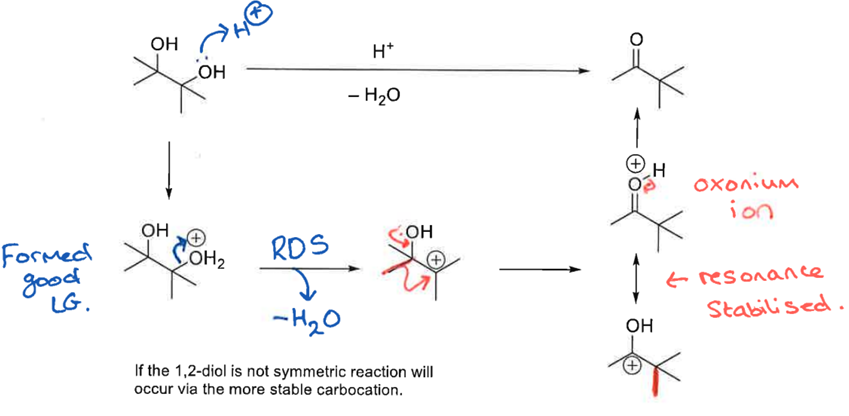
Do this pinacol rearegement rxn, hint: add H+
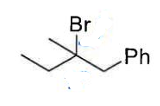
Do an E1 rxn and state what alkyl halides and configuration are favoured
favours E ,avoids steric hendrance,and prefers tertiary alkyl halides the most, and weaker Nu-. Favourable especially for steric release as any rxn