10 - group 2 elements
1/62
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
63 Terms
general equation for reaction between metal and water
M (s) + 2H2O (l) → M(OH)2 (s) + H2 (g)
The exceptions to this general equation are:
Be which does not react with water
Mg which forms MgO (s) and H2 (g)
which group 2 elements react with water
all except for beryllium
reactions of group 2 elements with water : magnesium
Magnesium reacts very slowly with cold water. Little hydrogen gas and magnesium hydroxide are formed over a few days.
Mg (s) + 2H2O (l) → Mg(OH)2 (s) + H2(g)
Magnesium reacts readily with steam in a vigorous reaction. When steam is passed over heated magnesium hydrogen gas and magnesium oxide are formed. Magnesium metal glows with a white light as the reaction takes place
Mg (s) + H2O (g) → MgO (s) + H2(g)
reactions of group 2 elements with water : calcium
The rest of the Group 2 metals, calcium to barium react with cold water with increasing vigor. Calcium reacts moderately with water and forms cloudy white precipitate due to the formation of sparingly soluble calcium hydroxide
Ca (s) + 2H2O (l) → Ca(OH)2( s) + H2(g)
reactions of group 2 elements with water : strontium
reacts rapidly with water and its hydroxide is formed.
Sr (s) + 2H2O (l) → Sr(OH)2 (aq) + H2 (g)
reactions of group 2 elements with water : barium
reacts vigorously with water to form its hydroxide.
Ba (s) + 2H2O (l) → Ba(OH)2 (aq) + H2(g)
general equation for reaction between metals and oxygen
The reaction of all metals with oxygen follows the following general equation:
2M (s) + O2 (g) → 2MO (s)
Where M is any metal in Group 2
Remember that Sr and Ba also form MO2
The reactions of these metals with oxygen, once started, are vigorous.
reaction of group 2 elements with oxygen : magnesium
Magnesium is covered with a superficial oxide layer due to oxidation by atmospheric oxygen. It rapidly/easily burns in air / oxygen with a brilliant/bright white flame.
2Mg (s) + O2 (g) → 2MgO (s)
reaction of group 2 elements with oxygen : calcium
Calcium is difficult to ignite, but burns with a brick red flame
2Ca (s) + O2 (g) → 2CaO (s)
reaction of group 2 elements with oxygen : strontium
difficult to ignite, burns with a crimson red flame
2Sr (s) + O2 (g) → 2SrO (s)
Sr (s) + O2 (g) → SrO2 (s)
reaction of group 2 elements with oxygen : barium
difficult to ignite, burns with an apple green flame to form its oxide
2Ba (s) + O2 (g) → 2BaO (s)
Ba (s) + O2 (g) → BaO2 (s)
general equation for reaction between metals and dilute HCl
M (s) + 2HCl (aq) → MCl2 (aq) + H2 (g)
The metals react with dilute hydrochloric acid, forming a colorless solution of the metal chloride and bubbles of hydrogen gas. Reaction is more vigorous down the group.
The metal chlorides formed (e.g., MgCl₂, CaCl₂, SrCl₂, BaCl₂) are generally soluble in water.
reaction of group 2 elements with dilute hcl : magnesium
reacts vigorously
Mg (s) + 2HCl (aq) → MgCl2 (aq) + H2 (g)
reaction of group 2 elements with dilute HCl : calcium
reacts vigorously
Ca (s) + 2HCl (aq) → CaCl2 (aq) + H2 (g)
reaction of group 2 elements with dilute HCl : strontium
reacts vigorously
Sr (s) + 2HCl (aq) → SrCl2 (aq) + H2 (g)
reaction of group 2 elements with dilute HCl - barium
reacts vigorously
Ba (s) + 2HCl (aq) → BaCl2 (aq) + H2 (g)
general equation for reaction between all metals and dilute sulfuric acid
M (s) + H2SO4 (aq) → MSO4 (aq) + H2 (g)
Remember that CaSO4, SrSO4 and BaSO4 are insoluble
reaction between group 2 elements and dilute H2SO4 : magnesium
reacts vigorously
Mg (s) + H2SO4 (aq) → MgSO4 (aq) + H2 (g)
reaction between group 2 elements and dilute H2SO4 : calcium
Reaction is slowed by the formation of a sparingly soluble (almost insoluble) sulfate layer which coats the metal surface stopping hydrogen bubbles from rising
Ca (s) + H2SO4 (aq) → CaSO4 (s) + H2 (g)
reaction between group 2 elements and dilute H2SO4 : strontium
Reaction is quickly stopped by the formation of an insoluble sulfate layer on the metal surface
Sr (s) + H2SO4 (aq) → SrSO4 (s) + H2 (g)
reaction between group 2 elements and dilute H2SO4 : barium
Reaction is quickly stopped by the formation of an insoluble sulfate layer on the metal surface
Ba (s) + H2SO4 (aq) → BaSO4 (s) + H2 (g)
group 2 oxides
All Group 2 oxides are basic and ionic, except for BeO which is amphoteric (it can act both as an acid and base)
group 2 oxides with water
Group 2 oxides react with water to form alkaline solutions which generally get more alkaline going down the group
This happens because the hydroxides that form become more soluble as you move down the group
This means that more hydroxide ions, OH–, dissociate into the solution (excess water) causing the pH to increase
The alkalinity,
and hence the pH of the solution, depends on the relative solubility of the hydroxide in water.
reaction of group 2 oxides with water : magnesium
MgO (s) + H2O (l) → Mg(OH)2(s)
Mg(OH)2 is only slightly soluble in water, therefore a weakly alkaline solution (pH 9-11) is formed
Mg(OH)2( s) + aq ⇌ Mg(OH)2
or : Mg(OH)2 (s) ⇌ Mg2+ + 2OH−(aq)
1st shows that it barely dissolves and is simplified, 2nd is more common one (i think)
reaction of group 2 oxides with water : calcium
CaO (s) + H2O (l) → Ca(OH)2(s)
A vigorous reaction which releases a lot of energy, causing some of the water to boil off as the solid lump seems to expand and open (pH 11.0)
calcium hydroxide is also called limewater
reaction of group 2 oxides with water : strontium
SrO (s) + H2O (l) → Sr(OH)2(aq)
reaction of group 2 oxides with water : barium
BaO (s) + H2O (l) → Ba(OH)2(aq)
Hydroxide is soluble in water and the pH of the medium is between 11 – 13 .
general equation for an oxide and water
oxide + water → hydroxide
group 2 metals with acid
The Group 2 metals will react with dilute acids to form colourless solutions of metal salts
For example, they will form colourless solutions of metal chlorides if reacted with hydrochloric acid
When metals react with an acid, the by-product of this reaction is hydrogen gas
the reactions are more vigorous down the group
When some Group 2 metals react with sulfuric acid rather than hydrochloric, an insoluble sulfate forms
Going down the group, the Group 2 sulfates become less and less soluble
Calcium sulfate is sparingly soluble, but strontium sulfate and barium sulfate are insoluble
group 2 oxides with acid (h2so4)
Group 2 sulfates also form when a Group 2 oxide is reacted with an acid
The insoluble sulfates form at the surface of the oxide, which means that the solid oxide beneath it can’t react with the acid (on calcium - partially -, strontium and barium)
This can be prevented to an extent by using the oxide in powder form and stirring, in which case neutralisation can take place
MgO( s) + 2 HCl (l) → MgCl2 ( aq ) + H2O ( l )
CaO ( s ) + H2SO4 ( aq) → CaSO4 ( s ) + H2O ( l )
general equations for metal oxides with dilute HCl, and dilute sulfuric acid
metal oxide + dilute hydrochloric acid → salt + water
metal oxide + dilute sulfuric acid → sulfate + water
reactions of group 2 elements with dilute HCl and dilute H2SO4 : magnesium
Mg (s) + 2HCl (aq) → MgCl2 (aq) + H2 (g)
Mg (s) + H2SO4 (aq) → MgSO4 (aq) + H2 (g)
reactions of group 2 elements with dilute HCl and dilute H2SO4 : calcium
Ca (s) + 2HCl (aq) → CaCl2 (aq) + H2 (g)
Ca (s) + H2SO4 (aq) → CaSO4 (aq) + H2 (g)
reactions of group 2 elements with dilute HCl and dilute H2SO4 : strontium
Sr (s) + 2HCl (aq) → SrCl2 (aq) + H2 (g)
Sr (s) + H2SO4 (aq) → SrSO4 (s) + H2 (g)
reactions of group 2 elements with dilute HCl and dilute H2SO4 : barium
Ba (s) + 2HCl (aq) → BaCl2 (aq) + H2 (g)
Ba (s) + H2SO4 (aq) → BaSO4 (s) + H2 (g)
group 2 hydroxides with dilute acid
The Group 2 metal hydroxides form colourless solutions of metal chlorides when they react with a dilute acid
The sulfates decrease in solubility going down the group (barium sulfate is an insoluble white precipitate)
reactions of group 2 hydroxides with dilute HCl and dilute H2SO4 : magnesium
Mg(OH)2 (s) + 2HCl (aq) → MgCl2 (aq) + 2H2O (l)
Mg(OH)2 (s) + H2SO4(aq) → MgSO4 (aq) + 2H2O (l)
reactions of group 2 hydroxides with dilute HCl and dilute H2SO4 : calcium
Ca(OH)2 (s) + 2HCl (aq) → CaCl2 (aq) + 2H2O (l)
Ca(OH)2 (s) + H2SO4(aq) → CaSO4 (aq) + 2H2O (l)
reactions of group 2 hydroxides with dilute HCl and dilute H2SO4 : strontium
Sr(OH)2 (s) + 2HCl (aq) → SrCl2 (aq) + 2H2O (l)
Sr(OH)2 (s) + H2SO4(aq) → SrSO4 (s) + 2H2O (l)
reactions of group 2 hydroxides with dilute HCl and dilute H2SO4 : barium
Ba(OH)2 (s) + 2HCl (aq) → BaCl2 (aq) + 2H2O (l)
Ba(OH)2 (s) + H2SO4(aq) → BaSO4 (s) + 2H2O (l)
general equations for reactions between metal hydroxides and dilute acid/dilute sulfuric acid
metal hydroxide + dilute acid → salt + water
metal hydroxide + dilute sulfuric acid → sulfate + water
reaction of group 2 carbonates (solubility, hcl, h2so4)
All Group 2 carbonates (except for BeCO3) are insoluble in water
All Group 2 carbonates will form soluble chloride salts, water and carbon dioxide gas when reacted with dilute hydrochloric acid
When reacted with sulfuric acid, the carbonates of Ca, Sr and Ba form an insoluble sulfate layer on their surface which stops any further reaction after the initial bubbling (effervescence) of carbon dioxide gas is seen
general reaction between metal carbonates and dilute HCl/dilute sulfuric acid
metal carbonate + dilute hydrochloric acid → salt + water + carbon dioxide
meatl carbonate + dilute sulfuric acid → sulfate + water + carbon dioxide
reactions of group 2 carbonates with dilute HCl and H2SO4 : magnesium
Magnesium carbonate shows rapid effervescence with sulfuric acid, but other carbonates stop the effervescence after the initial bubbling due to the formation of insoluble sulfate.
MgCO3 (s) + 2HCl (aq) → MgCl2 (aq) + H2O (l) + CO2 (g)
MgCO3 (s) + H2SO4(aq) → MgSO4 (aq) + H2O (l) + CO2 (g)
reactions of group 2 carbonates with dilute HCl and H2SO4 : calcium
CaCO3 (s) + 2HCl (aq) → CaCl2 (aq) + H2O (l) + CO2 (g)
CaCO3 (s) + H2SO4(aq) → CaSO4 (aq) + H2O (l) + CO2 (g)
reactions of group 2 carbonates with dilute HCl and H2SO4 : strontium
SrCO3 (s) + 2HCl (aq) → SrCl2 (aq) + H2O (l) + CO2 (g)
SrCO3 (s) + H2SO4 (aq) → SrSO4 (s) + H2O (l) + CO2 (g)
reactions of group 2 carbonates with dilute HCl and H2SO4 : barium
BaCO3 (s) + 2HCl (aq) → BaCl2 (aq) + H2O (l) + CO2 (g)
BaCO3 (s) + H2SO4(aq) → BaSO4 (s) + H2O (l) + CO2 (g)
solubility of group 2 hydroxides and sulfates
solubility of group 2 hydroxides increases down the group and group 2 sulfates decreases down the group.
thermal decomposition
the breakdown of a compound into two or more different substances using heat
thermal decomposition of group 2 carbonates
The Group 2 carbonates break down (decompose) when they are heated to form the metal oxide and give off carbon dioxide gas
The general equation for the decomposition of Group 2 carbonates is:
XCO3 (s) →(HEAT) XO (s) + CO2 (g)
X = Group 2 element
Going down the group, more heat is needed to break down the carbonates
MgCO3 (s) →(HEAT) MgO (s) + CO2 (g)
thermal decomposition of group 2 nitrates
Group 2 nitrates also undergo thermal decomposition
Group 2 nitrates decompose to form the metal oxide, nitrogen dioxide gas and oxygen gas
The general equation for the decomposition of Group 2 nitrates is:
X(NO3)2 (s) →{HEAT} XO (s) + 2NO2 (g) + ½O2 (g)
OR
2X(NO3)2 (s) →{HEAT} 2XO (s) + 4NO2 (g) + O2 (g)
X = Group 2 element
Nitrogen dioxide gas is observed as brown fumes and is toxic
This is often seen as brown fumes when heating Group 2 nitrates in test tube reactions
An example of this reaction is:
2Ca(NO3)2 (s) →{HEAT} 2CaO (s) + 4NO2 (g) + O2 (g)
trend in thermal stabilities of group 2 carbonates and nitrates
1. The basic trend
Group 2 carbonates (MCO₃) and nitrates (M(NO₃)₂) decompose when heated.
As you go down Group 2 (Be → Ba), you need more heat to make them decompose.
➡ So thermal stability increases down the group.
2. Why does this happen?
It’s all about polarisation of the large anions (CO₃²⁻ or NO₃⁻).
The Group 2 cation (Mg²⁺, Ca²⁺, Sr²⁺, Ba²⁺) is positively charged.
The anion (carbonate or nitrate) has lots of delocalised negative charge.
The positive ion can distort (polarise) the negative ion’s electron cloud.
3. Polarising power of the cation
Small, highly charged ions (like Mg²⁺ or Be²⁺) have a strong electric field.
They pull on the electron clouds of the big anions (CO₃²⁻, NO₃⁻).
This weakens the bonds inside the anion.
If the bonds are weakened, the anion breaks apart more easily when heated → less thermally stable.
So:
At the top (small cations like Mg²⁺): strong polarisation → bonds in carbonate/nitrate weakened → decomposes easily → low thermal stability.
At the bottom (large cations like Ba²⁺): weak polarisation → bonds in carbonate/nitrate remain strong → decomposes less easily → high thermal stability.
chemical trends in group 2
All elements in Group 2 (also called alkali earth metals)have the two electrons in their outermost principal quantum shell
All Group 2 metals can form ionic compounds in which they donate these two outermost electrons (so they act as reducing agents) to become an ion with +2 charge (so they themselves become oxidised)
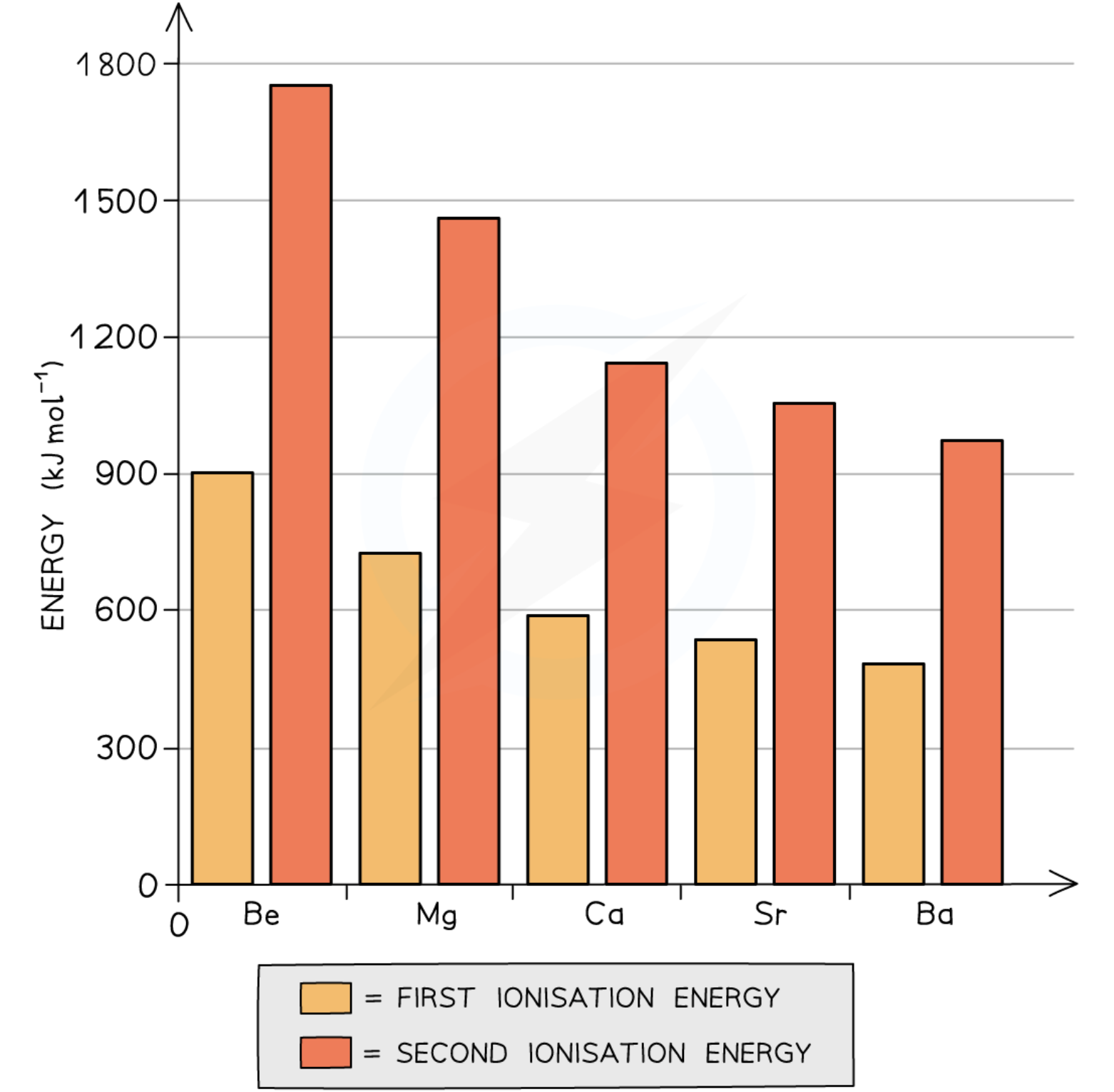
reactivity and ionisation energies of group 2 elements
Going down the group, the metals become more reactive
This can be explained by looking at the Group 2 ionisation energies (graph)
The first ionisation energy is the energy needed to remove the first outer electron of an atom
The second ionisation energy is the energy needed to remove the second outer electron of an atom
The graph above shows that going down the group, it becomes easier to remove the outer two electrons of the metals
Though the nuclear charge on the nucleus increases going down the group (because there are more protons);
Factors such as an increased shielding effect and a larger distance between the outermost electrons and the nucleus outweigh the attraction of the higher nuclear charge
The elements become more reactive going down the group as it gets easier for the atoms to lose two electrons and become 2+ ions
This trend is shown by looking at the reactions of the Group 2 metals:
With dilute hydrochloric acid;
Bubbles of hydrogen gas are given off much faster indicating that the reactions become more vigorous
With oxygen;
The metals get more reactive with oxygen down the group (Ba is so reactive, that it must be stored in oil to prevent it from reacting with oxygen in air)
physical trends in group 2
Going down the group, the elements become larger as the outer two electrons occupy a new principal quantum shell which is further away from the nucleus

melting points of group 2 elements
The melting point of the elements decreases going down the group as the outer electrons get further away from the nucleus
This means that the attraction between the nucleus and the bonding electrons decreases causing a decrease in melting point

density of group 2 elements
As you go down the group, the density of the alkali earth metals increases

general properties of group 2 elements
They are all metals and are good conductors of heat and electricity.
Their compounds are all white or colourless.
In all their compounds they exhibit the oxidation state of +2.
Their compounds are ionic (except for beryllium oxide which is mostly covalent)
They are called alkaline earth metals because their oxides and hydroxides are basic.
They react with acids to give their salts and hydrogen gas ; reactivity increases down the
group
They react with cold water to give their hydroxides and hydrogen gas ; magnesium reacts
with steam to give its oxide and hydrogen gas ; reactivity increases down the group.
Solubility of Group 2 hydroxides increases down the group.
Solubility of Group 2 sulfates decreases down the group.
In general, electronegativity decreases down the Group. As successive energy levels
are filled, the positive nucleus exerts less force on electrons and so has less ability to attract electrons.
group 2 hydroxides (formations down group)
Going down the group, the solutions formed from the reaction of Group 2 oxides with water become more alkaline
When the oxides are dissolved in water, the following ionic reaction takes place:
O2- (aq) + H2O(l) → 2OH- (aq)
The higher the concentration of OH- ions formed, the more alkaline the solution
The alkalinity of the formed solution can therefore be explained by the solubility of the Group 2 hydroxides
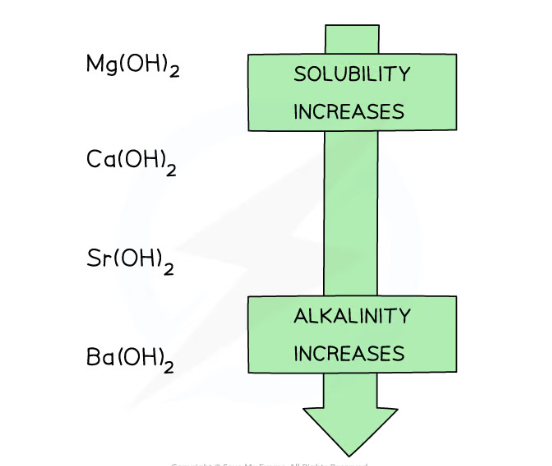
solubility of group 2 hydroxides table
Group 2 hydroxide | Solubility at 298 K (mol / 100 g of water) |
|---|---|
Mg(OH)2 | 2.0 x 10–5 (sparingly soluble) |
Ca(OH)2 | 1.5 x 10–3 |
Sr(OH)2 | 3.4 x 10–3 |
Ba(OH)2 | 1.5 x 10–2 (soluble) |
group 2 hydroxides dissolving in water
The hydroxides dissolve in water as follows:
X(OH)2 (aq) → X2+ (aq) + 2OH– (aq)
Where X is the Group 2 element
When the metal oxides react with water, a Group 2 hydroxide is formed:
XO (s) + H2O (l) → X(OH)2 (aq)
Going down the group, the solubility of these hydroxides increases
This increasing solubility is attributed to a decrease in lattice energy and a relative increase in hydration energy as the metal cation size increases.
This means that the concentration of OH- ions increases,increasing the pH of the solution
As a result, going down the group, the alkalinity of the solution formed increases when Group 2 oxides react with water
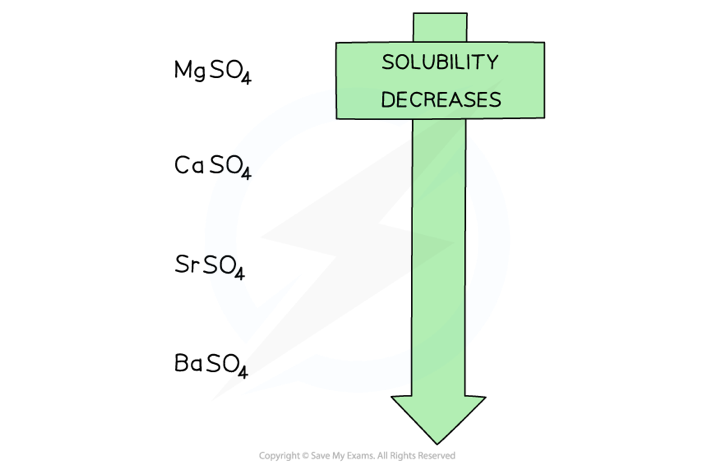
solubility table of group 2 sulfates
Group 2 sulfate | Solubility at 298 K (mol / 100 g of water) |
|---|---|
MgSO4 | Highly soluble |
CaSO4 | 1.2 x 10–3 |
SrSO4 | 7.6 x 10–4 |
BaSO4 | 1.0 x 10–4 (insoluble) |
group 2 sulfates dissolving in water
The sulfates dissolve in water as follows:
XSO4 (s) → X2+ (aq) + SO42- (aq)
Where X is the Group 2 element
When the metal oxides react with sulfuric acid, a Group 2 sulfate is formed:
XO (s) + H2SO4 (aq) → XSO4 (aq) + H2O (l)
Going down the group, the solubility of these sulfates decreases
This decreasing solubility is attributed to an increase in lattice energy and a relative decrease in hydration energy as the size of the Group 2 cation increases