chem paper 2 2023
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
what is limewater an aqeous solution of
calcium hydroxide
whats on compound to a product of the reaction of the compound with ehtanoic acid
sodium carbonate
carbon dioxide
which isnturment method can be used ot show the prescence of potassium ions in the medecine
flame emmision spectroscopy
whats a greenhouse gas nitrogen or water vapour
water vapour
explain why the increase in world population may have caused the increase in the concentration of methane in the atmosphere
more wast produced
which produces more methane on decomposition
describe two potential effects of the increase in the mean temperatrue of the atmosphere at the Earth’s surface
melting ice
rising sea levels
loss of habitat
The mean temperature of the atmosphere at the Earth’s surface has increased.
Most scientists think that this has been caused by an increase in the concentration of
greenhouse gases in the atmosphere.
Give one reason why some scientists do not accept this theory.
the earth goes through cycles of temp change
Explain why the Rf values for the red dye are different using the two types of chromatography paper.
Rf value is smaller for paper A
because red dye is more attracted to paper A than B
so red dye
what other changes to the investigation could result in a different Rf value for the red dye
use different solvent
whats the solvent
the water
whats the solute
the dye
Give the general name for the four different monomers which make up the structure shown
nucleotides
When 4.68 g of hydrated copper sulfate changes into anhydrous copper sulfate:
• 2.99 g of anhydrous copper sulfate is produced
• 1.47 kJ of energy is taken in from the surroundings.
The equation for the reversible reaction is:
hydrated copper sulfate ⇌ anhydrous copper sulfate + water
Calculate the maximum mass of water that can be produced from 11.7g of hydrated copper sulfate.
4.68 - 2.99 = 1.699
11.7 / 4.68 × 1.69 = 4.23g
When 4.68 g of hydrated copper sulfate changes into anhydrous copper sulfate:
• 2.99 g of anhydrous copper sulfate is produced
• 1.47 kJ of energy is taken in from the surroundings.
When 4.68 g of hydrated copper sulfate changes into anhydrous copper sulfate:
• 2.99 g of anhydrous copper sulfate is produced
• 1.47 kJ of energy is taken in from the surroundings.
15 / 2.99 × 1.47 = 7.37
What is the colour of the mixture when a new equilibrium position is reached?
darker shade of brown
same shade of brown
mixture is a lighter shade of brown
lighter shade
2 HI(g) ⇌ H2(g) + I2(g)
Explain the effect of increasing the pressure on the equilibrium position of this reaction.
no effect on equilibrium
because there’s an equal number of gas molecules on both
The reaction mixture is green when both copper compounds are present in a solution at equilibrium.
How can the equilibrium position be shifted to make the reaction mixture more yellow?
add more hydrochloric acid
add more water
leave the reaction mixture for 30 minutes
add more hydrochloric acid
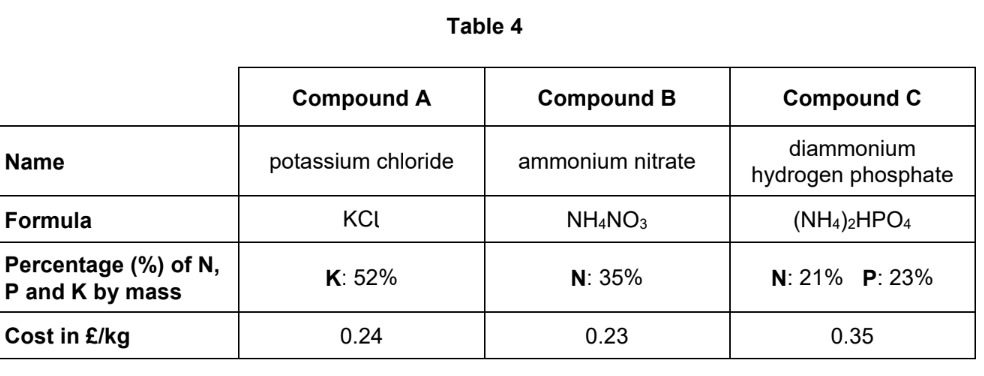
A scientist analysed the percentages of nitrogen, phosphorus and potassium in a soil.
The percentages of nitrogen and of potassium in the soil were lower than the percentages needed for high agricultural productivity.
There was sufficient phosphorus in the soil for high agricultural productivity.
Evaluate the use of the compounds in Table 4 to improve the agricultural productivity of this soil.
how is potassium chloride obtained form the eath
mining
Name one other compound that could be used instead of potassium chloride (compound A) to give a similar improvement in agricultural productivity.
potassium sulfate
Nitric acid is needed to produce ammonium nitrate (compound B).
Name a compound needed to produce nitric acid.
ammonia
Phosphate rock contains phosphorus compounds.
Plants absorb phosphorus from compounds dissolved in rainwater.
Suggest why phosphate rock cannot be used directly as a fertiliser.
insoluble in water
Phosphate rock can be treated with different acids to produce salts useful as fertilisers.
Name the salts which are produced by treating phosphate rock with:
• sulfuric acid
• phosphoric acid.
sulfuric acid - calcium sulfate
phosphoric acid - calcium phosphate