Hydroxy Compounds
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
General forumla of alcohols
R-OH
General formula of phenol
-OH DIRECTLY attached to an aromatic ring
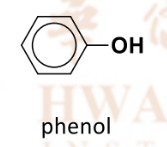
Why do alcohols/phenols have higher boiling points than their isoelectronic hydrocarbons?
Strength of dispersion forces is about the same due to similar size of electron cloud
More energy is required to overcome intermolecular hydrogen bonding in alcohols
which are stronger than the disperson forces in alkanes
Note: larger e- cloud —> higher probability of distortion of e- cloud —> stronger and more significant dispersion forces —> difference between bps decreases
boiling point increases/decreases as alkyl chain length increases
alkyl chain length increases
electron cloud size increases
more energy required to overcome stronger dispersion forces between alcohol molecules
Why are alcohols more soluble in water than corresponding alkanes(same carbon skeleton)?
more energy released from stronger hydrogen bonding formed between alcohol molecules and water molecules
is more able to overcome
the dispersion forces and intermolecular hydrogen bonding between alcohol molecules +
the hydrogen bonding between water molecules
As alkyl chain length increases, the solubility of alcohols increases/decreases
decreases:
hydrocarbon chain length increases
dispersion forces between alcohol molecules increases
energy released from hydrogen bonding between alcohol molecules and water molecules
is less able to overcome the stronger dispersion forces between alcohol molecules and hydrogen bonding between water molecules
Why are phenols moderately soluble in water?
large alkyl groups increase strength of dispersion forces
energy released from hydrogen bonding between alcohol molecules and water molecules
is less able to overcome the stronger dispersion forces between alcohol molecules and hydrogen bonding between water molecules
Alcohols can act as Bronsted acids or Bronsted bases because…
presence of OH group
O-H bond can break to give H+ and R-O-)alkoxide ion
electron-deficient hydrogen that can be removed in the presence of a sufficiently strong base (Bronsted acid: proton donor)
electron-rich oxygen in that can be protonated in the presence of a sufficiently strong acid (Bronsted base: proton acceptor)
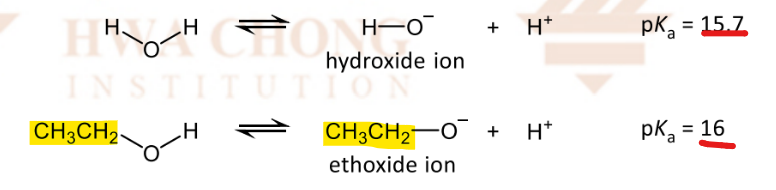
Why are alcohols weaker acids than water?
alkyl groups in alcohol are inductively electron-donating
intensifying the negative charge on the alkoxide ion, destabilising it
deprotonation is able to take place less easily
Note: length of alkly group does not have significant impact (focus on stability of alkoxide ion)
Effect of electron-withdrawing groups((e.g. −NO2, –F, –Cl, –Br, –I, –COCH3, –CO2H, –CN, −CO2R, –NH2, –OH, –OCH3) on acidity of alcohol
electron-wtihdrawing groups disperse the negative charge on the alkoxide ion/phenoxide
stabilising it and promoting ionisation of alcohol
increasing the acidity of the alcohol
higher Ka, lower pKa
Effect of electron-donating groups(e.g. –CH3) on acidity of the alochol
electron-withdrawing group intensifies the negative charge on the alkoxide/phenoxide
destabilising it, making it less easy for the alcohol to be deprotonated
lowering the acidity of the alcohol
lower Ka, higher pKa
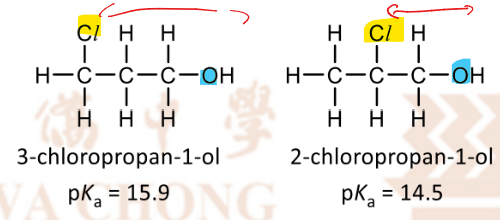
Effect of distance of electron-withdrawing group from the negatively oxygen of alkoxide ion on acidity of the alcohol
as distance increases
inductive effect decreases [inductive effect does NOT apply to phenols]
negative charge is less dispersed, alkoxide is less stable and less likely to be deprotonated
lower acidity, lower Ka, higher pKa
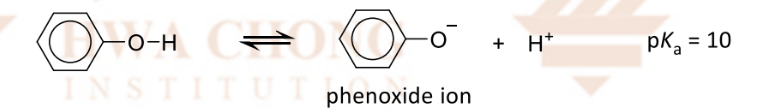
Why are phenols more acidic than alcohols?
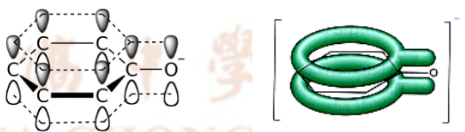
the negative charge of the oxygen an be delocalised into the benzene ring
making the alkoxide more stable and more likely to deprotonate
more acidic, higher Ka, lower pKa
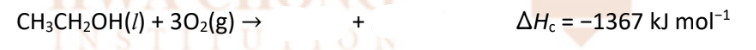
Combustion of alcohols
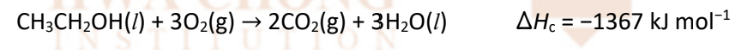
acid-metal reaction with sodium for alcohol
RnCs: Na(s0, room temperature
Observations: slow effervescence of hydrogen gas
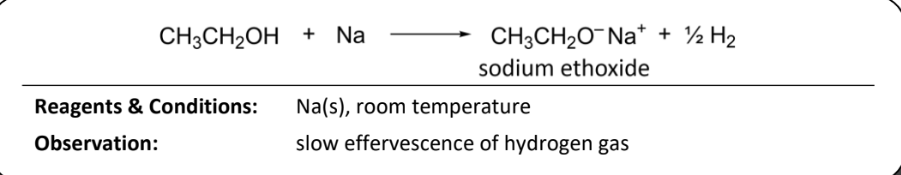
acid-metal reaction with sodium for phenol
RnCs: Na(s), room temperature
Observation: rapid effercsence of hydrogen gas
acid-base reaction for sodium hydroxide with alcohol
not possible
alcohols are not acidic enough to react with NaOH(aq)
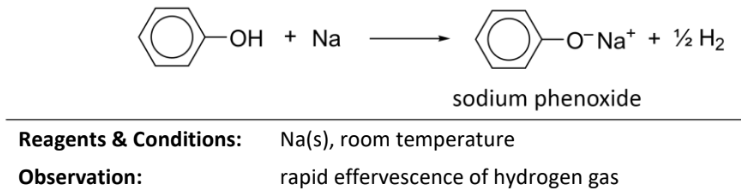
acid-base reaction for sodium hydroxide phenol
RnCs: NaOH (aq), room temperature
Observation: coudy mixture dissolves to form a colourless homogenous solution
Nucleophilic substituition with phosphorus halides
RnCs: PCl5 (phosphorus(V) chloride)
Observation: dense white fumes of HCl produced
Note: good distinguishing test for alcohol group (given carboxylic acid is absent)

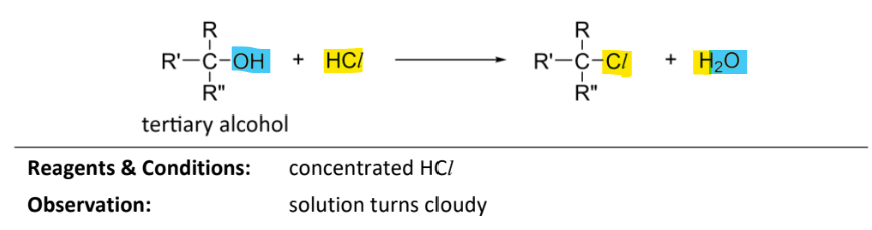
nucleophilic substitution of tertiary alcohol with hydrogen chloride
RnCs: concentrated HCl
Observation: solution turns cloudy
(faster rate than with primary or secondary alcohol)
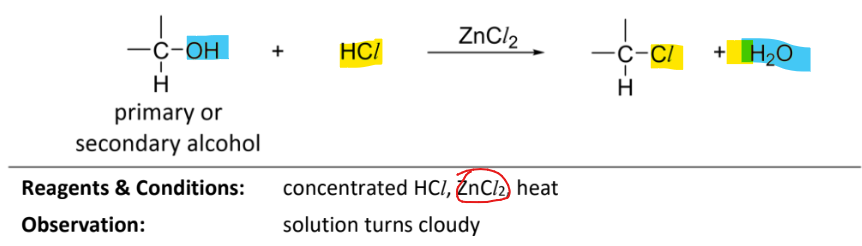
nucleophilic substitution of primary or secondary alcohol with hydrogen chloride
RnCs: concentrated HCl, ZnCl2(catalyst), heat
Observation: solution turns cloudy
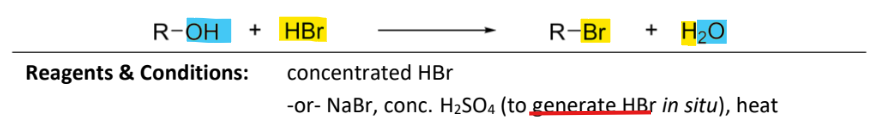
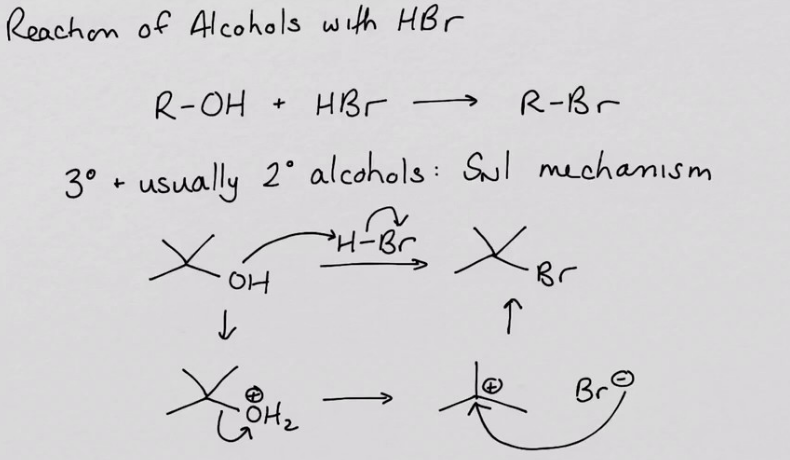
nucleic substitution with hydrogen bromide
RnCs: concentrated HBr (aka hydrobromic acid)
or NaBr, conc, H2So4 (to generate HBr), heat
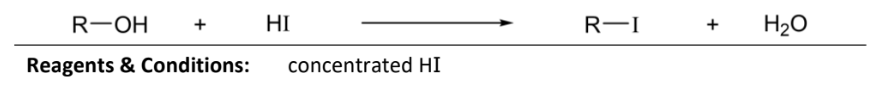
nucleic substituition with hydrogen iodide
RnC: concentrated HI (aka hydroiodic acid)
Why don’t phenols react in nucleic substitution reactions?
lone pair on oxygen atom delocaliseses into the benzene ring, resulting in a partial double bond character in the C-O bond. The C-O bond is stronger and harder to break
nucleophile approaching the C-O bond from the rear side is hindered by the benzene ring. electron cloud of benzene ring repels the lone pair of electrons on nucleophile
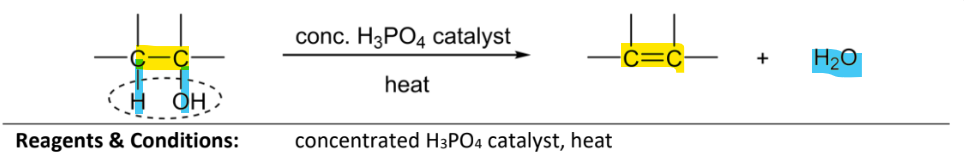
dehydration/elmitation of alcohol
RnCs: concentrated H3Po4 catalysst (Phosphoric acid), heat
or excess concentrated H2So4, heat (but not preferred as it is an OA and can produce unwanted side products)
or Al2O3, heat
Note:
alcohol must ave a H atom bonded to the C atom adjacent to another C atom bearin g the hydroxyl group
Why doesn’t phenol react in dehydration?
strong C-O bond that is not easily broken
carbon atom adjacent to C atom attached to hydroxy group is not attached to any H atom
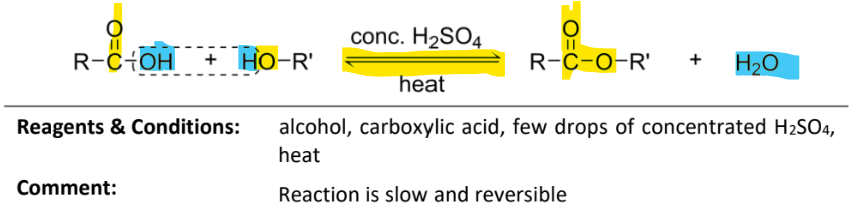
condenstation of alcohol with carboxylic acid
RnCs: carboxylic acid, few drops of concentrated H2So4, heat
Note: reaction is slow and reversible
Why can’t phenols react with carboxylic acids?
lone pair of electrons on oxygen atom are less availale to attack carboxyl carbon of carboxylic acid due to delocalisation of the lone pair into the benezene ring
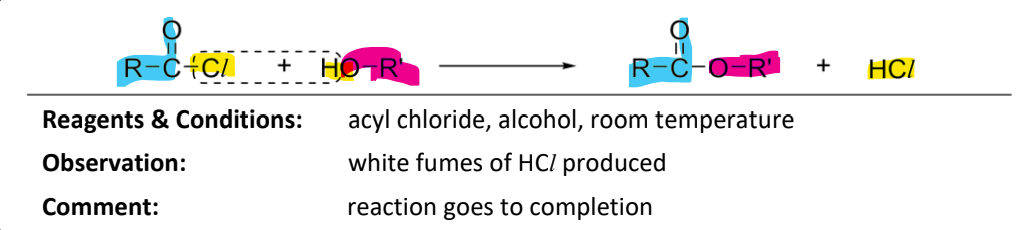
Condensation of alcohol with acyl chloride
RnC: acyl chloride, room temperature
observation: white fumes of HCl
note:
acyl chloride reacts more readily with alcohol, hence no catalysts required
reaction goes to completion and is irreversible
Condenstaion of phenol with acyl chloride
RnC: acyl chloride, room temperature
obersvation: whtie fumes of HCl
Note:
phenol can only react with acyl chloride for condensation as it is a weaker nucleophile
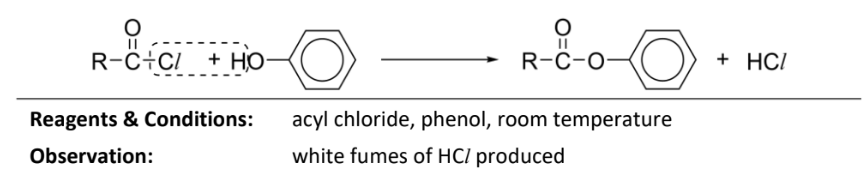
How to increase yield of condensation of phenol with acyl chloride?
reaction is conducted under basic conditions where Na or NaOH is present to generate a phenoxide ion
ion has a negative charge and is a stronger nucleophile
why can’t oxidation using K2Cr2O7 or KMnO4 occur for tertiary alcohols?
there must be at least one hydrogen atom bonded to carbon atom bearing the hydroxy group for oxidation to occur using the above reagents
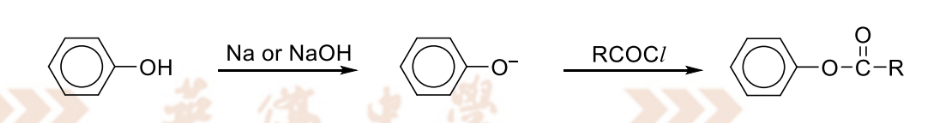
oxidation of primary alcohol to aldehyde
RnCs: K2Cr2O7, H2SO4(aq), heat with immediate distillation
Observation: orange(Cr2O7) solution turns green (Cr3+)
Note:
distillation is to prevent further oxidation to form carboxylic acid by distilling aldehyde away since it has a lower bp
KMnO4 cannot be used because it is stronger as a OA and wil oxidsie the alcohol directly into carboxylic acid
only K2Cr2O7(potassium dichromate) is selective enough
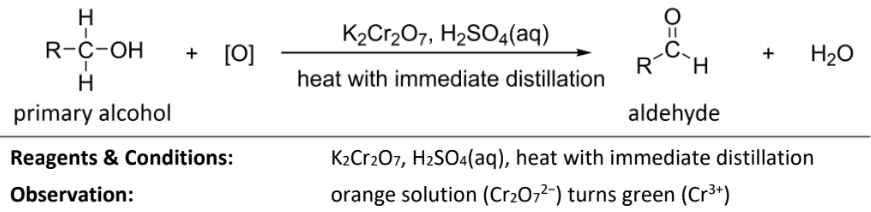
oxidation of primary alcohol to carboxylic acid
RnCs: KMnO4, H2SO4, heat under reflux
or K2Cr2O7, H2SO4, heat under reflux
Observations: purple(MnO4-) solution turns colourless (Mn2+)
orange(Cr2O7) solution turns green (Cr3+)
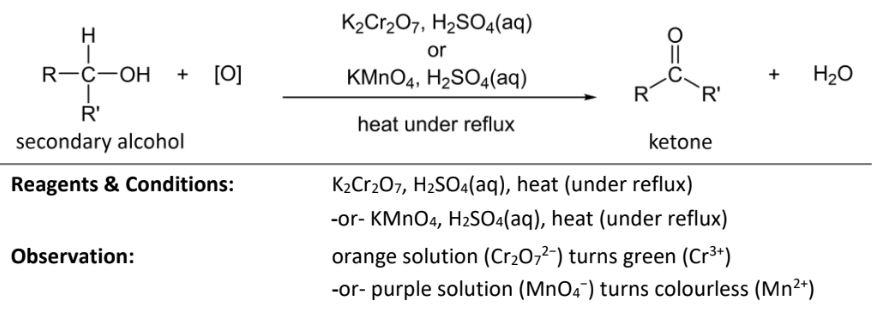
Oxidation of secondary alcohol to ketone
RnCs: KMnO4/K2Cr2O7
+H2SO4, heat under reflux
Observations: purple(MnO4-) solution turns colourless (Mn2+) OR
orange(Cr2O72-) solution turns green (Cr3+)
Using oxidation as a distinguishing test, what are the expected observations for primary, secondary and tertiary alcohols respectively?
primary and secondary: colour change (either orange to green OR purple to colourless depending on AI)
tertiary: no colour change
tri-iodomethane (iodoform) test
RnCs: I2(aq), NaOH(aq), warm
Observations: Yellow ppt(CHI3) formed
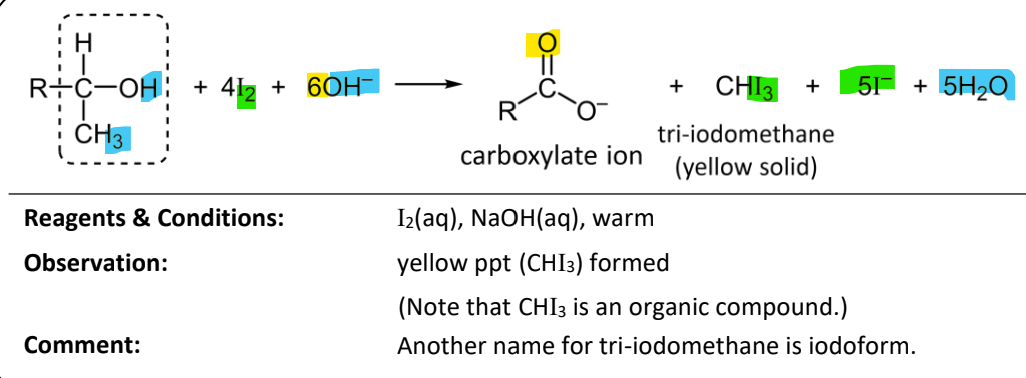
Why is the tri-iodomethane test known as a step-down reaction?
the reaction involves the breaking of the C-C bond to remove the methane group
this shortens the carbon chain by a single carbon
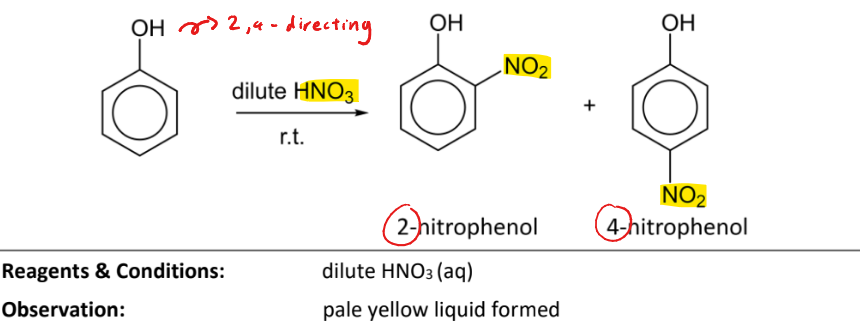
Electrophilic substitution with nitric acid
RnCs: dilute HNO3(aq)
Observation: pale yellow liquid formed
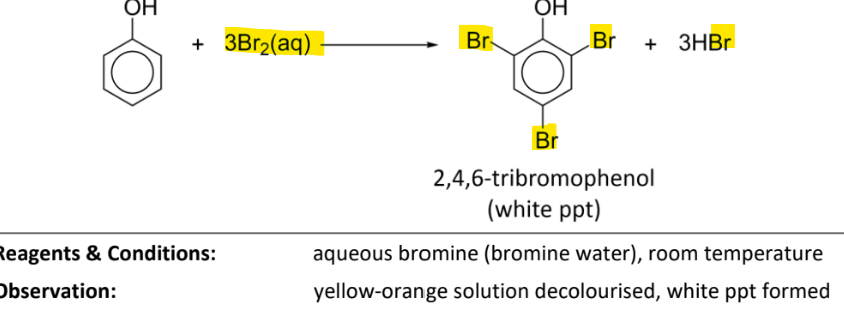
electrophilic substituition with bromine (bromination)
RnCs: Br2(aq) (bromine water), room temperature
observation: yellow-orange solution(bromine water) decolourised, white ppt formed
complex formation with iron(III) chloride
RnCs: neutral FeCl3 (iron(III) chloride), room temperature
conditions: violet colouration
