AQA A level Chemistry 3.1.4 Energetics
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
What is meant by enthalpy change? (1)
Heat energy change at a constant pressure
What are the units and symbol for enthalpy change? (2)
- Symbol: ΔH
- Unit: kJ mol-1
What is ΔH for an exothermic reaction? (1)
Negative
What is ΔH for an endothermic reaction? (1)
Positive
What happens in an exothermic reaction? (1)
The system gives out heat to the surroundings
What happens in an endothermic reaction? (1)
The system takes in heat from the surroundings
What are the conditions for standard enthalpy change (ΔH°)? (2)
- Temperature: 298 K
- Pressure: 100 kPa
What is the standard enthalpy of combustion (ΔH°c)? (2)
- The enthalpy change when 1 mole of a substance is completely burned in oxygen
- Under standard conditions, with all substances in their standard states.
Are enthalpies of combustion reactions exothermic or endothermic? (1)
Always exothermic.
How do you balance combustion equations? (3)
E.g. complete combustion of C3H8
1. Balance carbons on the CO2 / CO / C -> 3CO2
2. Balance hydrogens on the water molecule -> 4H20
3. Balance oxygens on the O2 molecule - 502
C3H8(g) + 5O2(g) -> 3CO2(g) + 4H20(l)
What is the standard enthalpy of formation (ΔH°f)? (2)
- The enthalpy change when 1 mole of a compound is formed from its elements
- Under standard conditions, with all substances in their standard states.
What is the enthalpy of formation of elements? (1)
Zero
Write an equation that represents a reaction equal to the standard enthalpy of formation of NAHCO3(s)
Na(s) + ½ H2 (g) + C (s) + 1.5 O2(g) → NaHCO3 (s)
What does Hess' law state? (1)
The enthalpy change for a reaction is independent of the route taken.
When is hess' law needed for enthalpy change calculations? (1)
For enthalpy changes that cannot be determined experimentally e.g. due to a solid dissolving etc
If enthalpy of combustion values are given, how can you calculate enthalpy change?

If enthalpy of formation values are given, how can you calculate enthalpy change?
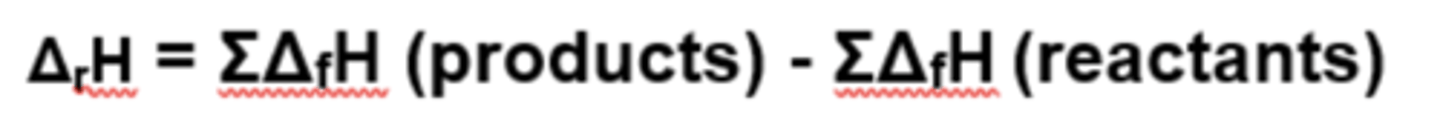
What is the formula for heat energy transferred to water? (5)
- q = m x c x ΔT
- q = heat energy (J)
- m = mass of water (g)
- c = specific heat capacity of water (given)
- ΔT = temperature (either K or degrees)
How do you convert 1cm3 of water to g? (1)
1cm3 = 1g
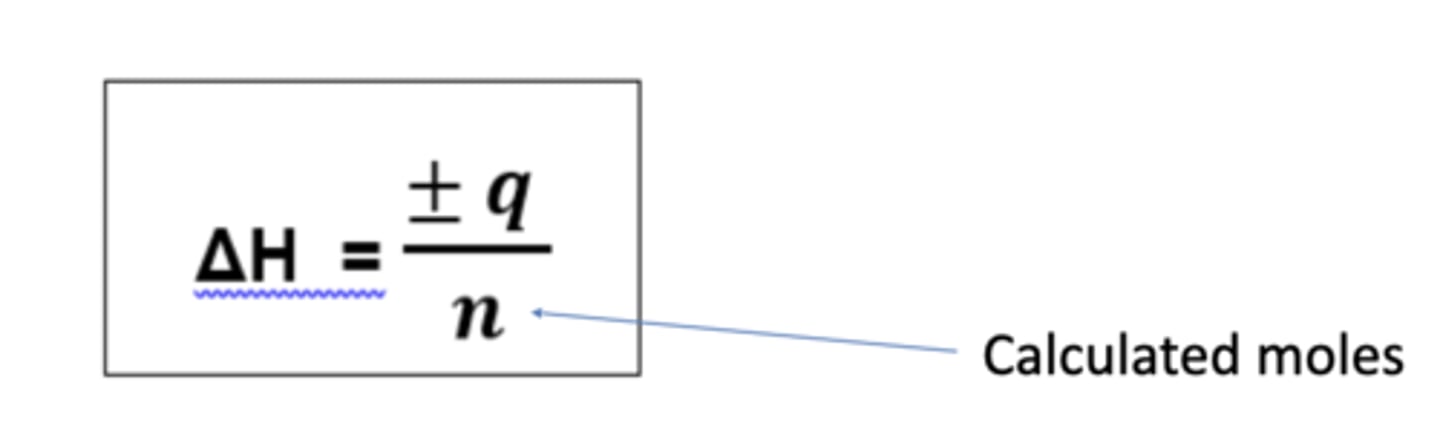
How do you calculate enthalpy change from q? (3)
- ΔH = kJ mol-1 (so divide q by 1000 to get it into kJ)
What are the two ways of carrying out calorimetry? (2)
1. A fuel is used to heat up a known volume of water
2. A reaction in soloution is carried out in an insulated container
What is the first step in using a fuel to heat up water? (1)
Record the mass of the spirit burner and the fuel with the cap on
What do you do after recording the mass of the spirit burner? (1)
Place a known volume of water in a beaker and record its temperature.
How is the fuel used to heat the water? (2)
- The flame is placed close to the beaker of water, and heat-proof guards are used to minimise heat loss.
- Stir the water while heating.
What should you do when the water's temperature has increased by a reasonable amount? (2)
- Extinguish the flame by placing the lid on the burner to avoid fuel evaporation.
- Monitor and record the highest temperature reached by the water.
How do you calculate the mass of fuel burned? (1)
Reweigh the spirit burner with the lid on and calculate the difference in mass from the initial measurement.
What are two reasons why the value of enthalpy change obtained is less than the data book value? (2)
Heat loss to the surroundings.
Incomplete combustion.
What are three other reasons why the value obtained is less exothermic than the data book value? (3)
Non-standard conditions.
Evaporation of fuel/alcohol.
Heat loss to the can.
How can poor stirring of the water affect the result? (1)
It can cause the temperature reading to be incorrect.
What is used as the insulated container in method 2 of carrying out calorimetry ? (1)
A polystyrene coffee cup
What is the first step in method 2 of carrying out calorimetry? (2)
1. Pour the aqueous reactant into the cup
2. Record the temperature every minute for three minutes to ensure the solution is at the same temperature as the room.
What do you do on the fourth minute in method 2 of carrying out calorimetry? (2)
- Add the other reactant and stir to ensure the reactants mix thoroughly for a complete reaction and to ensure the temperature is even throughout the solution
- Do not record the temperature
What should you do from the fifth minute onwards? (1)
Record the temperature at the fifth minute and continue to record it every minute for up to ten minutes
What is bond dissociation energy? (1)
The enthalpy change to break the bond in 1 mole of gaseous molecules, forming gaseous atoms.
What is mean bond enthalpy? (1)
The enthalpy required to break 1 mole of covalent bonds, averaged over a range of compounds.
Why is the enthalpy change calculated from mean bond enthalpy data not 100% accurate? (1)
Bond enthalpies are average values from a range of compounds.
What is the formula to calculate enthalpy change (ΔH) using bond enthalpies? (1)
ΔH = ΣΔH (bonds broken) - ΣΔH (bonds formed)