Lec 7 - Pathology of the Exocrine Pancreas
1/96
Earn XP
Description and Tags
Exam 1 - Dr. Sandy
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
97 Terms
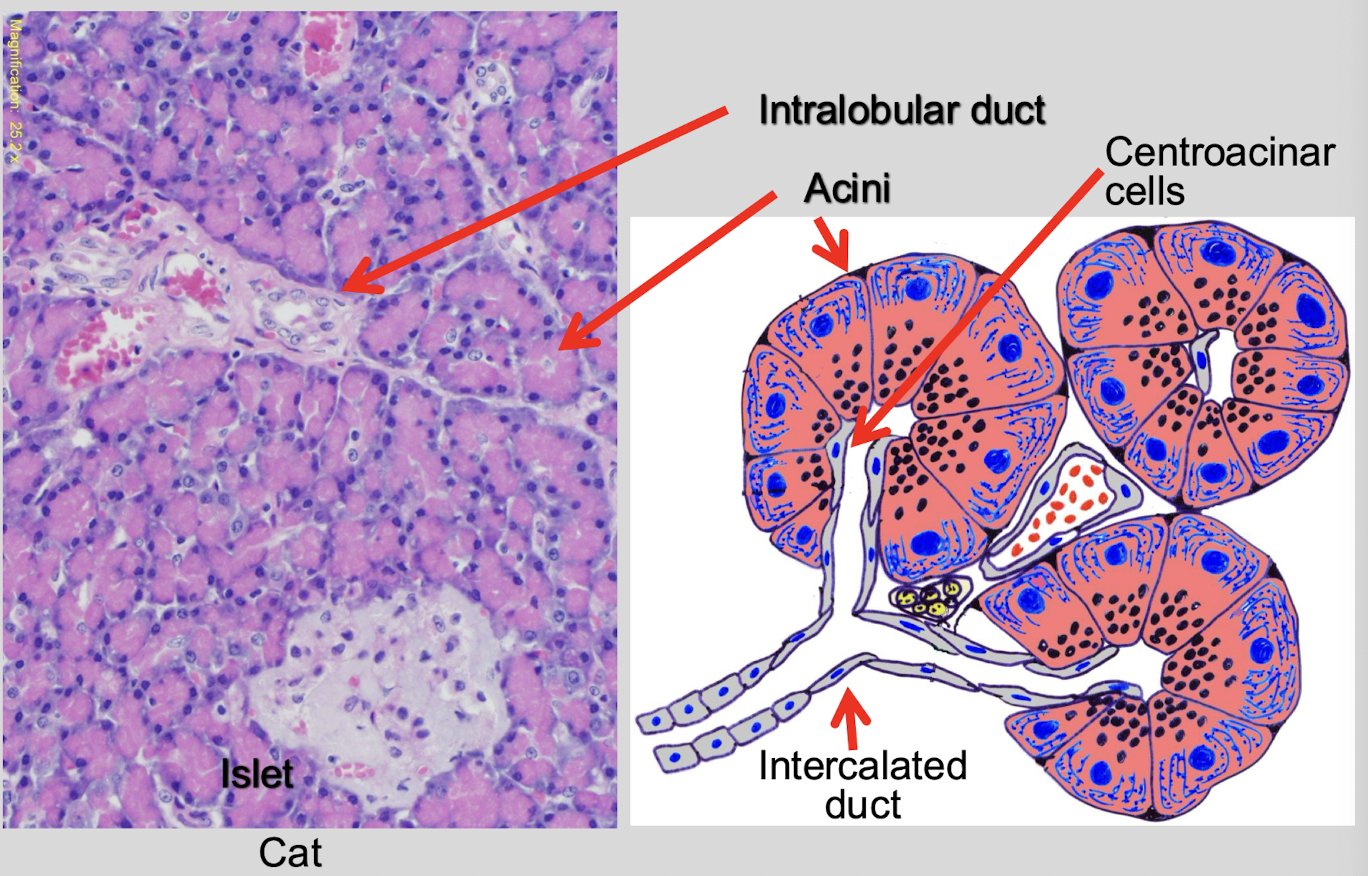
Describe the structure of the pancreas.
lobulated, tubuloalveolar gland
What percentage of the pancreas parenchyma is the exocrine component? Endocrine?
exocrine = 85%
endocrine = 15%
T/F: The subgross lobules of the pancreas are separated by delicate connective tissue.
TRUE
Pancreatic ducts drain into which organ?
the duodenum
Each pancreatic lobule contains what?
tightly packed acini, ducts, vessels, & scattered islets
What are the primary secretions of the exocrine pancreas?
digestive enzymes
bicarbonate rich fluid for neutralization of gastric acid
What are the 2 ways secretions of the exocrine pancreas are controlled?
neural regulation → vagal stimulation
hormonal stimulation (by neuroendocrine cells)
Where are S cells located and what do they secrete?
duodenal crypts
secretin
What does secretin stimulate?
secretion of H2O & bicarbonate from centroacinar & ductal cells of pancreas, duodenum, & biliary epithelium
What stimulates S cells to secrete secretin?
acidic chyme
Where are the I cells located and what do they secrete?
small intestine
cholecystokinin (CCK)
What does the secretion of CCK stimulate?
secretion of digestive enzymes from acinar cells
What stimulates I cells to secrete CCK?
fat & protein in partially digested food from stomach
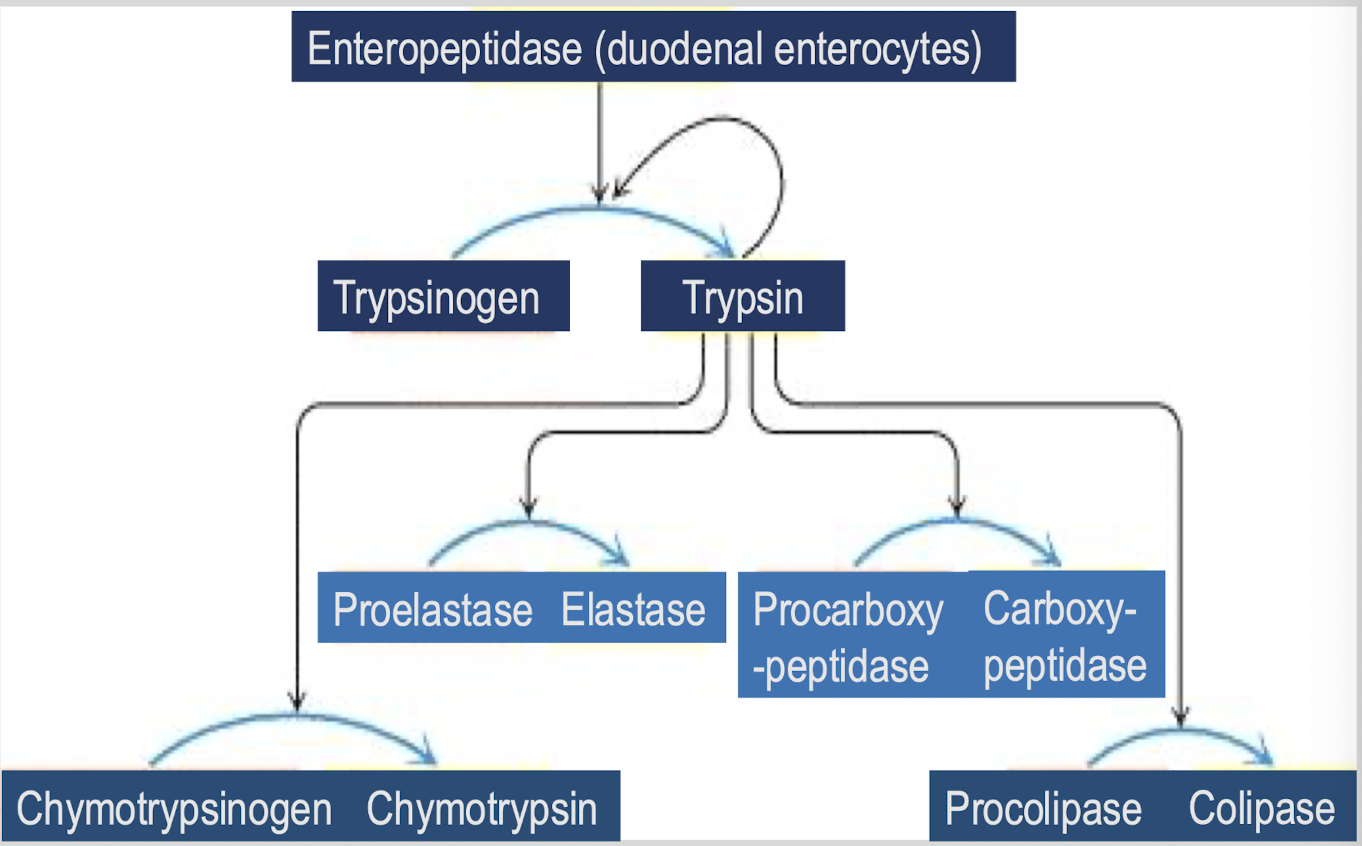
Which digestive enzymes aid in fat digestion?
lipase, procolipase, prophospholipase A2
Which digestive enzymes aid in protein digestion?
typsinogen, chymotrysinogen, proelastase, procollagenase
Which digestive enzymes aid in carbohydrate digestion?
amylase
Which digestive enzymes aid in nucleic acid digestion?
nuclease (DNAse I and RNAse)
Which digestive enzymes are secreted as zymogens (inactive forms)?
procolipase, prophospholipase A2, trypsinogen, chymotrypsinogen, proelastase, procollagenase
How is trypsinogen activated?
activated in the intestinal lumen by enteropeptidase form duodenal epithelium
What does active trypsin do?
activates other pancreatic proenzymes
What are the 3 major diseases dues to dysfunction of the exocrine pancreas?
exocrine pancreas insufficiency (EPI)
acute pancreatitis
chronic pancreatitis
What is EPI and what does it result in?
inadequate production of digestive enzymes → maldigestion & malabsorption
What are the clinical signs of EPI? What species/breeds is it common in?
weight loss, diarrhea, steatorrhea
GSD
What is acute pancreatitis and what does it lead to?
acute inflammation of the pancreas due to premature activation of digestive enzymes → pancreatic autodigestion & necrosis
T/F: Acute pancreatitis only causes severe local inflammation.
FALSE - causes local and systemic
What is chronic pancreatitis and what does it lead to?
progressive inflammation w/ fibrosis & acinar loss → gradual reduction in enzyme production
T/F: Chronic pancreatitis often results in secondary EPI in advanced stages.
TRUE
What is the prominent & defining feature of acute pancreatitis (acute pancreatic necrosis)?
necrosis
What are the potential risk factors associated with acute pancreatitis?
exact cause unknown in dogs & cats
hereditary (mini schnauzers w hyperlipidemia, yorkies)
obese, sedentary female dogs
What is acute pancreatitis frequently associated with in cats?
triaditis → pancreatitis, cholangitis, IBD
What are the 3 major mechanisms of acute pancreatitis?
obstruction of pancreatic ducts
direct injury to acinar cells
disturbed intracellular trafficking within acinar cells
What can cause obstruction of the pancreatic duct? What does this do?
calculi or parasites → impairs enzyme outflow & promotes intrapancreatic activation
What may cause direct injury to acinar cells?
drugs (sulfonamindes, KBr, phenobarbital)
chemicals (T-2 mycotoxin, Cassia occidentalis, zinc)
trauma (may occur at surgery)
ischemia (may occur at surgery)
What is associated with disturbed intracellular trafficking within acinar cells? What may cause this?
abnormal stimulation of secretion → dietary indiscretion, obesity, elevated corticosteroids, hyperlipidemia
Pancreatic damage causes further decrease in pancreatic secretion into the small intestine. What does this lead to?
overstimulation of acinar cells by CCK, increasing trypsinogen secretion & promoting further damage
Excess trypsin may overwhelm the activity of local trypsin inhibitors. What are these inhibitors called?
pancreatic secretory trypsin inhibitor (PSTI)
alpha-1-antitrypsin
What are the systemic complications associated with pancreatic injury?
excessive production of pro-inflammatory cytokines
vascular injury (hemorrhage, shock, SIRS, MODS, DIC)
2o islet injury (10-40% risk diabetes)
liver necrosis (inc ALT, focal necrosis)
What is grossly seen in severe cases of acute pancreatic necrosis in dogs?
hemorrhagic pancreas w prominent edema & multi-colored areas
chalky white areas of fat necrosis by lipolytic enzymes (w 2o mineralization)
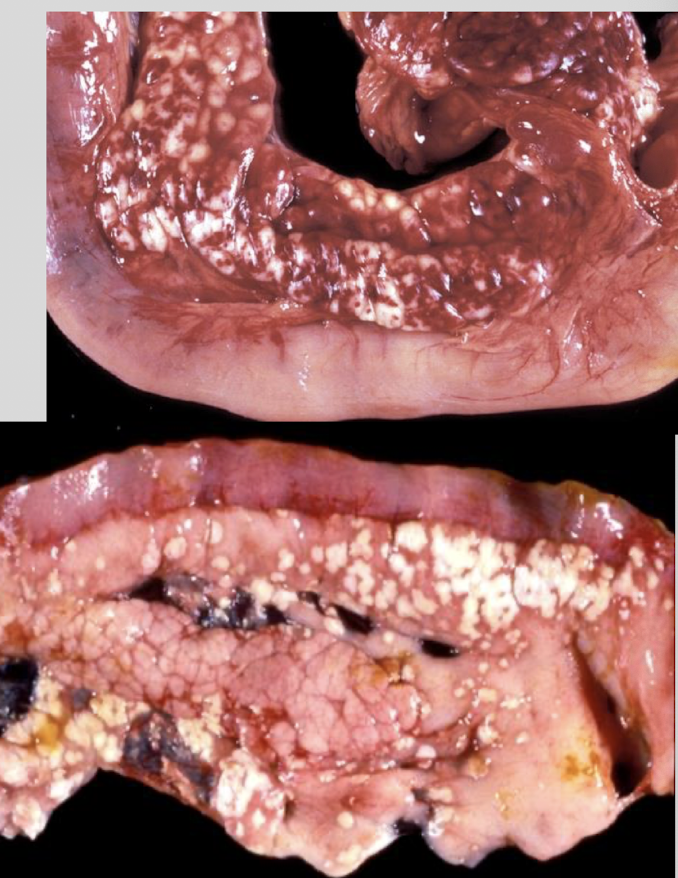
What does the peritoneal fluid of a dog with acute pancreatic necrosis look like? What does peritonitis look like in these cases?
blood stained ± oil droplets
fibrinous adhesions
What is seen in addition to necrosis of acinar tissue, neutrophils, & fat necrosis in dogs with acute pancreatic necrosis?
calicum deposits in saponified fats → peripancreatic fat necrosis
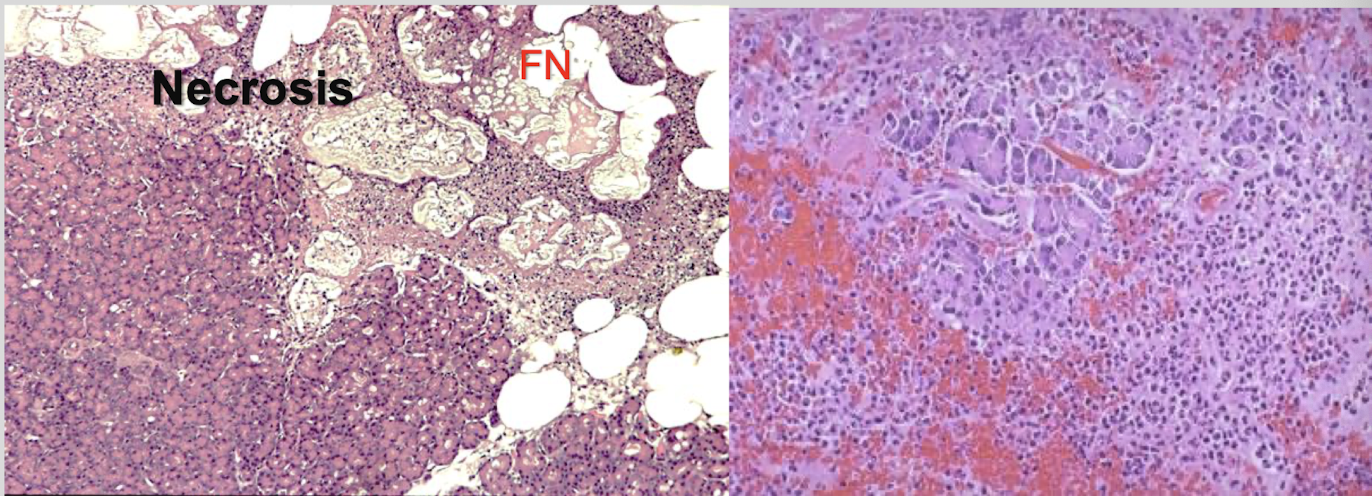
What are the 2 distinct clinical syndromes of acute pancreatitis seen in cats?
acute pancreatic necrosis (like in dogs)
suppurative pancreatitis from ascending bacterial infection (associated w/ cholangiohepatitis - triaditis)
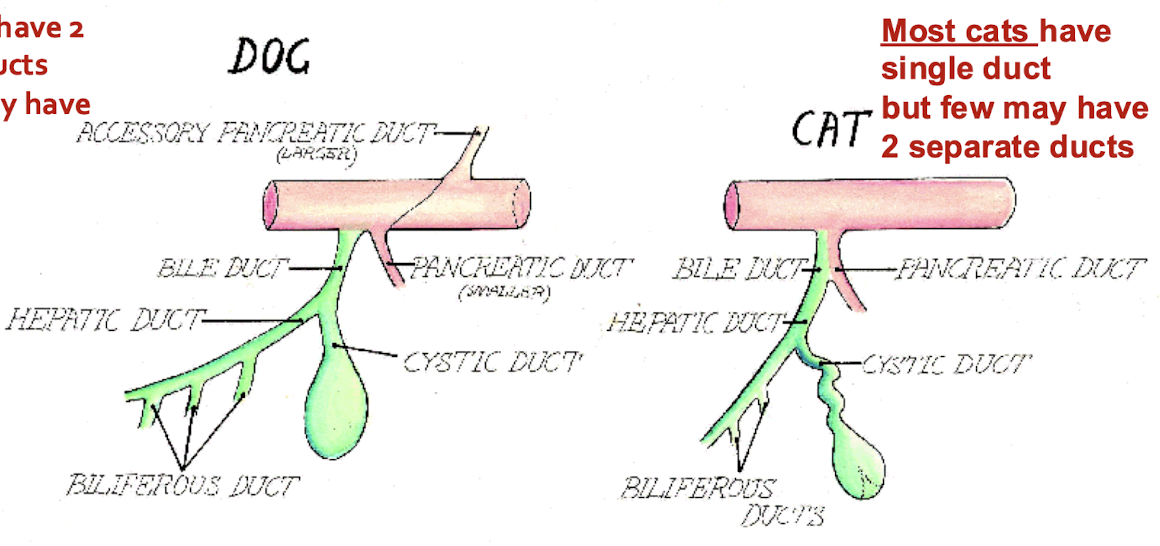
How many pancreatic ducts do most dogs have? What about cats?
dogs: 2 but few may have 1
cats: 1 but few may have 2
Chronic pancreatitis occurs in ALL species due to what?
duct obstruction (may be 2o to flukes)
T/F: The pancreas has poor regenerative capacity of parenchymal cells.
FALSE - modest regen → repair typically involves replacement fibrosis & atrophy
T/F: The pancreas has substantial functional reserve.
TRUE
When does EPI clinically occur?
only after extensive loss of exocrine tissue (>90%)
In what species does EPI occur more commonly?
dogs
What are common causes of EPI in dogs? Cats?
dogs = juvenile pancreatic acinar atrophy
cats = chronic pancreatitis
Exocrine pancreatic atrophy is __________ destruction. Which cells are responsible for this?
immune-mediated
T cells target pancreatic acinar cells
Exocrine pancreatic atrophy is typically preceded by what?
lymphocytic infiltration → termed “subclinical pancreatitis”
What structure is usually spared during exocrine pancreatic atrophy?
islets of Langerhans
T/F: There is a strong genetic association with exocrine pancreatic atrophy.
TRUE - hereditary predisposition
What breeds are most commonly affected by hereditary exocrine pancreatic atrophy?
GSD, rough collies, eurasier dogs
What is the typical age of onset hereditary exocrine pancreatic atrophy?
between 6 and 12 months
What breed is the important exception to exocrine pancreatic atrophy?
greyhounds → disease may affect both exocrine & endocrine pancreas, loss of islets w development of diabetes mellitus
What are the histologic findings of early lesions of exocrine pancreatic acinar atrophy?
intense T cell infiltration (preceding acinar atrophy) → CD4+ and CD8+ lymphocytes infiltrate bt & w/in acini → lymphoid follicles, plasma cells, & marcrophages present during subclinical stage
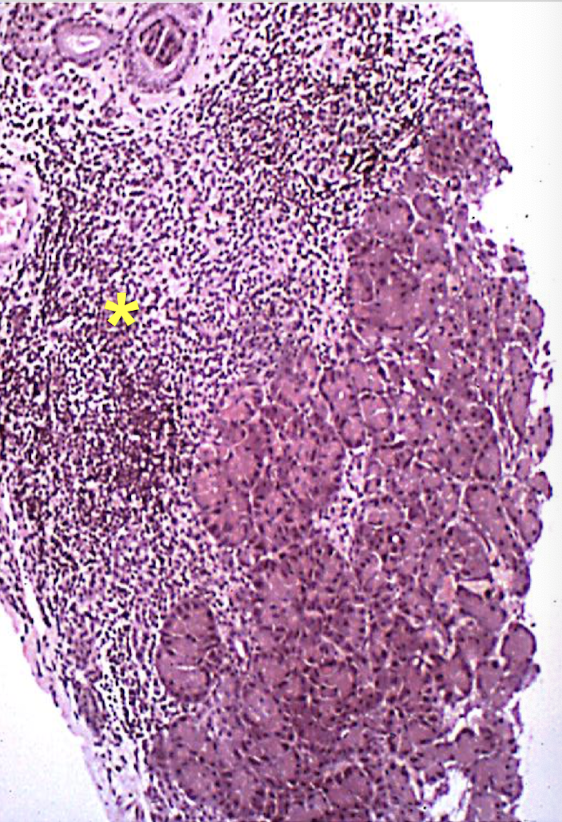
Explain the disease progression of exocrine pancreatic acinar atrophy.
progressive loss of acinar tissue → minimal inflammation in late stage dz → preservation of islets (most breeds)
What is the gross pathology of exocrine pancreatic acinar atrophy?
markedly reduced pancreatic size, thin, translucent, membranous appearance, near complete loss of exocrine parenchyma
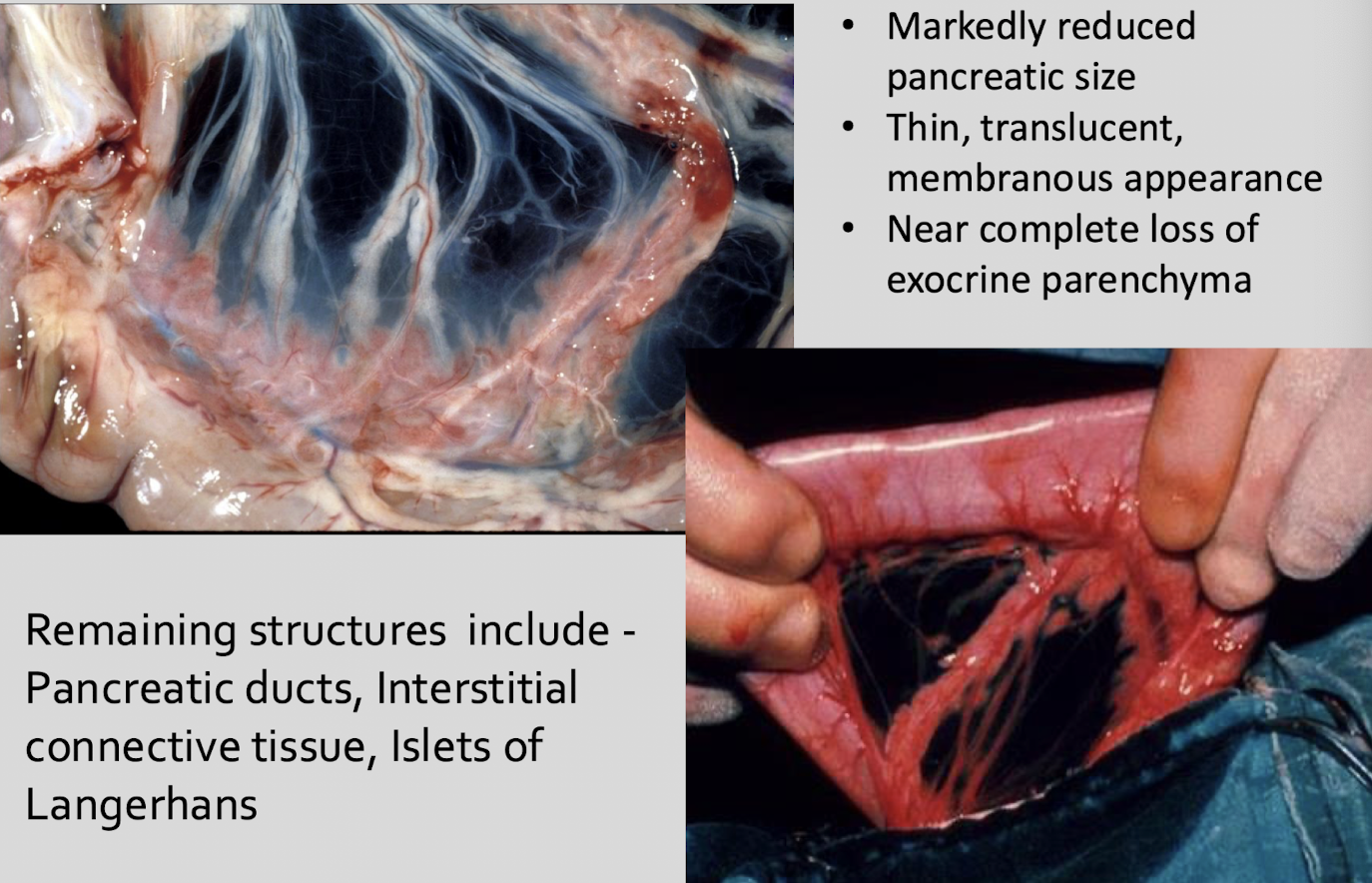
What structures remain in exocrine pancreatic acinar atrophy?
pancreatic ducts, interstitial connective tissue, islets of Langerhans
What is the consequence of amylase deficiency?
impaired carbohydrate digestion
What is the consequence of lipase deficiency?
impaired fat digestion
steatorrhea w pale, greasy stools
What is the consequence of protease deficiency?
impaired protein digestion
muscle wasting & malnutrition
What are the consequences of malabsorption & malnutrition?
undigested nutrients in lumen lead to weight loss, chronic diarrhea, & poor body condition despite normal or increased appetitie
What secondary effect is seen with EPI? What does this cause?
small intestinal dysbiosis (undigested nutrients promote bacterial overgrowth)
further worsens malabsorption & diarrhea
Untreated EPI leads to what?
emaciation
What are the predisposing factors of small intestinal bacterial overgrowth?
loss of normal antibacterial effects of pancreatic secretions
increased availability of undigested nutrients w/in intestinal lumen
increased bacterial populations in duodenum & jejunum
What are the 2 consequences of bacterial overgrowth?
fermentation of carbs
deconjugation of bile acids by bacteria
What does fermentation of carbs produce?
osmotic effects leading to diarrhea
What does deconjugation of bile acids lead to?
impairs fat digestion & absorption → results in increased fecal fat loss
What organisms contribute to the mucosal injury seen with small intestinal bacterial overgrowth? How do they cause injury?
obligate anaerobes like Clostridium & Bacteroides by producing proteases & glycosidases
What is the result of mucosal injury secondary to SIBO?
damaged brush border enzymes & epithelium leads to villous epithelial atrophy & reduced absorptive surface area
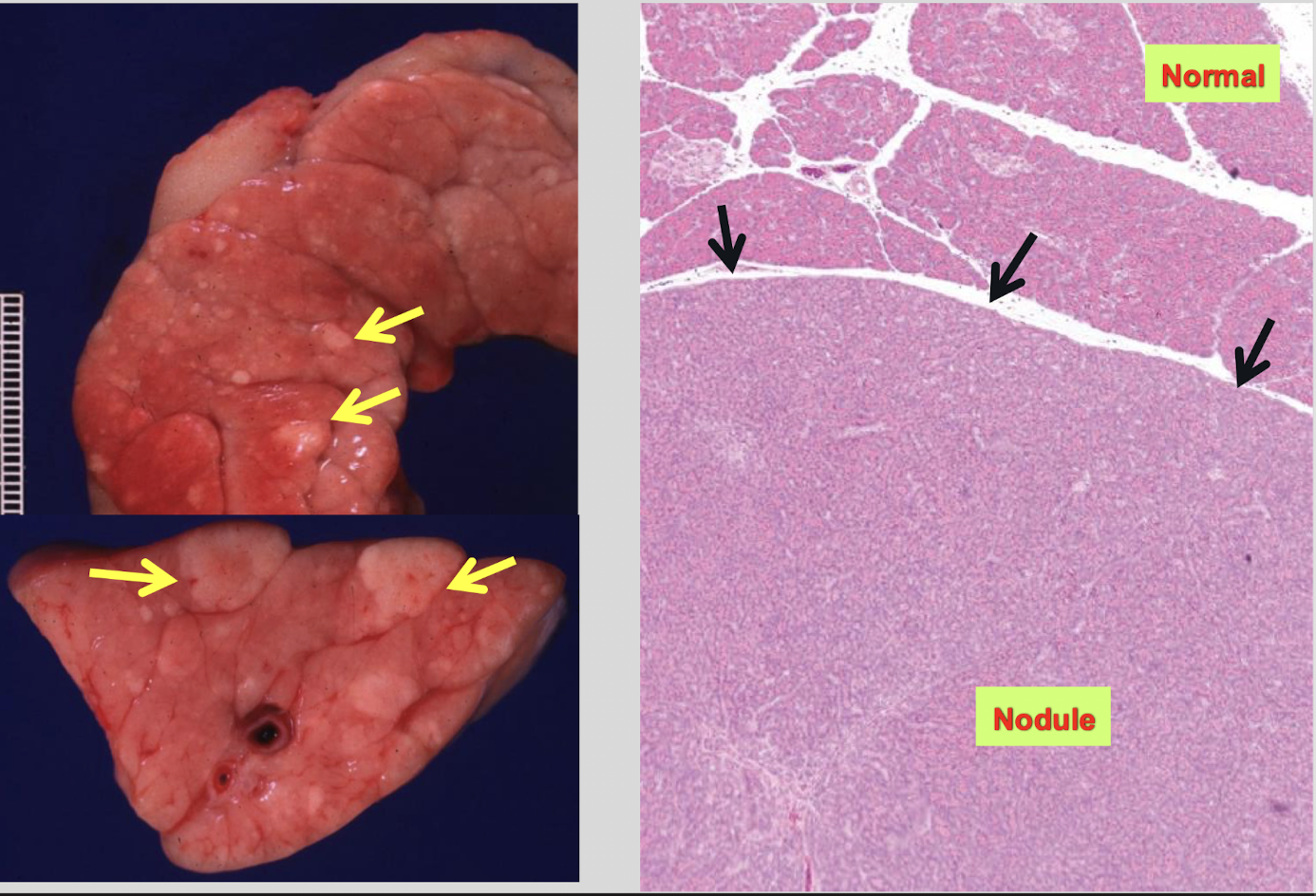
T/F: Pancreatic nodular hyperplasia is a common incidental finding in cats & dogs and is a benign, age related change with no clinical significance.
TRUE
What are the gross features of pancreatic hyperplasia? Where is it found?
multiple small nodules, typically 1-5 mm in diameter
located on the pancreatic surface or within the parenchyma
How do you differentiate pancreatic hyperplasia from neoplasia?
hyperplasia = multifocal, small, well-circumscribed, non-invasive
neoplasia = adenoma or adenocarcinoma, typically solitary & larger, invasive growth & cytologic atypia
Pancreatic carcinoma is a rare neoplasm in both cats & dogs that arises from which cells (in most cases)?
pancreatic acinar cells
What is the gross pathology of pancreatic carcinoma?
usually presents as single mass, gray to yellow w/ frequent hemorrhage & necrosis, firm to schirrous consistency, adhesions to adjacent organs common
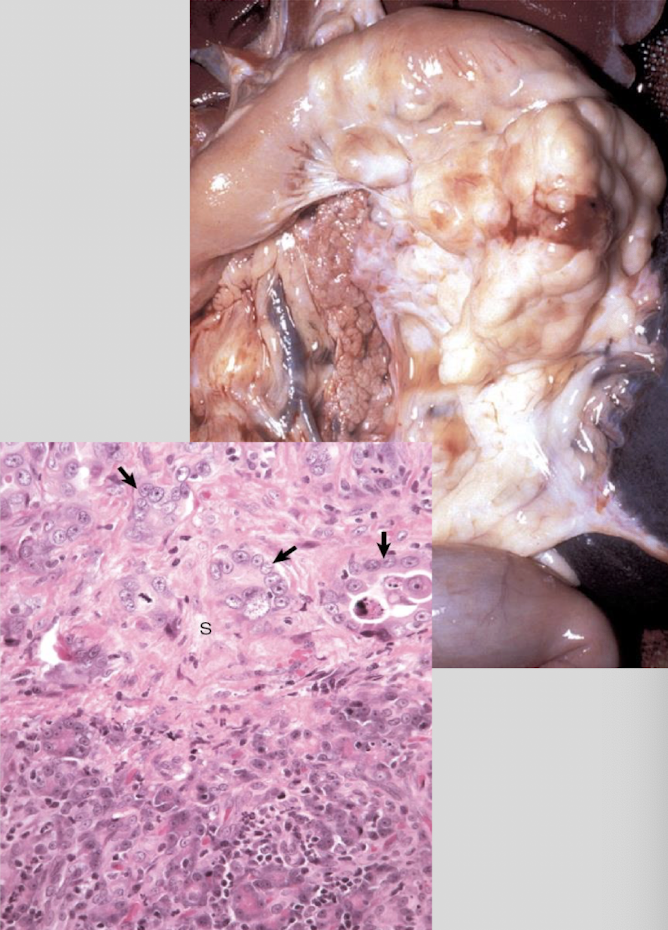
What is the biologic behavior of pancreatic carcinoma?
locally invasive, early metastasis & poor prognosis, peritoneal spread & metastasis typical
What is a frequent metastasis site of pancreatic carcinoma?
liver
What must occur for bile to be released from the gallbladder?
contraction of muscular layer of gallbladder → relaxation of sphincter of heptaopancreatic ampulla
What species are missing a gallbladder?
horses, rats, elephants, & camelids
What happens to bile after a cholecystectomy?
bile continues to flow directly from the liver to intestine → some functional changes but low morbidity
What are the major gallbladder diseases?
cholelithiasis & neoplasia
T/F: Gallstones (chloelithiasis) are common in dogs & cats.
FALSE - uncommon
While primary gallbladder neoplasms are rare, what are they often assoicated with when present?
chronic biliary disease
obstruction or inflammation
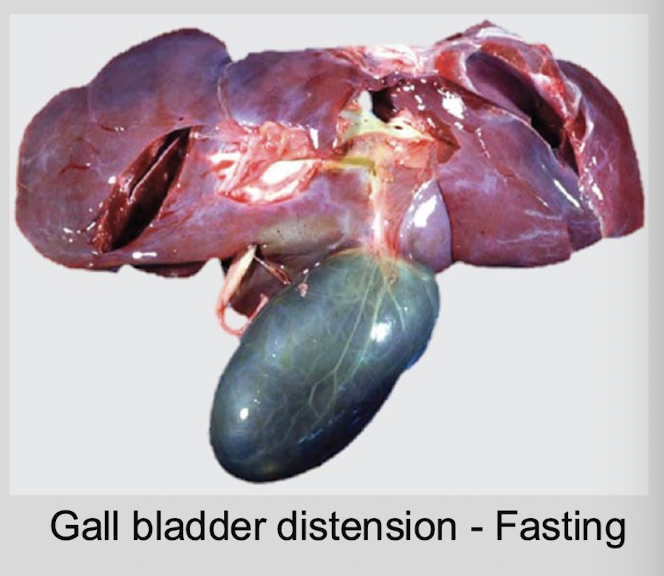
Physiologic distension of the gallbladder is a common incidental finding and is associated with __________. Why is this?
fasting → lack of CCK stimulation
What is the gross appearance of a distented gallbladder?
enlarged & thin walled, filled with bile → NOT a pathologic condition
What are the 2 distinct gallbladder lesions in dogs?
gallbladder mucocoele
cystic mucinous hyperplasia
What is a gallbladder mucocoele?
abnormal accumulation of thick, inspissated mucus w/in gallbladder → gallbladder markedly distended & filled w/ immobile bile & mucus
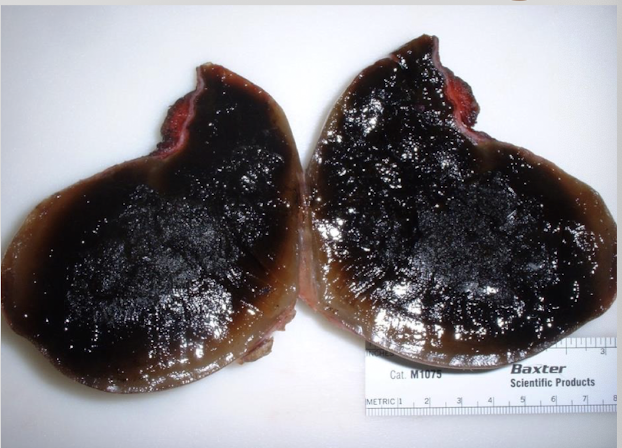
What are the predisposing factors of a gallbladder mucocoele?
hyperadrenocorticism, hyperlipidemia, Shetland Sheepdogs, Mini Schnauzers
T/F: The pathogenesis of gallbladder mucocoeles is incompletely understood.
TRUE
What is the clinical significance of gallbladder mucocoeles?
abdominal pain, biliary obstruction, risk of rupture, bile peritonitis
clnically significant & potentially life-threatening
What is cystic mucinous hyperplasia?
benign proliferative lesion of gallbladder mucosa, characterized by mucin filled epithelial cysts
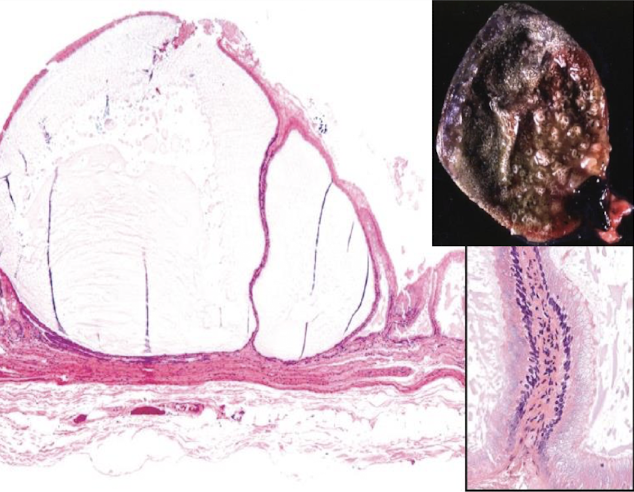
How does cystic mucinous hyperplasia develop?
spontaneous or hormone related → associated w/ progestins
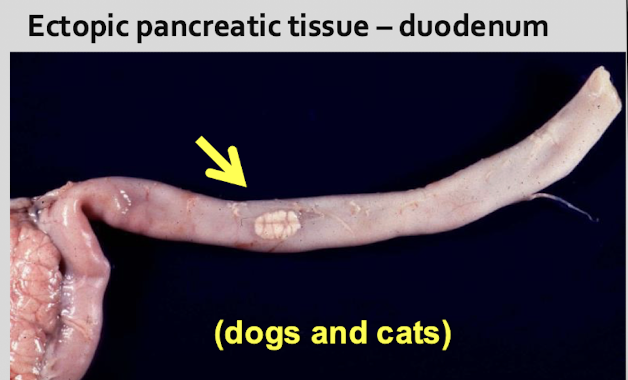
Where are nodules of ectopic pancreatic tissue found? What species are affected by this condition?
duodenum, stomach, spleen, gallbladder, mesentery (= chorisoma)
dogs & cats
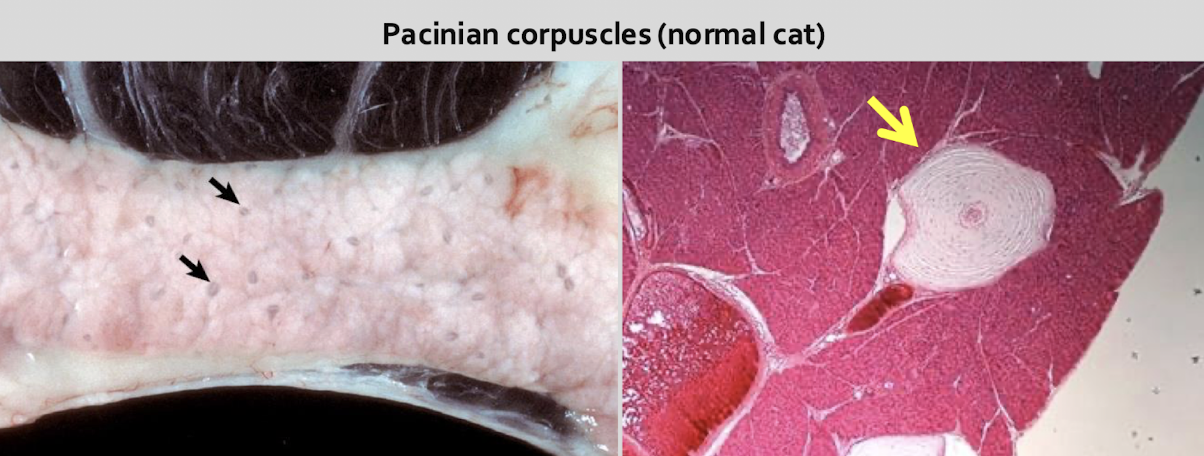
What is an incidental finding specific to cats?
pacinian corpuscles in interlobular connective tissue & mesentery → specialized nerve ending (mechanoreceptor)
What is autolysis?
very rapid, in situ enzymes postmortem, gut bacteria → tissue pink → dark red → green (hemolysis)
What demographic of cats is affected by stromal fat cell infiltration?
obese cats