Chemistry CH6 Study Guide
1/135
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
136 Terms
What is the Buckminister Fuller made of, and what is it sometimes called?
geodesic domes, buckeyball (looks like the EPCOT ball)

What must geometric isomers have?
double bond
What are unsaturated hydrocarbons?
not all carbon atoms have 4 single covalent bonds
What is one common use of an alkene?
ethene, plant hormone that triggers ripening
What is the ending for an alkyne with one triple bond?
-yne (ex. hexyne)
What is an aromatic hydrocarbon?
6-membered carbon rings and delocalized electrons
What halogen atoms can be in alykyl halides?
F, Cl, Br, I
What is the general formula for alkyl halides?
R-X ("r" being the rest of the formula and "x" being the halide/s)
What are aldehydes?
organic compounds where carbonyl group is attached to a carbon at the END of a carbon chain
What is a carbonyl group?
double bonded O
What is the simplest aldehyde?
formaldehyde (preservative)
What is the ending for a carboxylic acid with ONE carboxyl group?
-oic acid
When are amines common?
breakdown of proteins in animal cells
Where can you find carbon?
all living things and fuels like coal and gasoline
What pattern is common with single bonds?
zig zags
What shape is common with single bonds?
tetrahedral
Where are hybrid orbitals in single bonds?
sp3
Where are hybrid orbitals in double bonds?
sp2
With double bonds, where will atoms be?
same plane
Where are hybrid orbitals in triple bonds?
sp
What is allotrophy?
existence of 2 or more different molecular forms of an element exist in the same physical state
What is an example of an allotrophy?
O2 is oxygen, O3 is ozone
What are some examples of carbon allotrophies? (name 2)
diamond, graphite
What are fullerenes?
dark solids made of spherically networked carbon cages
What are carbon nanotubes used for?
circuits, fiberoptics, electricity
What are organic compounds?
covalently bonded compounds w/ carbon, excluding carbonates and oxides
What is catenation?
covalent binding of an element to itself to form chains or rings (ex. 2 carbons together, or hundreds)
What are hydrocarbons?
compounds w/ only C and H
Hydorcarbons are some of the most ___ organic compounds and are referred to as ___.
simple, backbone
What are isomers?
compounds w/ same molecular formula but different structures and properties (in the picture, both molecules are C4H10, but bond orders are different)
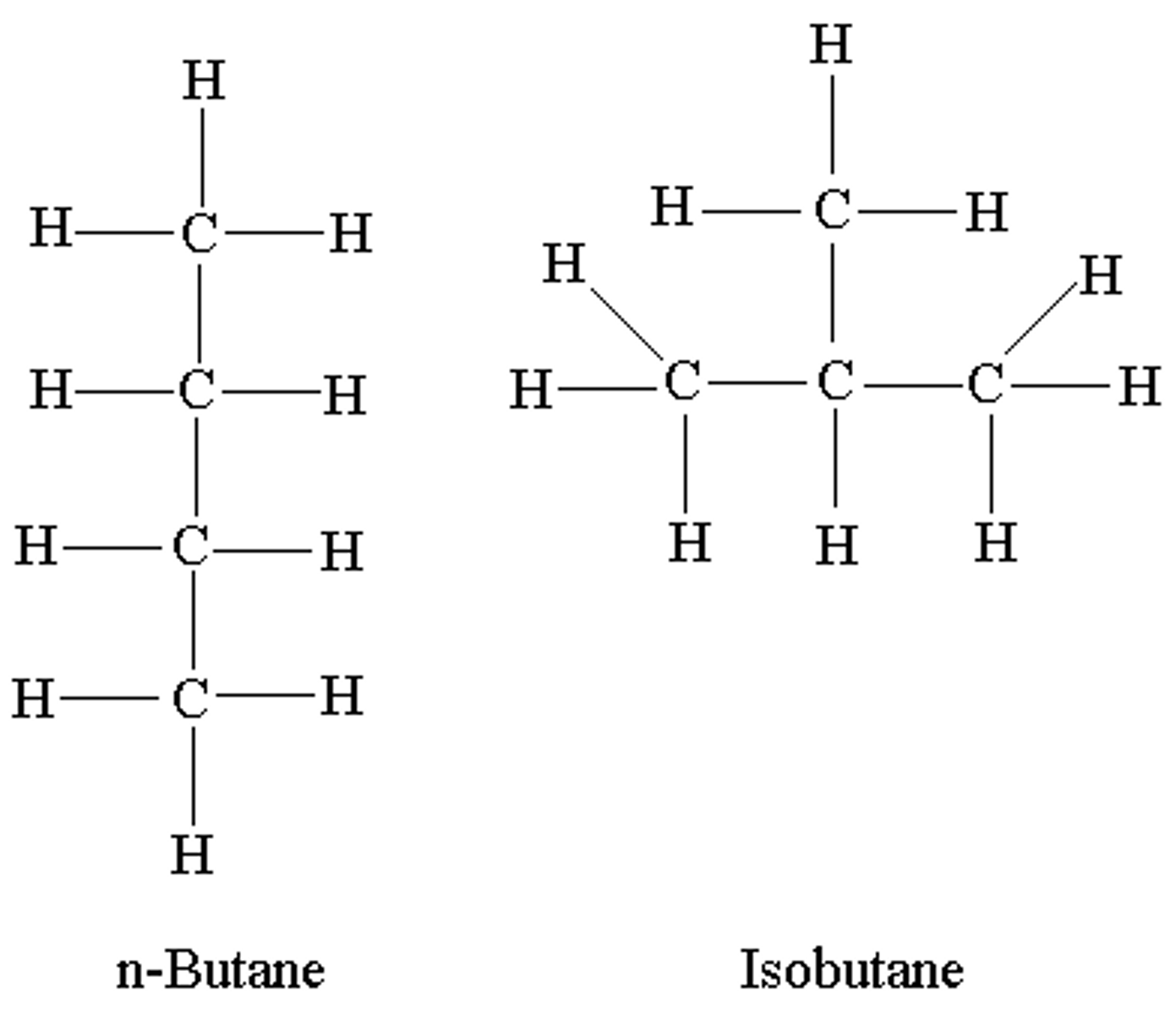
What are structural isomers?
isomers whose atoms are bonded together in different orders
What are geometric isomers?
isomers where the orders of atom bonds are the same, but ARRANGEMENT is different
Double bonds prevent what?
rotation
What are the two types of geometric isomers?
cis and trans
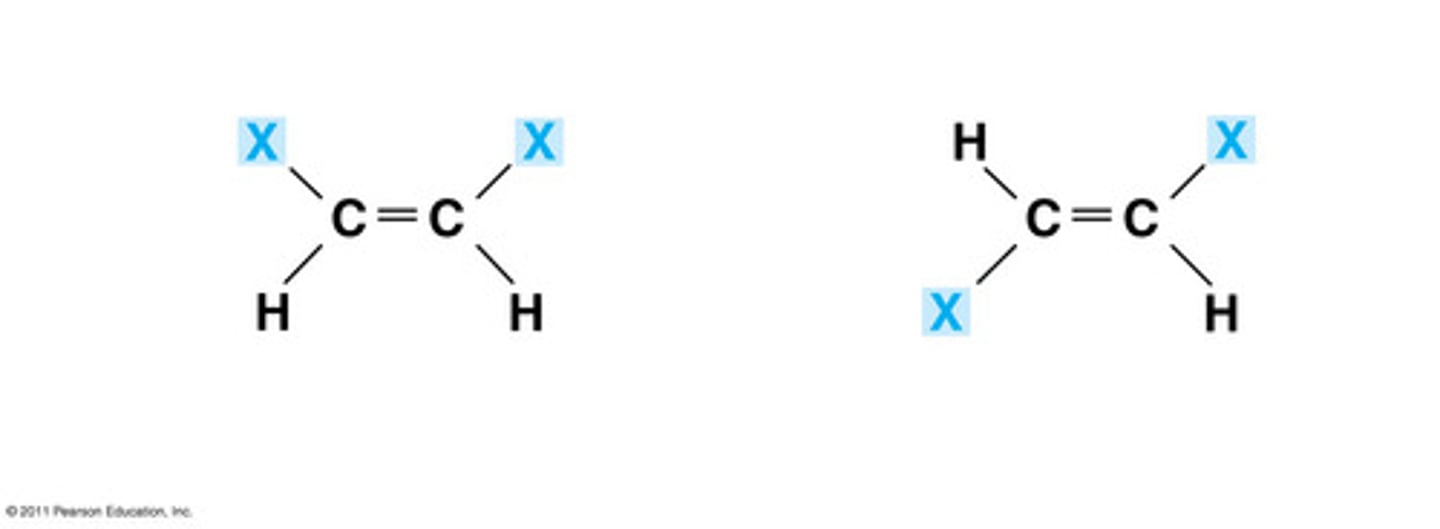
Be able to draw geometric and structural isomers
no prob
What are saturated hydrocarbons?
each carbon atom forms from single bonds with other atoms (so the carbons only have single bonds with each other, and then as many hydrogens as possible)
What are alkanes?
hydrocarbons with only single bonds
Know are your prefixes and word roots, I'm not putting them all here
fine
What is the general formula for ALKANES?
CnH2n+2 (for every C, one H on top, one on the bottom, and then the extra two on the ends)
What is the formula for an alkane with 15 carbon atoms?
C15H32 (CnH2n+2)
What is the formula for an alkane with 22 carbon atoms?
C22H46 (CnH2n+2)
Adjacent members differ by a constant unit in a _____ series.
homologous (so if every unit varies by two or something)
Know how to do the condensed/super condensed/line formats
alrighty
What are cycloalkanes?
alkanes in which the carbon atoms are arranged in a ring, or cyclic, structure
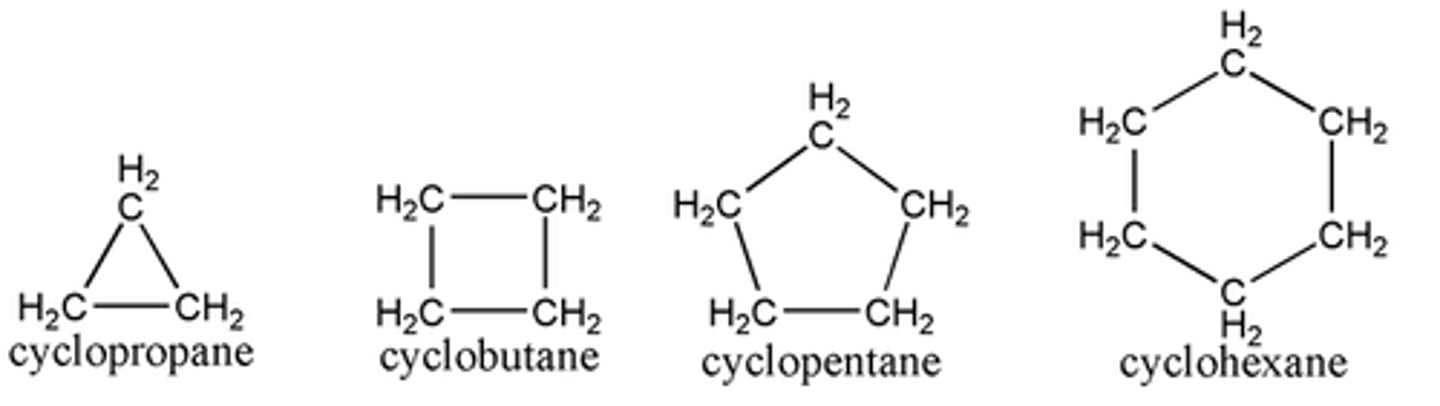
What is the general formula for CYCLOALKANES?
CnH2n (each C has one H on top and one H on bottom, all other bonds are between the C's in a ring)
What is the ending used after the wood root for ALKANES?
-ane (ex. hexane)
Name this molecule: CH3-CH2-CH2-CH2-CH3
pentane
Branched alkane chains are referred to as what?
alkyl groups
Be able to draw out molecules when given the names and name molecules when given the drawing, I can't really put those here
k
C-H bonds are NEARLY ___. The only forces of attraction are ___ Dispersion Forces.
nonpolar, London
What kinds of alkanes are gases, smaller or longer chains?
smaller
What is natural gas made up of?
mainly methane (like 98%), also butane
What is petroleum made up of?
hydrocarbons with varying compositions
What is fractional distillation?
components of a mixture are separated based on boiling point by condensation of vapor in a fractionating column
Where is fractional distillation used?
oil refining
What is combustion?
reaction involving the burning of a substance, often an alkane hydrocarbon
Pure alkanes ___ easily.
burn
What is the basic equation for combustion?
CH4 + 2O2 --> CO2 + 2H2O
What is an octane rating?
measure of fuel's burning efficiency
Octane ratings depend on the ___ of a hydrocarbon. The more branches you have, the ___ the rating.
structure, higher
What are alkenes?
hydrocarbons w/ double bonds
Where is the general formula for ALKENES?
CnH2n (lost the 2 H's from alkanes when C comes together to form a double bond)
What is the ending for an alkene with ONE double bond?
-ene (ex. hexene)
What is the ending for an alkene with TWO double bonds? Three?
-adiene, -atriene (ex. hexadiene, hexatriene)
Name this: CH3-CH2-CH2-CH2-CH=CH2
hexene (no position number is needed here. The double bond is on the end, so it automatically gets position #1. That being said, if you really want to put a position number, you won't get counted off
What is an alkyne?
hydrocarbon with a triple bond
What is the general formula for alkynes with 1 triple bond?
CnH2n-2 (if you ever forget a general formula, just draw out a simple formula like CH---C-CH3 and figure it out from there)
What is the ending for an alkyne with two triple bonds? Three?
-adiyne, -atriyne (ex. hexadiyne, hexatriyne)
Name (the 3 dashes mean it's a triple bond): CH---C-C---C-CH3
1,3-pentadiyne
What is one common use of an alkyne?
ethyne torches, used for welding
know what benzene is. I don't really know how to make a question about it
okie dokie
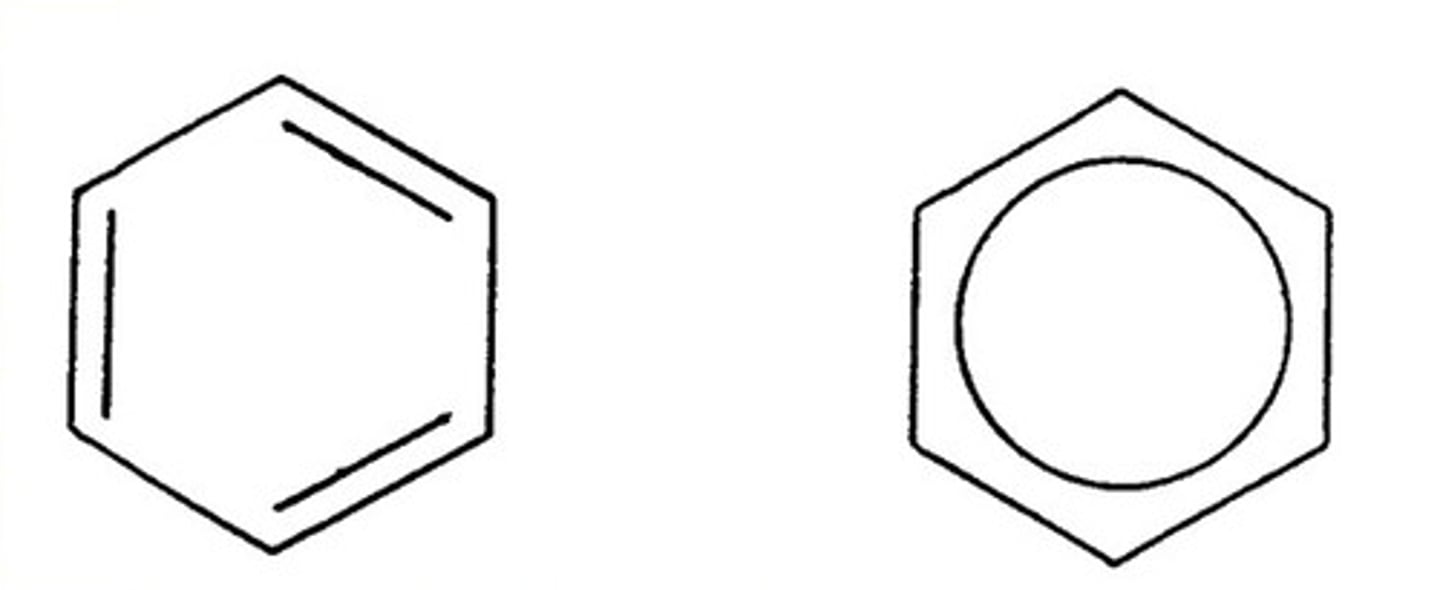
What does heterocyclic mean?
in a benzene or cycloalkane, a carbon atom in the ring is replaced by another element
What is it called when a benzene acts as a branch?
benzyl/phenyl
Are aromatic hydrocarbons stable? What does this mean when they are alone?
yes, less active
Aromatic hydrocarbons are ___ that produce toxic molecules when your body tries to break them down.
carcinogens
Where is benzene found?
gasoline (it can be absorbed through your skin)
What is a functional group?
atom/group of atoms responsible for specific properties of an organic compound
The location of a functional group is usually the site of ___ ___.
chemical reactivity
All molecules with the same functional group will have similar ___.
properties
What are alcohols? (the CHEMICAL definition not the sarcastic one I know some of you are thinking about)
organic compounds with 1 or more hydroxyl groups
What is the general formula for alcohols?
R-OH
What is a hydroxyl group?
OH
What is the ending for an alcohol with ONE hydroxyl group?
-ol (ex. hexanol)
What is the ending for an alcohol with two hydroxyl groups? Three?
DO NOT DROP ANY LETTERS, -diol, -triol (ex. hexanediol, hexanetriol)
If an OH is a branch, what is it called?
hydroxy (ex. 3-hydroxy-hexene)
Alcohols' boiling points are generally ___ than alkanes.
higher
Why are alcohols' boiling points generally higher than alkanes?
hydrogen bonding is stronger than alkanes' London dispersion forces and are harder to break
Smaller alcohols are ___ in water.
soluble
Alcohols can be used to make certain products what?
moist (it retains moisture. sorry to whoever is bugged by this word ¯\_(ツ)_/¯)
All alcohols are ___ to some extent.
poisonous
What are alkyl halides?
organic compounds with halogen atoms are substituted for hydrogens
What are the halogen stems used as prefixes for naming alkyl halides?
fluoro, chloro, bromo, iodo
Name: CH2(F)CH2CH2(F)
1,3-difluoropropane
What is one use of alkyl halides?
CFC's (chloro-fluoro-carbons) in foams, aerosols
What is one risk of using alkyl halides?
destroys ozone
What are ethers?
organic compounds with 2 hydrocarbon groups bonded to a single oxygen
What is the general formula for ethers?
R-O-R'
Name: CH3-CH2-O-CH2-CH3
diethyl ether
What is the ending for an ether?
ether (just add either. ex. cyclobutyl methyl ether. separate words.)
Name: C3H7-O-C3H7
dipropyl ether