Organic Chem 1 Reactions
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
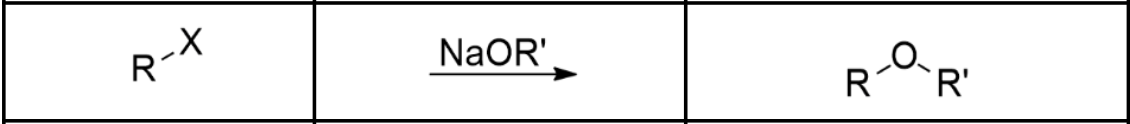
Alkyl Halide —> Ether
1º Alkyl Halide with the salt of an alkoxide reagent (Williamson Ether Synthesis via Sn2)
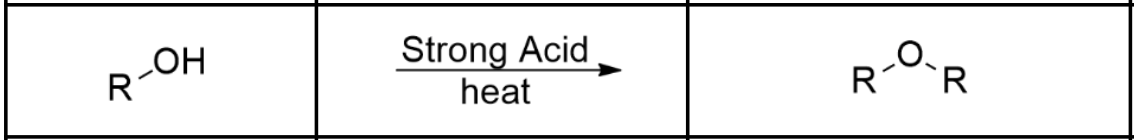
Alcohol —> Ether
Presence of a strong acid (usu. H2SO4 or H3PO4) and heat (Condensation Reaction, via Sn1/Sn2)
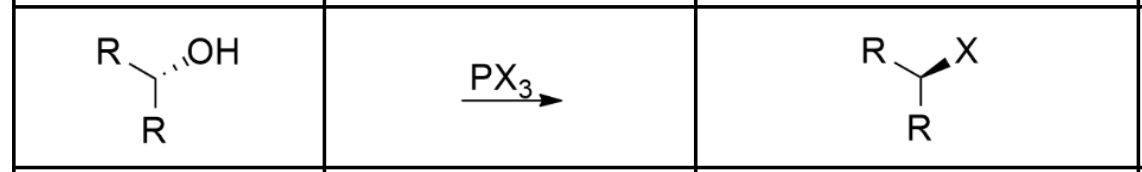
Alcohol to Alkyl Halide (via Sn2)
PX3: PBr3 or PCl3
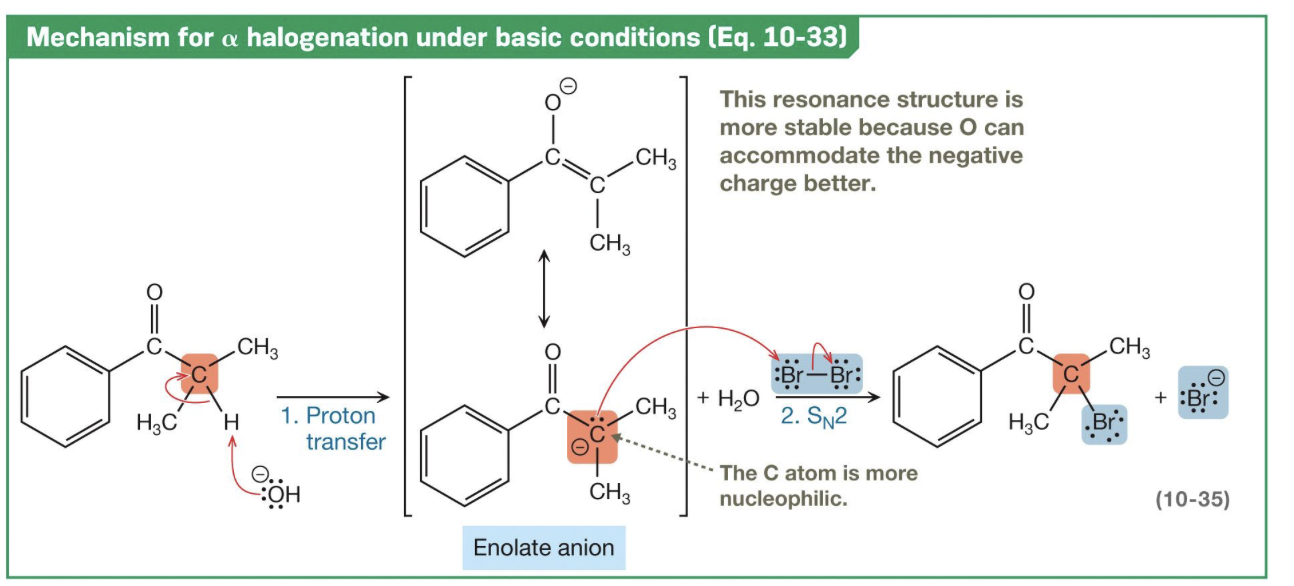
⍺-carbon halogenation of a ketone or aldehyde
I2, Br2, Cl2 — Forms an enol (acid)/enolate (alkaline) intermediate. Can occur in either basic or acidic conditions. ** If product is chiral, produces racemic stereoisomers.
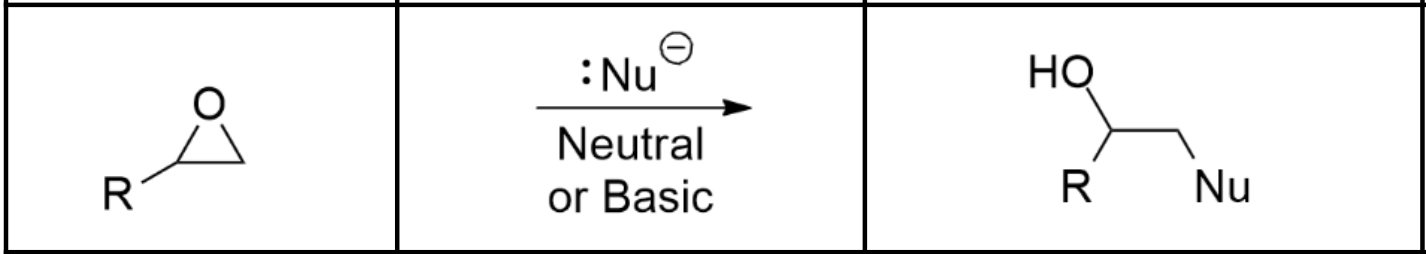
Alcohol synthesis via ether under neutral or basic conditions
Nu- (via Sn2) ex. NaCl or NaOCH3. Note that (under basic and neutral conditions) the Nu- only attacks the least substituted carbon opposite of the O.
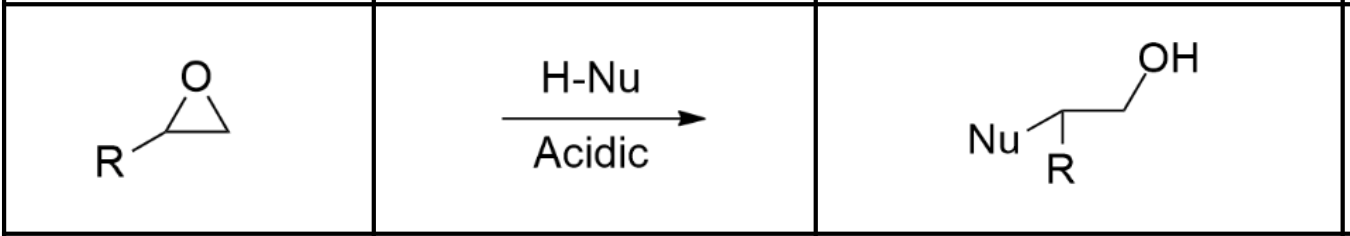
Alcohol synthesis via ether under acidic conditions
Use an acid (such as HBr). Nu- will react with the MOST substituted carbon, since the first-step proton transfer creates a positive charge on the O that is mitigated by partially breaking bonds with the bonded carbons. The more substituted carbon has electron donating groups, resulting in an even weaker C-O bond.
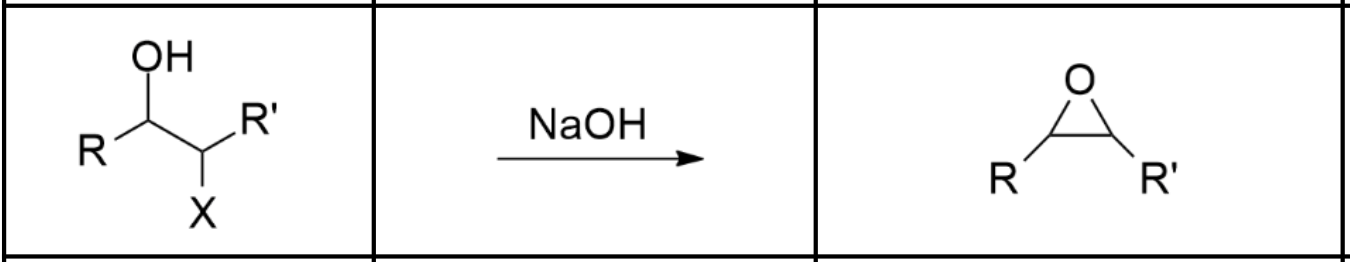
Halohydrin —> Epoxide
Requires a base (ex. NaOH). Occurs via Sn2.
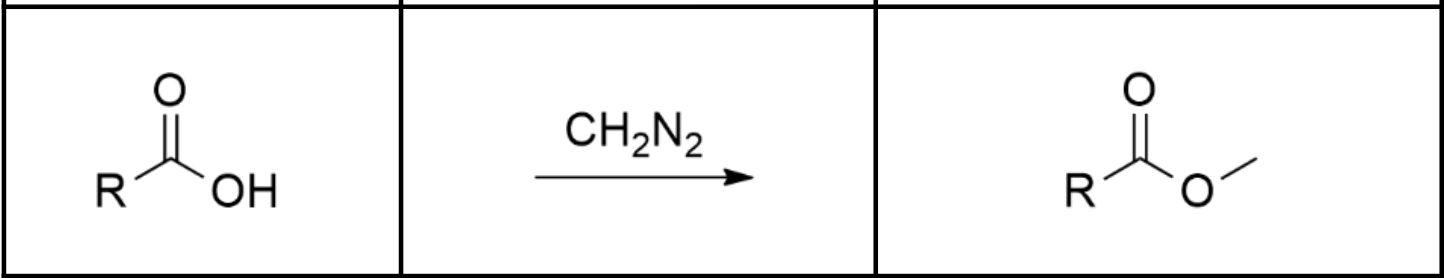
Synthesizing a methyl ester by treating a carboxylic acid with ___
diazomethane: CH2N2. Often in a neutral solvent like diethyl ether.
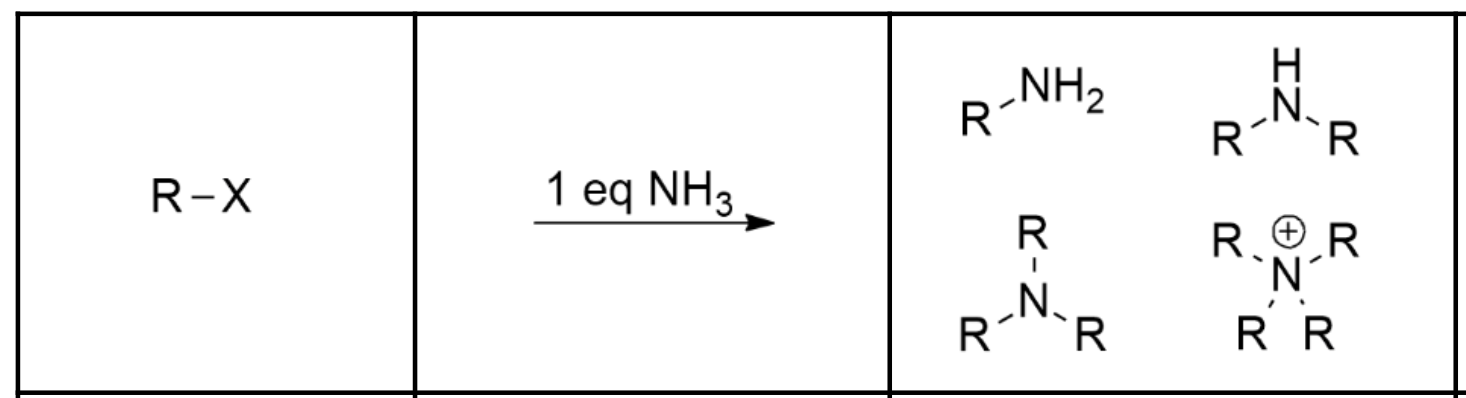
Synthesis of 1-4º alkyl halides:
Use an alkyl halide. The NH3 will act as a nucleophile as well as a base, and will proceed via Sn2 —> Proton Transfer to yield a primary amine.
From there, the primary amine will act as the nucleophile on the alkyl halide to produce a secondary amine… and again and again until quartenary ammonium ion.
Because of this, alkylation is NOT a very efficient way of synthesizing amines from alkyl halides (esp. 1º amines).
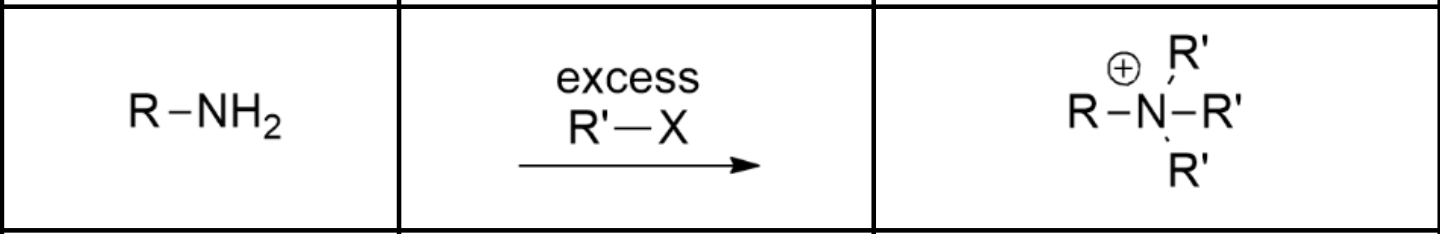
Synthesis of quaternary ammonium ion:
Ammonia with an excess of an alkyl halide.
Rather than having the alkyl halides react with ammonia like when we try to synth a 1º amine via alkylation, we WANT polyalkylation to occur, meaning we add an excess of alkyl halide compared to our ammonia.
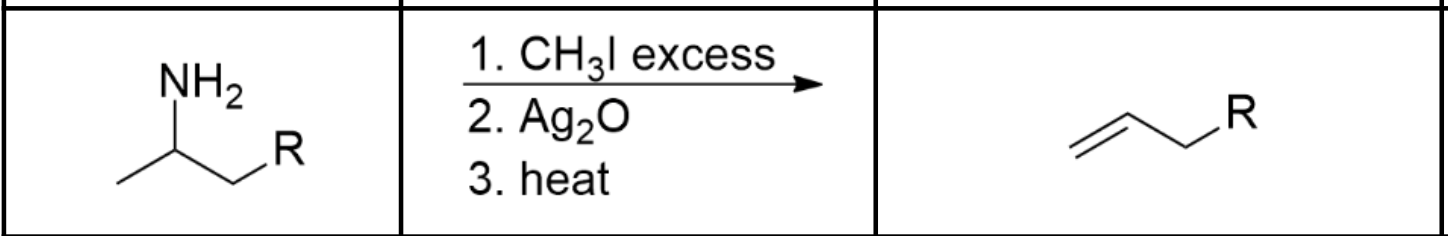
Formation of an alkene and a tertiary amine
Use excess CH3I (alkyl halide), which will proceed via Sn2 and polyalkylate the amine until it’s a quaternary ammonium ion. Then add Ag2O, which will cause the I- in solution to precipitate and replace it with OH-. The OH- now acts as a base and proceeds via E2 on the least substituted ß-hydrogen, resulting in an anti-zaitsev product.
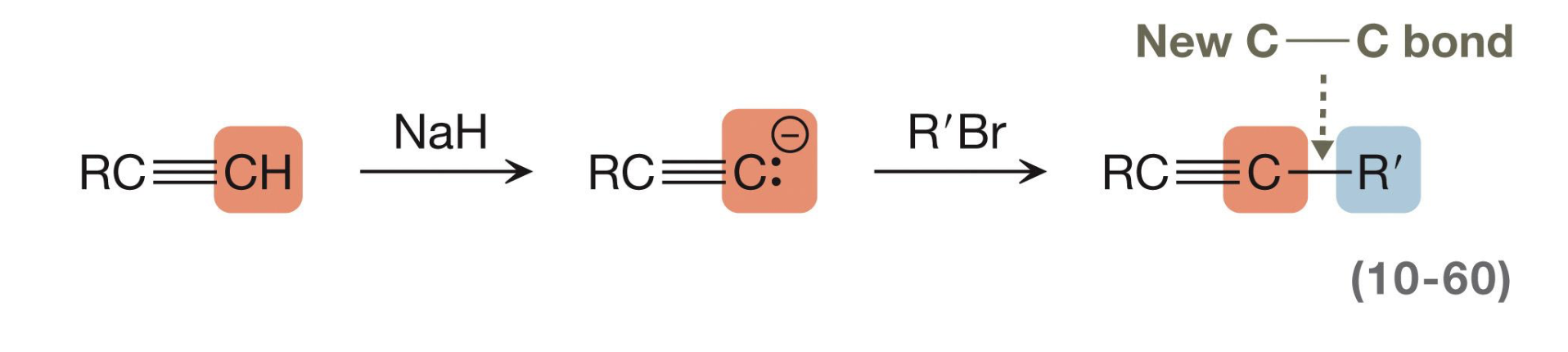
Alkyne extension
First, remove the hydrogen via proton transfer using NaH. From there, add an alkyl halide which will proceed via Sn2 to yield a longer alkyne chain.