Chp. 1-4
1/221
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
222 Terms
Homeostasis
steady state of body systems that living organisms maintain
Catabolism
the metabolic process of breaking down complex molecules into simpler ones, releasing energy in the process.
Anabolism
the metabolic process of building up complex molecules from simpler ones, usually requiring energy.
Your body can assemble, by utilizing energy, the complex chemicals it needs by combining small molecules derived from the foods you eat
ATP
Stores and releases energy
composed of a ribose sugar, an adenine base, and three phosphate groups.
releases free energy when its phosphate bonds are broken, and thus supplies ready energy to the cell. More is produced in the presence of oxygen (O2) than in pathways that do not use oxygen.
ATP process
The cell stores energy in the synthesis (anabolism) of ATP, then moves the ATP molecules to the location where energy is needed to fuel cellular activities. Then the ATP is broken down (catabolism) and a controlled amount of energy is released, which is used by the cell to perform a particular job.
cell
smallest independently functioning unit of a living organism.
organelles
specialized structures within a cell that perform distinct functions, such as energy production and protein synthesis.
tissue
a group of many similar cells (though sometimes composed of a few related types) that work together to perform a specific function.
organ
an anatomically distinct structure of the body composed of two or more tissue types. Each one performs one or more specific physiological functions.
organ system
a group of organs that work together to perform major functions or meet physiological needs of the body.
Differentation
where unspecialized cells become specialized in structure and function to perform certain tasks in the body
Set point
the physiological value around which the normal range fluctuates.
Normal range
the restricted set of values that is optimally healthful and stable.
ex: body temperature 98.6
negative feedback
a mechanism that reverses a deviation from the set point.
maintains body parameters within their normal range.
effector
is the component in a feedback system that causes a change to reverse the situation and return the value to the normal range.
positive feedback
intensifies a change in the body’s physiological condition rather than reversing it. A deviation from the normal range results in more change, and the system moves farther away from the normal range.
normal only when there is a definite end point.
Ex: pooping and childbirth
integumentary system
encloses internal body structures
site of many sensory receptors
hair, skin, nails
skeletal system
supports the body
enables movement
cartilage, bones, joints
muscular system
enables movement
helps maintain body temperature
skeletal muscles, tendons
nervous system
detects and processes sensory information
activates bodily responses
brain, spinal cord, peripheral nerves
endocrine system
secretes hormones
regulates bodily processes
pituitary gland, thyroid gland, pancreas, adrenal glands, testes, ovaries
cardiovascular system
delivers oxygen and nutrients to tissues
equalizes temperature in the body
heart, blood vessels
lymphatic system
returms fluid to blood
defends against pathogens
thymus, lymph nodes, spleen, lymphatic vessels
respiratory system
removes carbon dioxide from the body
delivers oxygen to blood
nasal passage, trachea, lungs
digestive system
processes food for use by the body
removes wastes from undigested food
stomach, liver, gall bladder, large intestine, small intestine
urinary system
controls water balance in the body
removes waste from blood and excretes them
kidneys, urinary bladder
organism
a living being that has a cellular structure and that can independently perform all physiologic functions necessary for life.
Metabolsim
the sum of all anabolic and catabolic reactions that take place in the body
prone
face-down position
supine
face up position
sagittal plane
is the plane that divides the body or an organ vertically into right and left sides. If this vertical plane runs directly down the middle of the body, it is called the midsagittal or median plane. If it divides the body into unequal right and left sides, it is called a parasagittal plane or less commonly a longitudinal section.
frontal (coronal) plane
the plane that divides the body or an organ into an anterior (front) portion and a posterior (rear) portion.
transverse plane
the plane that divides the body or organ horizontally into upper and lower portions. plane that produces cross sections
oblique plane
intersects the body at an angle and is not parallel or perpendicular to any of the other three commonly used planes.
dorsal (posterior) cavity
cranial and spinal cavaties
Ventral (anterior) cavity
thoracic and abdominopelvic cavaties
pleura
the serous membrane that encloses the pleural cavity; the pleural cavity surrounds the lungs
serous membrane
(also referred to a serosa) is one of the thin membranes that cover the walls and organs in the thoracic and abdominopelvic cavities.
parietal layer
lines the walls of the body cavity (pariet- refers to a cavity wall).
visceral layer
covers the organs (the viscera).
element
a pure substance that is distinguished from all other matter by the fact that it cannot be created or broken down by ordinary chemical means.
Together, just four elements make up more than 95 percent of the body’s mass. These include
oxygen, carbon, hydrogen, and nitrogen
compound
a substance composed of two or more elements joined by chemical bonds.
atom
the smallest quantity of an element that retains the unique properties of that element
typically has the same number of positively charged protons and negatively charged electrons.
proton
number determines the element
electron
The number of negatively-charged electrons that “spin” around the nucleus at close to the speed of light equals the number of protons.
atomic number
the number of protons (and electrons) in the nucleus of the atom, identifies the element.
mass number
the sum of the number of protons and neutrons in its nucleus.
isotope
one of the different forms of an element, distinguished from one another by different numbers of neutrons.
electron shell
a layer of electrons that encircle the nucleus at a distinct energy level.
valence shell
atom’s outermost electron shell.
atoms (except hydrogen andf helium) and single electron shells and are most stable when their valence shells are full (8 electrons)
bond
is a weak or strong electrical attraction that holds atoms in the same vicinity.
molecule
A more or less stable grouping of two or more atoms held together by chemical bonds
compound
When a molecule is made up of two or more atoms of different elements
H2O
ion
An atom that has an electrical charge; whether positive (cation) or negative (anion)
ionic bond
formed by the attraction of ions of opposite charges
table salt
covalent bond
share electrons in a mutually stabilizing relationship. Like next-door neighbors whose kids hang out first at one home and then at the other, the atoms do not lose or gain electrons permanently
polar molecule
a molecule that contains regions that have opposite electrical charges. occur when atoms share electrons unequally, in polar covalent bonds.
hydrogen bond
is formed when a weakly positive hydrogen atom already bonded to one electronegative atom (for example, the oxygen in the water molecule) is attracted to another electronegative atom from another molecule. In other words, hydrogen bonds always include hydrogen that is already part of a polar molecule.
Mechanical energy
When you lift a brick into place on a wall, your muscles provide the mechanical energy that moves the brick.
Radiant energy
The body uses the ultraviolet energy of sunlight to convert a compound in skin cells to vitamin D, which is essential to human functioning. The human eye evolved to see the wavelengths that comprise the colors of the rainbow, from red to violet, so that range in the spectrum is called “visible light.”
Electrical energy
supplied by electrolytes in cells and body fluids, contributes to the voltage changes that help transmit impulses in nerve and muscle cells.
synthesis reaction
a chemical reaction that results in the synthesis (joining) of components that were formerly separate
A + B —> AB
decomposition reaction
chemical reaction that breaks down or “de-composes” something larger into its constituent parts
AB —> A + B
exchange reaction
chemical reaction in which both synthesis and decomposition occur, chemical bonds are both formed and broken, and chemical energy is absorbed, stored, and released
A + BC —> AB + C // AB + CD —> AC + BD
catalyst
a substance that increases the rate of a chemical reaction without itself undergoing any change.
enzyme
a catalyst composed of protein or ribonucleic acid (RNA)
lower the activation energy in a chemical reaction
inorganic compound
is a substance that does not contain both carbon and hydrogen
organic compound
a substance that contains both carbon and hydrogen
solution
consists of a solvent that dissolves a substance called a solute.
homogenous (molecules are distributed evenly throughout the solution)
colloid
a mixture that is somewhat like a heavy solution. Consists of tiny clumps of molecules large enough to make the liquid mixture opaque (because the particles are large enough to scatter light).
milk and cream
suspension
is a liquid mixture in which a heavier substance is suspended temporarily in a liquid, but over time, settles out.
Acid
Substance that releases hydrogen ions (H+) in solution
Base
Substance that releases hydroxyl ions (OH–) in solution, or one that accepts H+ already present in solution
Buffer
A solution of a weak acid and its conjugate base. Can neutralize small amounts of acids or bases in body fluids.
Functional group
A group of atoms linked by strong covalent bonds and tending to function in chemical reactions as a single unit.
Ttightly knit “cliques” whose members are unlikely to be parted.
Functional groups important in human physiology
Hydroxyl, carboxyl, amino, methyl
Carbohydrate
molecule composed of carbon, hydrogen, and oxygen
saccharides (sugars)
lipid
one of a highly diverse group of compounds made up mostly of hydrocarbons
hydrophobic compounds that provide body fuel and are important components of many biological compounds.
triglyceride
one of the most common dietary lipid groups, and the type found most abundantly in body tissues. This compound, which is commonly referred to as a fat, is formed from the synthesis of two types of molecules
major fuel source for the body
protein
is an organic molecule composed of amino acids linked by peptide bonds.
found in the keratin in the epidermis of skin that protects underlying tissues, the collagen found in the dermis of skin, in bones, and in the meninges that cover the brain and spinal cord.
components of many of the body’s functional chemicals, including digestive enzymes in the digestive tract, antibodies, the neurotransmitters that neurons use to communicate with other cells, and the peptide-based hormones that regulate certain body functions (for instance, growth hormone).
RNA
ribose-containing nucleotide that helps manifest the genetic code as protein.
tissue
a group of cells found together in the body.
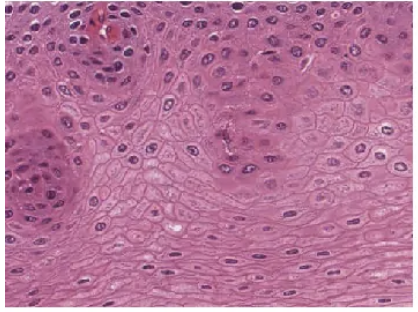
epithelial tissue
the sheets of cells that cover exterior surfaces of the body, line internal cavities and passageways, and form certain glands.
lining of GI tract organs and other hollow organs
skin surface (epidermis)
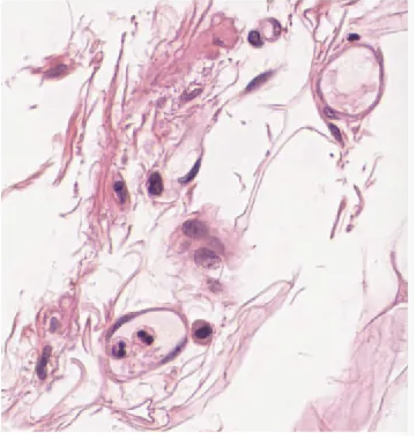
connective tissue
implies, binds the cells and organs of the body together and functions in the protection, support, and integration of all parts of the body.
fat and other soft padding tissue
bone
tendon
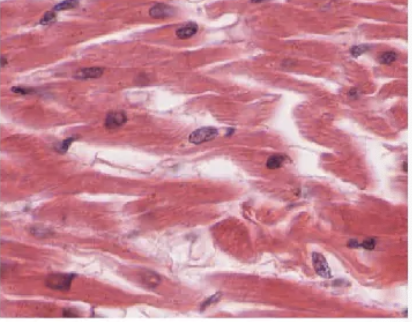
muscle tissue
excitable, responding to stimulation and contracting to provide movement, and occurs as three major types: skeletal (voluntary) muscle, smooth muscle, and cardiac muscle in the heart.
cardiac muscles
smooth muscle
skeletal muscle
nervous tissue
also excitable, allowing the propagation of electrochemical signals in the form of nerve impulses that communicate between different regions of the body
brain, spinal cord, nerves
ectoderm
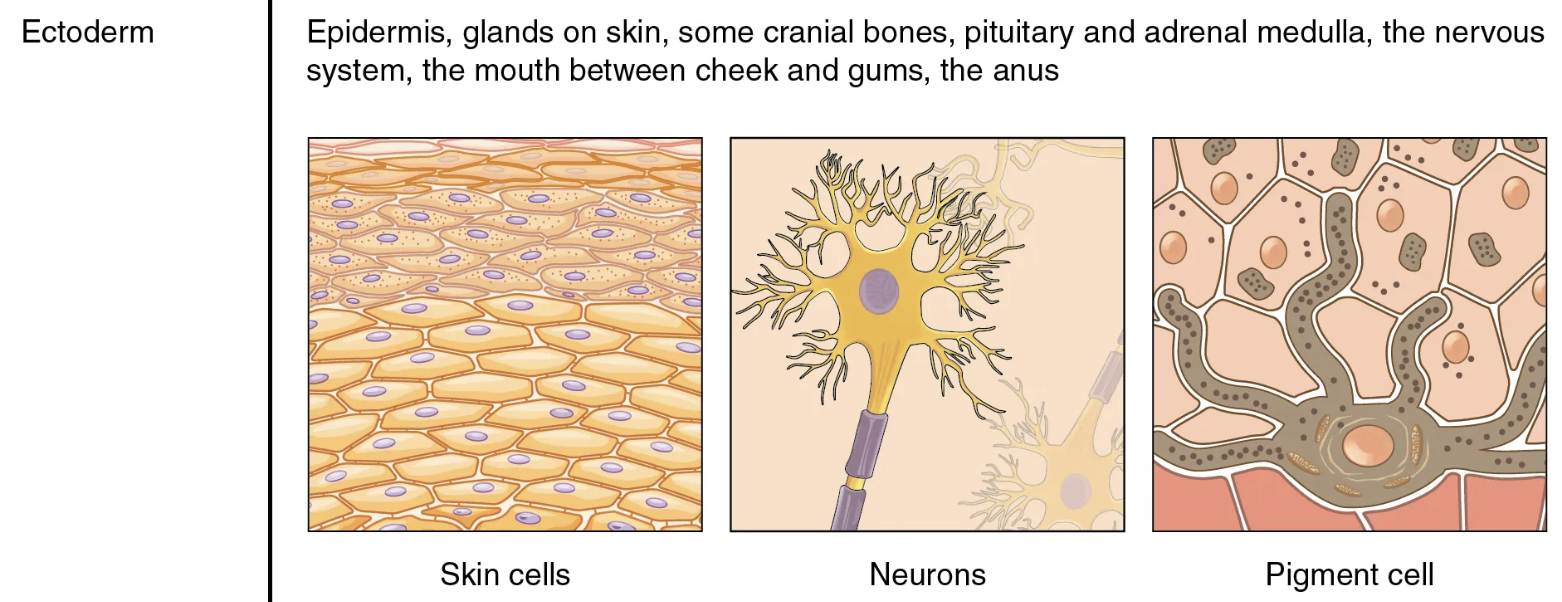
endoderm
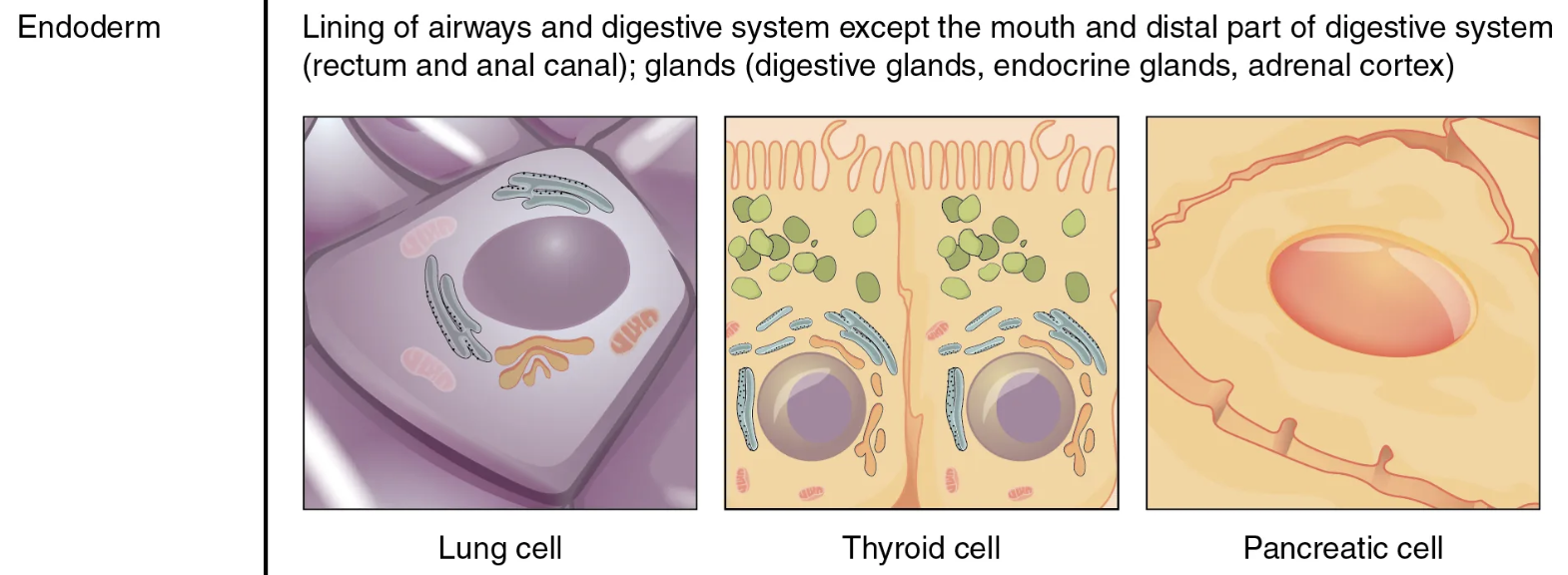
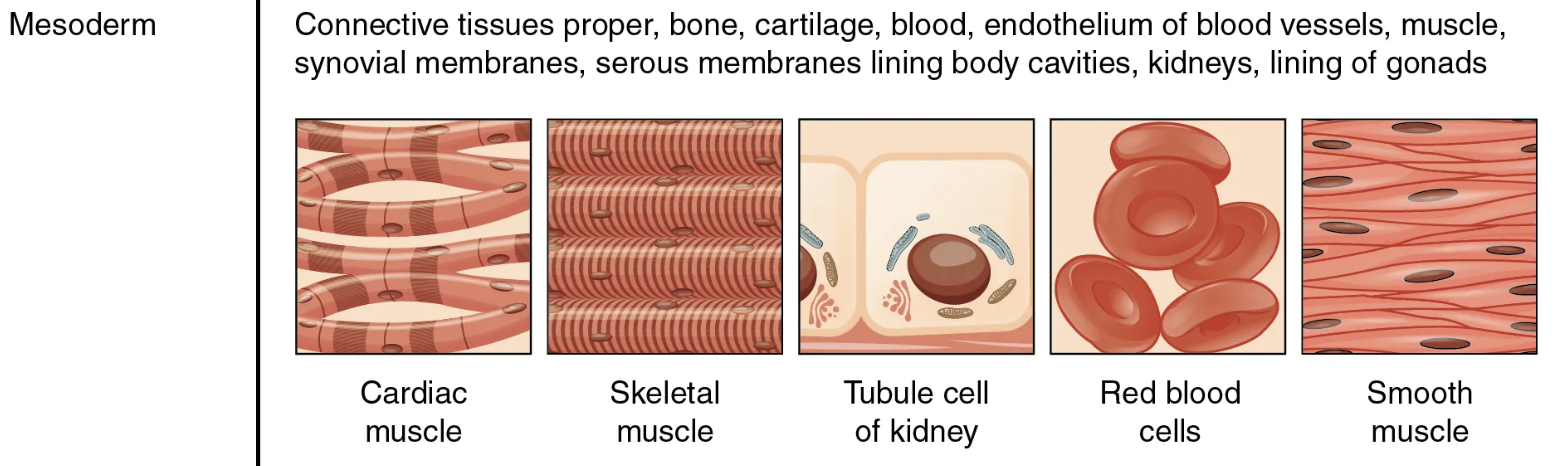
mesoderm
tissue membrane
a thin layer or sheet of cells that covers the outside of the body (for example, skin), the organs (for example, pericardium), internal passageways that lead to the exterior of the body (for example, mucosa of stomach), and the lining of the moveable joint cavities. There are two basic types of tissue membranes: connective tissue and epithelial membranes
connective tissue membrane
formed solely from connective tissue
These membranes encapsulate organs, such as the kidneys, and line our movable joints.
synovial membrane
type of connective tissue membrane that lines the cavity of a freely movable joint.
For example, they surround the joints of the shoulder, elbow, and knee.
Intracellular fluid (ICF)
the fluid interior of the cell.
Extracellular fluid (ECF)
the fluid environment outside the enclosure of the cell membrane.
Interstitial fluid
the term given to extracellular fluid not contained within blood vessels.
integral protein
protein that is embedded in the membrane.
channel protein
an example of an integral protein that selectively allows particular materials, such as certain ions, to pass into or out of the cell.
receptor
a type of recognition protein that can selectively bind a specific molecule outside the cell, and this binding induces a chemical reaction within the cell.
ligand
the specific molecule that binds to and activates a receptor.