Chemistry IV
1/52
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
Law of Conservation of Mass
In a chemical reaction, the total mass of
the substances before and after the
reaction remains the same.
It can neither be created nor destroyed but it can rearrange.
Reactants
The substances which undergoes chemical reactions.
Products
The substances which are the end products of a chemical reaction
Activation Energy
The minimum amount of energy that is required to activate molecules or atoms so that they can undergo chemical transformation.
React with
In chemical reaction or equation, what does + read as?
Yield / Produces
In chemical reaction or equation, what does → read as?
Decomposition

Synthesis

Single-replacement
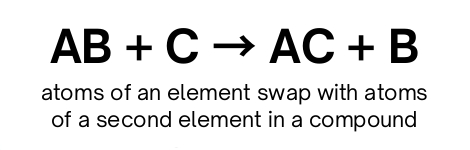
Double-replacement
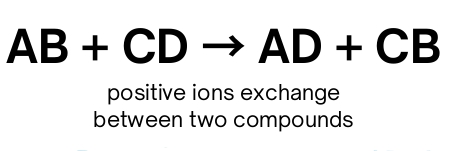
Combustion Reaction
Composed of fuel/organic compound, air, heat, and water.
e.g. CH4 + 2O2 → CO2 + 2H2O
Neutralization Reaction
Formed by an acid base.
e.g. NaOH + HClO3 → NaCl + H2O
Rate of reaction
The speed at which reactants are converted into products.
Nature of reaction
The rate of reaction highly depends on the type and ______. Few reactions are naturally faster than others while some reactions are very slow. (Fastest to slowest: Gas, liquid, solid — because it has more space/ readily avaliable)
Collision theory
According to the ______, the rate of reaction increases with the increase in the concentration of the reactants.
Law of mass action
The chemical reaction rate is directly proportional to the concentration of reactants.
Concentration of reactants
This implies that the chemical reaction rate increases with the increase in concentration and decreases with the decrease in the concentration of reactants.
1.) Collisions should be effective.
2.) The right orientation of reactant molecules towards each other.
3.) All molecules should possess a minimum amount of energy
The conditions for the collisions to form products are?
Pressure factor
Pressure increases the concentration of gases which in turn results in the increase of the rate of reaction.
Temperature
According to collision theory, a chemical reaction that takes place at a higher temperature generates more energy than a reaction at a lower temperature. This is because colliding particles will have the required activation energy at high temperature and more successful collisions will take place.
Solvent
Much soluble ____ leads to higher rate of reaction.
Electromagnetic radiation
Form of energy (lights) and its presence at the chemical reaction may increase the rate of reaction as it gives the particles of reactants more energy.
Presence of catalyst
Substance that increases the rate of the reaction without actually participating in the reaction. Providing an alternate pathway which has lower activation energy
Surface area
Exposed parts
Rate law
The ____ expresses the relationship of the rate of a reaction to the rate constant and the concentration of the reactants raised to the same powers.
Kinetics
Refers to the study of how things move and how those movements are caused by forces.
Rate = M/s
Rate law equation.
Reaction order
The ____ provides an indication of how changing the concentration of the reactant will affect the reaction's speed.
First order
If the rate doubles
Second order
If the rate quadruples
Zero order
When nothing happened to the rate
Negative 1 order
When the rate is divided into half or slows down
Overall reaction
The sum of individual order
Thermodynamics
Is an extensive scientific discipline that deals with the interconversion of heat and other forms of energy.
First law of thermodynamics
It states that energy cannot be created nor destroyed; It can only be converted from one form to another.
2nd law of thermodynamics: Entropy
States that energy naturally spreads out or becomes more disordered over time, and heat always flows from something hotter to something cooler, not the other way around— unless you use extra energy to make it happen.
Energy spreads out, and things tend to get
messier or more disorganized unless you do work to keep them ordered.
More entropy
More energy dispersal means …
Favorable
Increasing the Entropy of a system, is thermodynamically.
3rd law of thermodynamics
It says that as something gets colder and colder—closer to absolute zero—it loses all disorder (entropy) and becomes perfectly organized.
All molecular motion would stop, and the substance would have no more energy to give off.
Entropy = S(J/K)
Entropy equation
True
Entropy is not a measure of energy itself, but how energy is distributed within a system. (True or False)
Enthalpy
Internal energy of a system.
Gibbs Free Energy
is a way to tell whether a chemical reaction or physical process can happen on its own (spontaneously) under constant temperature and pressure.
Chemical energy budget
Gibbs free energy is like a “_____” that tells us if a reaction is worth doing.
can happen spontaneously
If ΔG < 0 (negative), the process…
won’t happen spontaneously
If ΔG > 0 (positive), the process…
Equilibrium
If ΔG = 0, the system is at…
Always spontaneous
If ΔG is negative it is
Never spontaneous
If ΔG is positive it is
Energetically favorable
If ΔH is negative it is
Energetically unfavorable
If ΔH is positive it is
Entropically unfavorable
If ΔS is negative it is
Entropically favorable
If ΔS is positive it is