Kinetics & Equilibrium
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
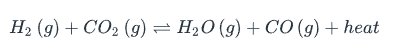
In this reaction, which of the following factors will not change the concentration of H2O in the reaction? | A) Increasing the pressure of the system B) Removing some hydrogen C) Decreasing the temperature of the system D) Adding some carbon dioxide
Increasing the pressure of the system
If carbon monoxide and oxygen can be made into carbon dioxide, what is the reverse reaction? | A) 2CO + O2 → 2CO2 B) C + O2 → CO2 C) 2CO2 → 2CO + O2 D) CO2 → CO + O2
CO2 → CO + O2
Which of the following correctly summarizes collision theory? | A) Reactants must collide with any amount force and in any orientation to form products B) Reactants must collide with enough force and proper orientation to form products C) Reactants must collide with any amount of force but proper orientation to form products D) Reactants must collide with enough force in any orientation to form products
Reactants must collide with enough force and proper orientation to form products
Reaction rate and concentration are related in a _____. | A) rate law B) reaction intermediate C) catalyst D) reaction mechanism
rate law
A _____ exists in a different physical state as the reaction it catalyzes. | A) homogeneous catalyst B) heterogeneous catalyst C) product D) inhibitor
heterogeneous catalyst
When equilibrium is established, products and reactants _____. | A) are equal to zero B) form at different rates C) are at equal concentrations D) form at the same rates
form at the same rates
In an endothermic reaction, equilibrium shifts _____ when temperature is increased. | A) in both directions B) to the left (towards the reactants) C) to the right (towards the products) D) in an unpredictable direction
to the right (towards the products)
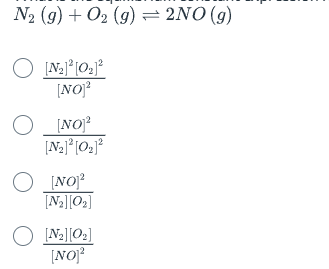
What is the equilibrium constant expression for the following reaction? | (options not provided)
B
When Keq ___ 1, reactants are favored at equilibrium. | A) > B) = C) < D) +
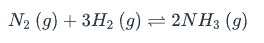
In the reaction shown, what will be the result if the volume of the reaction vessel is increased? | A) The equilibrium shifts to the left (towards the reactants) B) The equilibrium constant changes C) The equilibrium shifts to the right (towards the products) D) The equilibrium does not change
The equilibrium shifts to the left (towards the reactants)
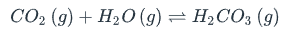
What is the effect of adding more carbon dioxide to the following equilibrium reaction? | A) The equilibrium is pushed in the direction of reactants B) CO2 concentration increases C) There is no effect D) More H2CO3 is produced
More H2CO3 is produced
What happens to a reaction at equilibrium when product is removed from the system? | A) The reaction makes more reactants B) The answer cannot be determined C) The reaction makes more products D) The reaction is unchanged
The reaction makes more products
According to the method of initial rates, if the initial concentration of a reactant doubles, and its initial rate also doubles, then that is a _____ reactant. | A) zeroth-order B) first-order C) third-order D) second-order
first-order
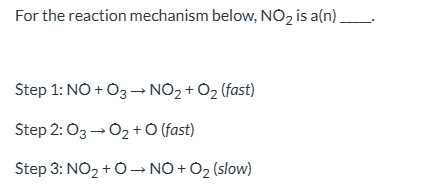
For the reaction mechanism below, NO2 is a(n) _____. | A) first-order reactant B) rate constant C) catalyst D) intermediate
intermediate
In an equilibrium reaction with a Keq of 2.1x10^4, the _____. | A) reaction is spontaneous B) products are favored C) reactants are favored D) reaction is exothermic
products are favored
_____ equilibrium is when the reactants and products are present in only one physical state. | A) Heterogeneous B) Le Chatelier’s C) Homogeneous D) Reversible
Homogeneous
A decrease in the concentration of reactants causes a shift in the reaction _____. | A) in both directions B) to the left (towards the reactants) C) to the right (towards the products) D) that is unpredictable
to the left (towards the reactants)
Which of the following statements about a catalyst is true? | A) A catalyst is not consumed during a reaction B) A catalyst decreases the concentration of reactants during a reaction C) A catalyst must always be a liquid D) A catalyst can raise the activation energy of a reaction
A catalyst is not consumed during a reaction
When equilibrium is reached, what are the relative amounts of reactants and products present? | A) There will be some reactants and also some products in the end B) Only reactants will remain in the end C) Relative amounts cannot be predicted D) Only products will remain in the end
There will be some reactants and also some products in the end
What is the rate law for the following reaction if it is first-order in A and second-order in B? A + 2B → C + D | A) rate = k[A]^2[B] B) rate = k[A][B]^2 C) rate = k[A][B] D) rate = k[A]^2[B]^2
rate = k[A][B]^2
For the reaction mechanism below, which statement is true? | A) Step 1 is the rate-determining step B) Step 2 is the rate-determining step C) Steps 1 and 2 are both rate-determining D) Step 3 is the rate-determining step
Step 3 is the rate-determining step
What is the overall reaction order for the following reaction if it is zeroth-order in A and first-order in B? A + 2B → C + D | A) Zeroth-Order B) Second-Order C) Third-Order D) First-Order
First-Order
Complex reactions must have at least _____. | A) an overall reaction order of two B) three elementary steps C) two elementary steps D) one catalyst
two elementary steps
Why does a higher concentration cause a reaction to go faster? | A) There are more collisions per second or the collisions are of greater energy B) There are more collisions per second and the collisions are of greater energy C) There are more collisions per second only D) Collisions occur with greater energy only
There are more collisions per second only
Which of the following factors does not affect the rate of the reaction? | A) The presence of an inhibitor B) The nature of the reactants C) The color of the reactants D) The presence of a catalyst
The color of the reactants
The rate of a chemical reaction normally _____. | A) decreases as temperature increases B) decreases as reactant concentration increases C) increases as temperature increases D) is slowed down by a catalyst
increases as temperature increases
![<p>In the equation below, what represents the concentrations of the products? | A) [Y] and [Z] B) (not provided) C) [W] and [X] D) (not provided)</p>](https://assets.knowt.com/user-attachments/9694d895-d9e9-402c-ae77-71dcdbe04385.png)
In the equation below, what represents the concentrations of the products? | A) [Y] and [Z] B) (not provided) C) [W] and [X] D) (not provided)
[W] and [X]
An inhibitor will _____. | A) increase the rate of reaction by increasing the temperature B) decrease the rate of reaction by binding to a catalyst C) decrease the rate of reaction by increasing the temperature D) increase the rate of reaction by binding to a catalyst
decrease the rate of reaction by binding to a catalyst
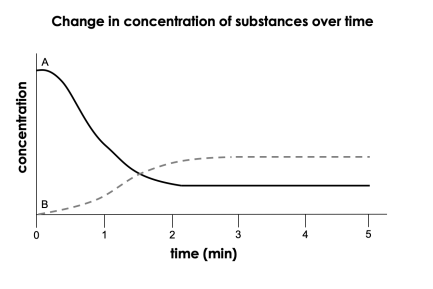
Substance A is a _____ and Substance B is a _____. | A) product; product B) reactant; reactant C) reactant; product D) product; reactant
reactant; product
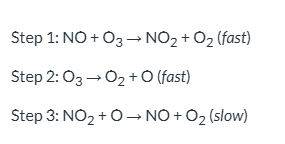
For the reaction mechanism below, NO is a(n) _____. | A) intermediate B) rate constant C) catalyst D) first-order reactant
catalyst