BIOL3060 Lecture #19 CRISPR-Cas9
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
Staggering Impact
Didn't invent CRISPR, discovered it
Life existed on Earth 4 billion years
99.9% of time past before homo sapiens arrived
Then didn’t know anything about DNA until 65 years ago (only 1 lifetime)
Learned structure of DNA, genetic info copying and storage, how to read/write DNA, and now learned how to edit DNA
Evolution of CRISPR-Cas9
Didn’t invent CRISPR, discovered it in bacteria
1987: Repetitive DNA sequences found in E. coli
Name
Clustered
Regularly
Interspaced
Short
Palindromic
Repeats
2005: Mojica proposed that CRISPR sequences serves as a bacterial immune system
2012: Doudna and Charpentier publish a paper describing how you can progran CRISPR to edit specific genes
Programmable Dual-RNA-Guided DNA Endonuclease in Adaptive Bacterial Immunity
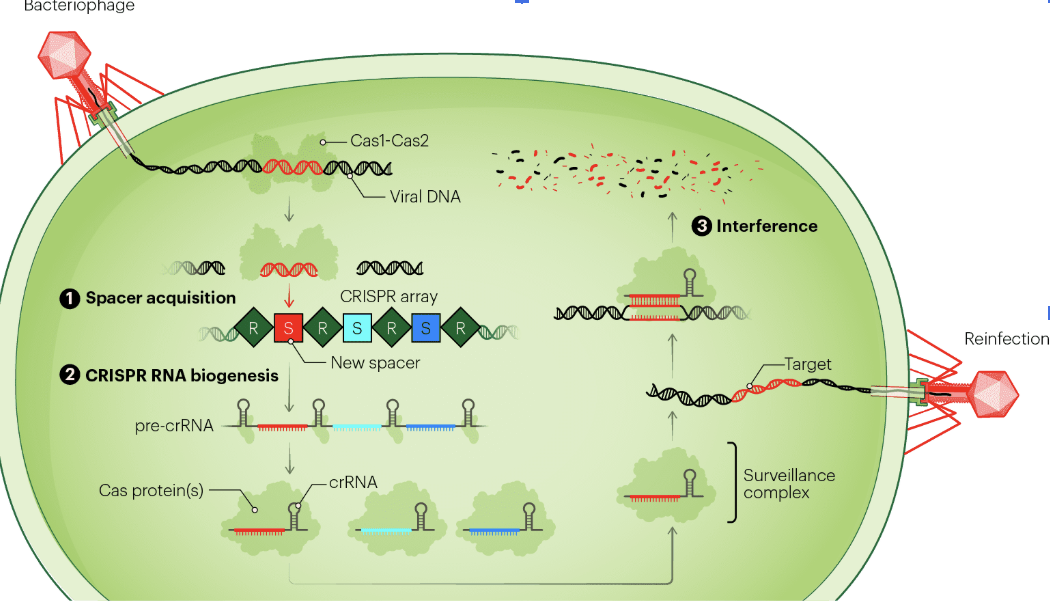
How Does CRISPR Work in Bacteria?
Bacteriophages inject genetic material into the cell, hijack cell(transcription/translation machinery)
Bacteria has to find a way to defend against viral DNA
Cas1 and Cas2 recognize viral DNA and cut it into short sequences
Viral sequences are inserted in between repeated palindromic sequences: Spacer acquisition (create molecular memory)
CRISPR RNA Biogenesis: cRNA is CRISPR RNA and trans-activating RNA, both created and come together to make structure wit- h hairpin loop: able to recognize viral sequence and bnd to CAS9 (a nonspecific DNA nuclease)
Hairpin loop structure is a surveillance complex, which moves around cell looking for the viral sequence which is recgnizes with complementary base pairing and then nuclease Cas9 can double strand break viral genome
Interference: process by which surveillance complex with Cas9 cuts DNA
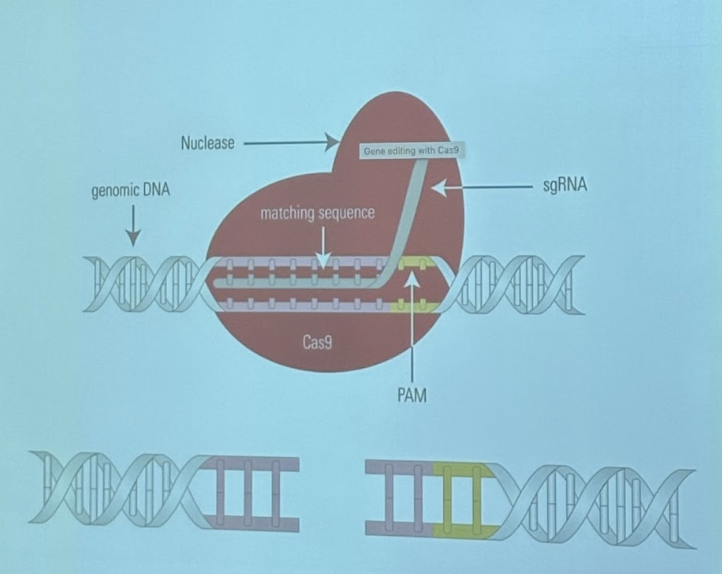
Found that you can cut whatever you want, just have to provide it with the 20 nucleotide guide sequence
Tracker RNA can be attached to CRISPR RNA, so can provide a single guide RNA
Cas9 protein creates double stranded breaks in DNA
After break is made, goal is to repair it right away

From Bacterial Immune System to Precise Molecular Scissors
Target the right gene
Bind the target gene
Cut the DNA with complexed Cas9 protein
Repair and edit the sequence

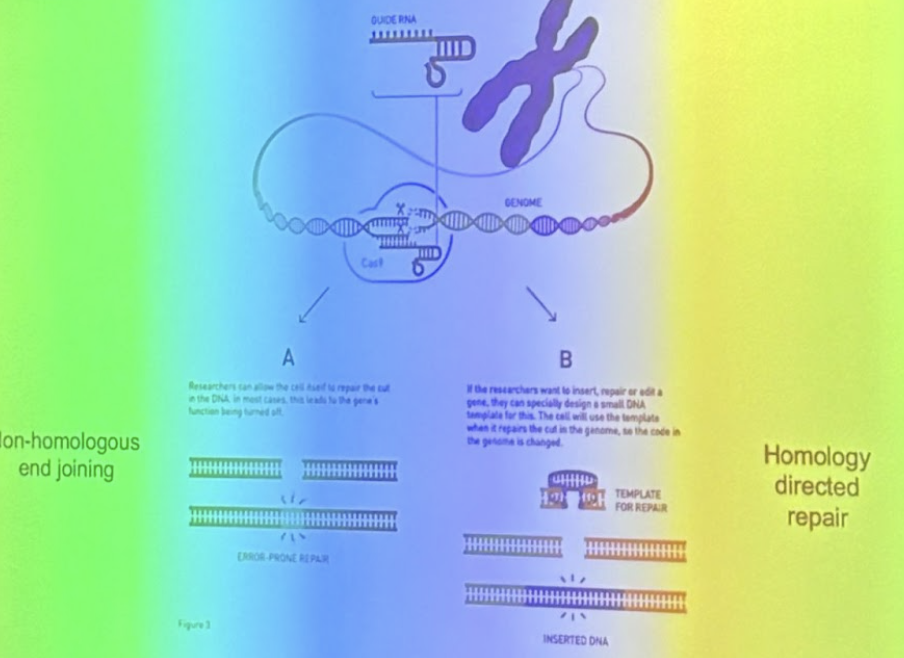
Two Mechanisms for Repairing Double-Stranded Breaks
A: Non-homologouse end joining
Quick but error-prone
Can cause small insertions or deletions that disrupt gene function
Used to disable genes
Used for knockouts: helps people figure out what the gene does bc can see what happens when it’s not working
B: Homology Directed Repair
Precise but slow
DNA template required
A custom DNA template can be inserted
Used for corrections, replacements, or insertions

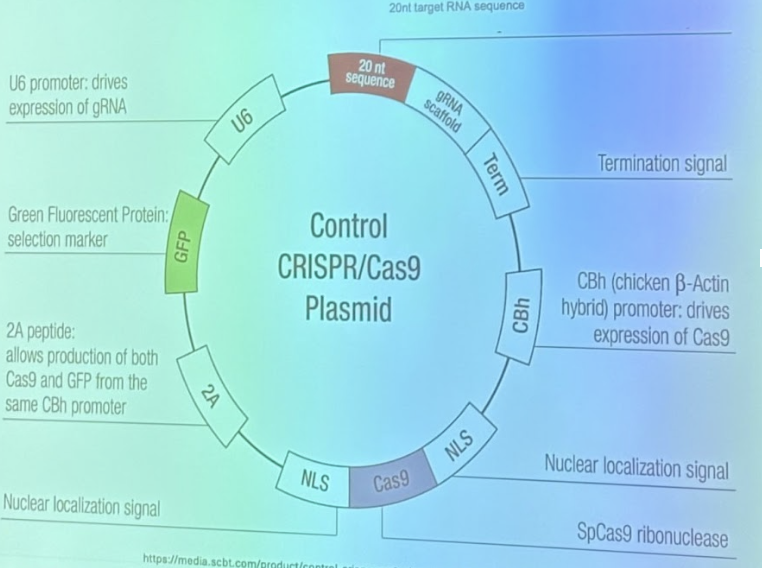
CRISPR-Cas9 Components
Key Components
Gene for Cas9
Complexed with Nuclear localization signal: so Cas9 will go to the nucleus, where the DNA is
20 nt target RNA sequence
Selection marker: makes the cell fluorescent green so you know CRISPR made it in to the cell

CRISPR-Cas9 Delivery Into Cells (In vitro: in a dish)
CRISPR-Cas9 on a plasmid
Ways to get plasmid into the cell
Microinjection: Take a needle and inject into cell
Electroporation: Electrocute the cell which creates transient holes in the cell for the plasmid to slip in
Could potentially kill the cell, only 10% efficiency
Viral Mediated Delivery Vehicles
Hallow out the inside of specific vral vectors, put plasmid in, then infect the cell with the virus
Non-Viral Mediated Delivery Vehicles
Cover DNA with lipid nanoparticles (LNPs) that will fuse to the cell membrane and allow entry into the cell
The In vivo (in a person) Delivery Problem
The gene therapy delivery problem in humans is that it is VERY difficult to efficiently deliver these therapeutic genes directly into a human (need to get it to many cells, not just one)
If, for example, trying to address something in lungs like cystic fibrosis, can use a virus that affects the respiratory tract only, like using a flu or common cold virus (use of viral mediated delivery vehicles)
Can also use the LNPs lipid nanoparticles
Genome Editing with CRISPR-Cas9
Sickle Cell Anemia is caused by a single base change in a gene (approved by FDA)
We have access to bone marrow, can get CRISPR into cells and return bone marrow
Collect blood stem cells
Modify cells
Inject back to body through infusion
Price: 2.2 or 3.1 million dollars for a single infusion
CRISPR Assisted CAR-T Cell Therapy (for cancer)
Can take T-cells out in blood and provde them with CAR-T gene that recognizes cancer cells so then Cas9 can recognize cancer cells and kill them
Price: 500k for infusion, but more like 1M with indirect cost of hospitalization
How Can CRISPR Be Used?
Edits in somatic cells stop with you, so any change to disease in you will not be passed to children, so children can still get the disease
IMPORTANT: differences in editing babies v. embryos, somatic v. germ line
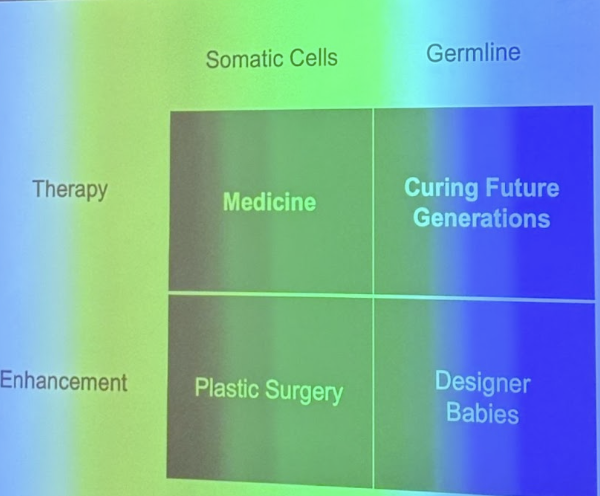

Debates about CRISPR
Concerns:
Access issues: only the wealthy will be able to cure their diseases, widens healthcare disparity
Can’t ensure there won’t be some off-site mutations, potential detrimental longterm affects
Religious reasons
Debate on what is or isn’t a “serious disease” that needs eradicated
Ethics of using CRISPR for enhancement (like attractiveness and intelligence) → Crosses into Eugenics
Crossing into the germline has severe ethical complications
Chinese scientist did already genetically engineer babies
Benefits:
Therapeutic applications: can be used to eliminate disease from the human genome