Lecture 2: Polyethylene (PE) and Polypropylene (PP)
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
Polyethylene has a low Tg (Tg= -120°C) but is still hard at room temperature. Why?
Polyethylene is a semi-crystalline thermoplastic polymer. It consists of:
Crystalline regions (ordered chain packing)
Amorphous regions (disordered chains)
The crystalline regions behave like physical crosslinks and give the material stiffness and hardness.
But the amorphous regions are flexible at room temperature (since Tg is so low) that gives polyethylene its ductility and it is therefore NOT brittle.
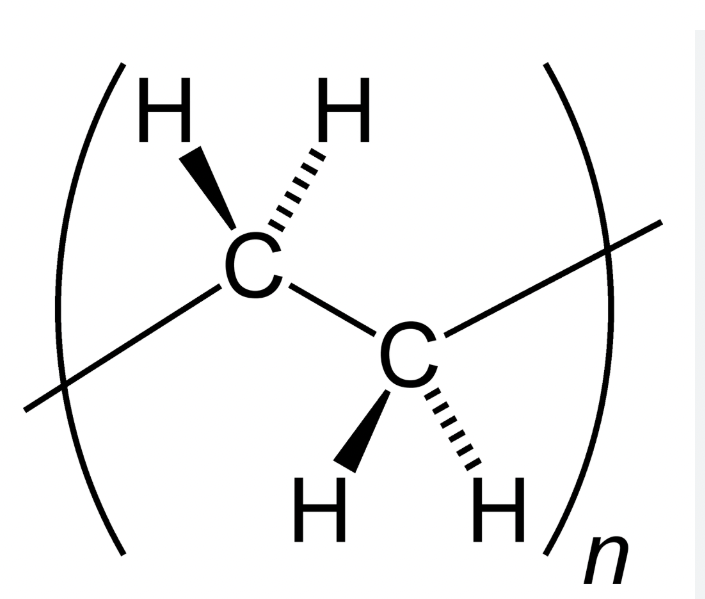
Draw the structure of Polyethylene (PE)
(C2H4)n
Mention the density, Tm and Tg for Polyethylene
Density: 0.905 -0.965 g/cm3 , will float in water! Most plastics sinks
Tm= 105-140°C
Tg= -120°C
Where does the majority of the monomer of PE (ethylene) come from?
Large majority of ethylene comes from petroleum sources such as Naphta
Explain one synthesis method for LDPE
Synthesis method: High-pressure polymerization
Polymerization technique: Free Radical Polymerization
Monomer: Ethylene
Initiator: Oxygen
The process occurs under high temperature and pressure in a tube reactor
The degree of branching is controlled by the temperature, pressure, and reactor type. High pressure gives a higher molecular mass of polymer.
Risk: The reaction is highly exothermic which can lead to uncontrolled temperature increase. To avoid this, powerful stirring and cooling systems are used.
How is branches formed in LDPE?
Branches are usually formed during Free Radical Polymerization of LDPE via chain transfer reactions.
Explain one synthesis method for HDPE and LLDPE
Polymerization technique: Coordination polymerization
Catalyst: Ziegler-Natta Catalysts
What is LLDPE?
Linear low density polyethylene (LLDPE) is a copolymer of ethylene and alpha-olefins (such as 1-butene or 1-hexene)
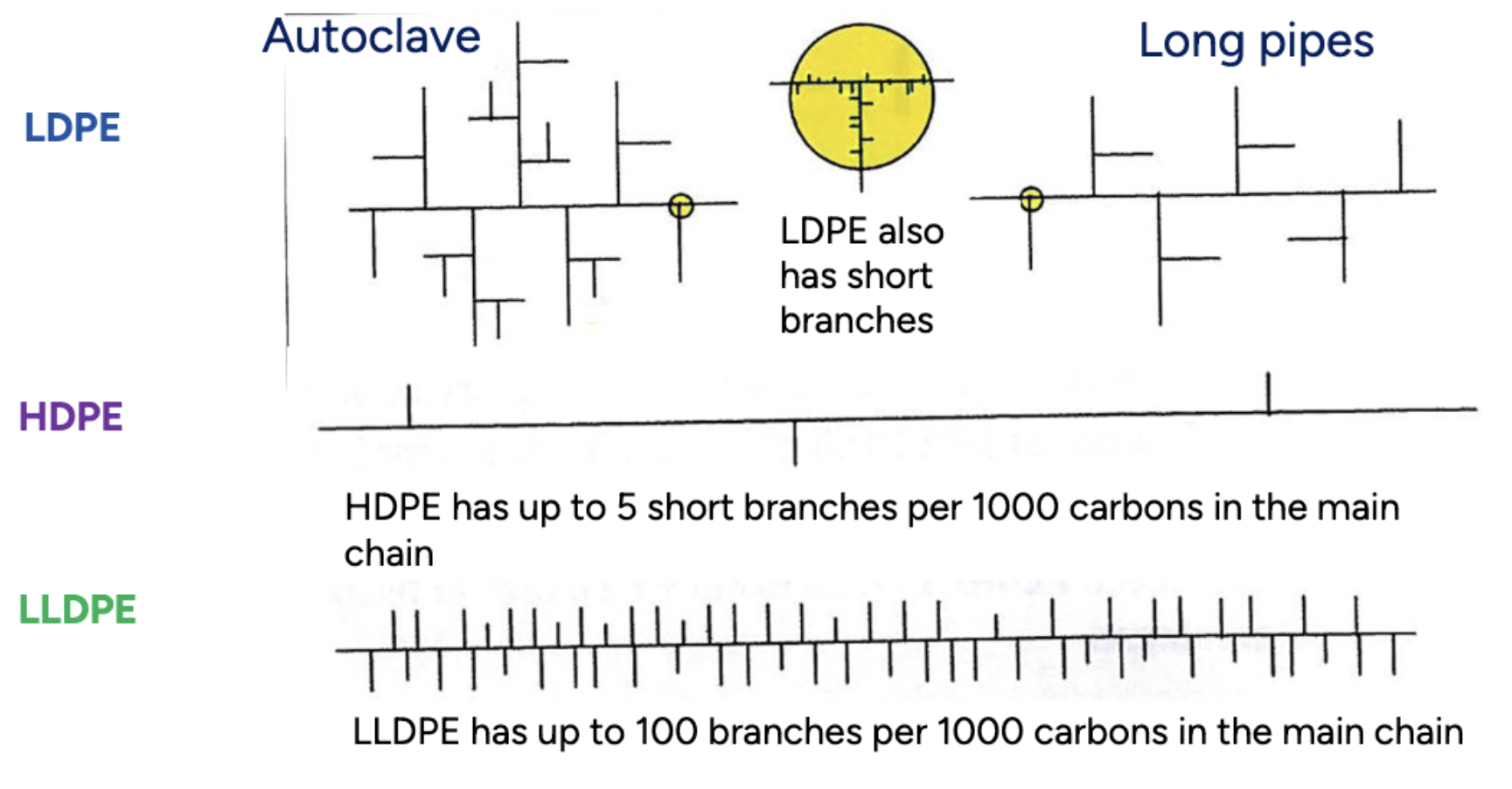
Compare the properties between LDPE, HDPE and LLDPE
LDPE has a low density and a high degree of branching. The degree of crystallinity is about 40%. Melting temperature is 105°C.
HDPE has a high density and a low degree of branching. The degree of crystallinity is about 95% (because of the low degree of branching). Melting temperature is 140°C.
LLDPE has a medium density, but more toward the lower end. The degree of crystallinity is higher than 50%. LLDPE has intermediate properties between LDPE and HDPE. Melting temperature is 130°C.
Compare the structures between LDPE, HDPE and LLDPE
LDPE has a highly branched structure
HDPE has a linear structure with minimal branching
LLDPE has a linear structure with short branches
Mention some application of Polyethylene
Toys
Pipes
Bags
Food containers
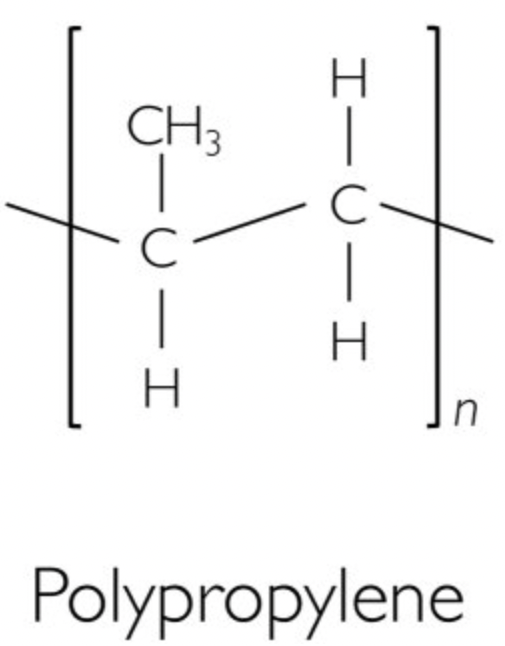
Mention the crystallinity and monomer of Polypropylene (PP)
Polypropylene is a semicrystalline thermoplastic.
Monomer: Propylene
Mention the density, Tg and Tm for Polypropylene.
Density: 0.9 g/cm3
Tg= -10°C
Tm= 160-170°C
Where does the monomer of Polypropylene come from?
Polypropylene comes from petroleum based sources such as Naphta
Explain one polymerization technique of Polypropylene
Spheripol-process:
Ziegler-Natta catalyst
No atactic polypropylene is formed
97% of PP produced is isotactic
Explain some important structure - properties for PP regarding chain stiffness and thermal stability.
Polypropylene has a methyl group on every other carbon atom in the chain. The methyl group will give steric hindrance and therefore, the chain can not rotate as freely. This will give a higher stiffness and Tg compared to polyethylene.
The tertiary carbon in the chain is more reactive and sensitive towards oxidation. Therefore, Polypropylene is more sensitive towards heat and UV radiation compared to polyethylene.
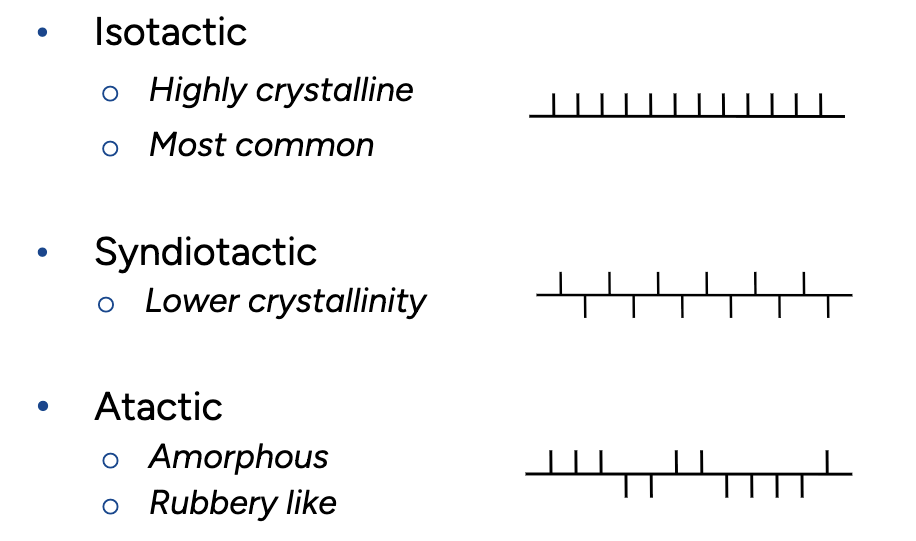
Describe the structure-properties for Isotactic PP, Syndiotactic PP and Atactic PP
Isotactic PP: Highly crystalline - stiff and strong material because all methyl groups is on the same side
Syndiotactic PP: Lower crystallinity because of alternating placement of methyl group, less rigid and higher impact resistance (because it doesnt crack easily)
Atactic PP: Amorphous, rubbery like because of random placement of methyl groups
Mention 3 applications for Polypropylene
Bottles
Packaging
Instrument panel
Name one important additive for polypropylene (PP)
Antioxidants is an important additive because it hinders thermal degradation by oxidation of the tertiary carbon.