Lecture 3 - Amino Acids & Protein Structures
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
What is the function of enzymes?
catalyze covalent bond breakage or formation. ex: pepsin degrades dietary proteins
What is the function of structural proteins?
provide mechanical support to cells and tissues ex: collagen
What is the function of transport proteins?
carry small molecules or ions. ex: hemoglobin
What is the function of motor proteins?
generate movement in cells and tissues. ex: myosin
What is the function of storage proteins?
store amino acids or ions. ex: ferritin stores iron
What is the function of signal proteins?
carry extracellular signals from cell to cell. ex: insulin
What is the function of receptor proteins?
detect signals and transmit them to the cell's response machinery. ex: rhodopsin in the eye detects light
What is the function of transcription regulators?
bind to DNA to switch genes on or off. ex: the Lac repressor in bacteria silences the genes for enzymes that degrade lactose
What is the function of special-purpose proteins?
highly variable (LOL). ex: antifreeze proteins of artic fishes protect their blood from freezing.
How many peptide chains are antibodies made up of?
4 peptide chains - 2 light, 2 heavy
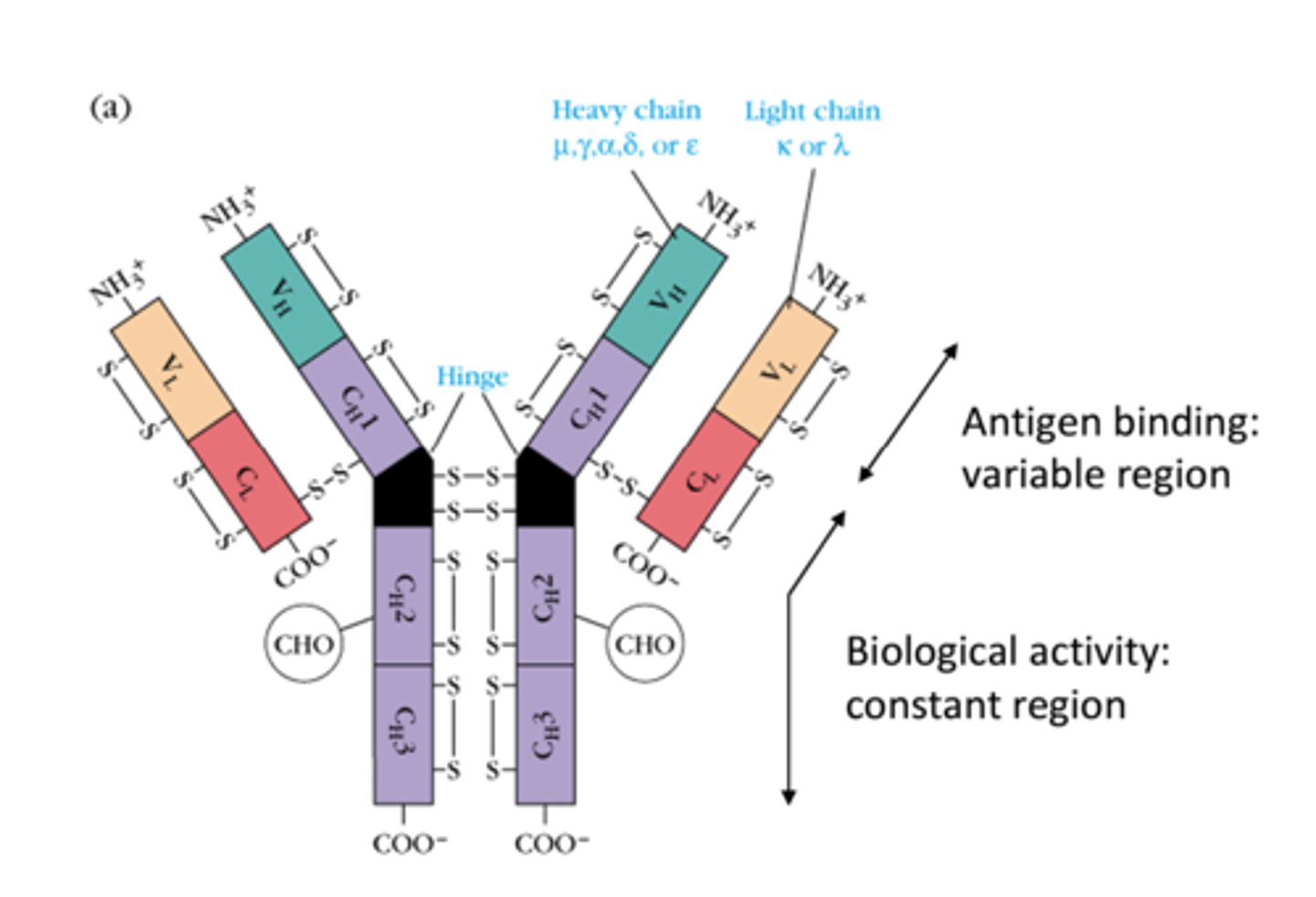
What are the peptide chains in the antibodies held by?
disulfide bonds
What part of amino acids are unique from the other 19 amino acids?
R-groups. backbone is the same.
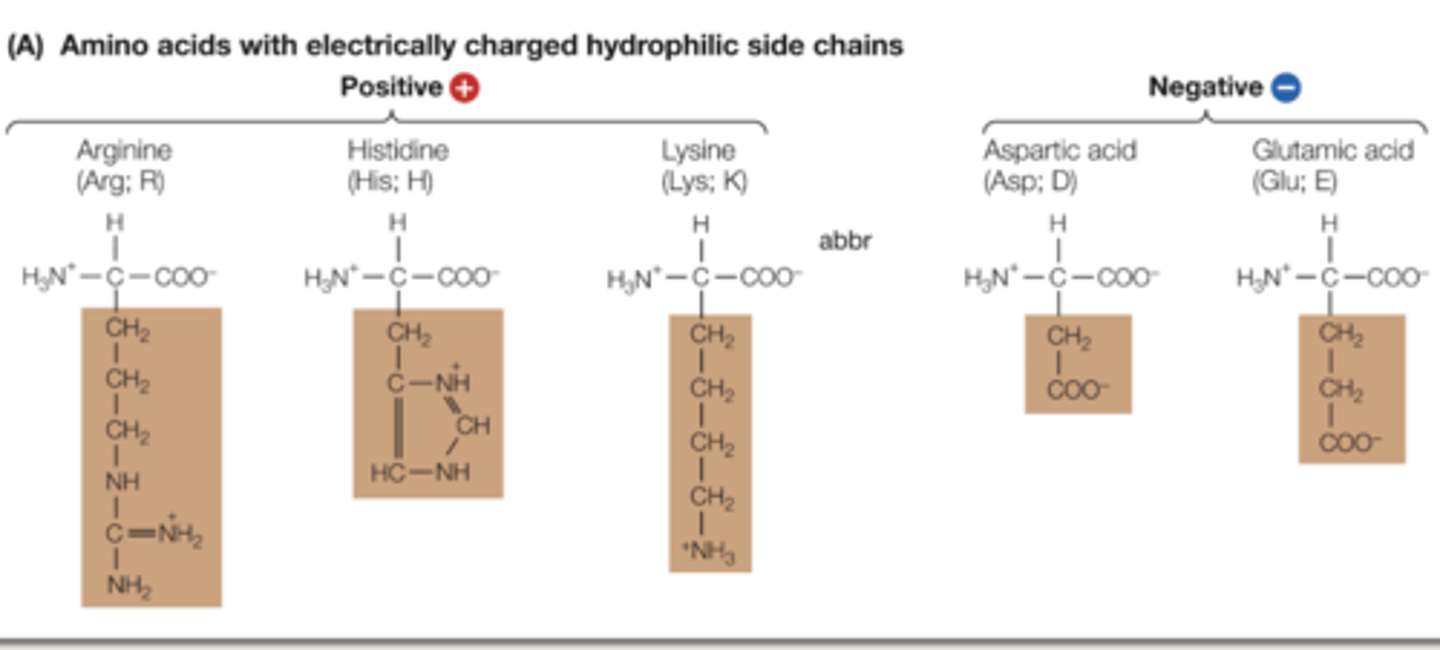
Explain characteristics of strongly hydrophilic amino acids.
they contain polar covalent bonds, and are positively or negatively charged. interacts with water. Have 4 things connecting to the N in the R group
Positive: Arginine, Histidine, Lysine. (have NH)
Negative: Aspartic acid, Glutamic acid. (have COO-)
(do not need to memorize)
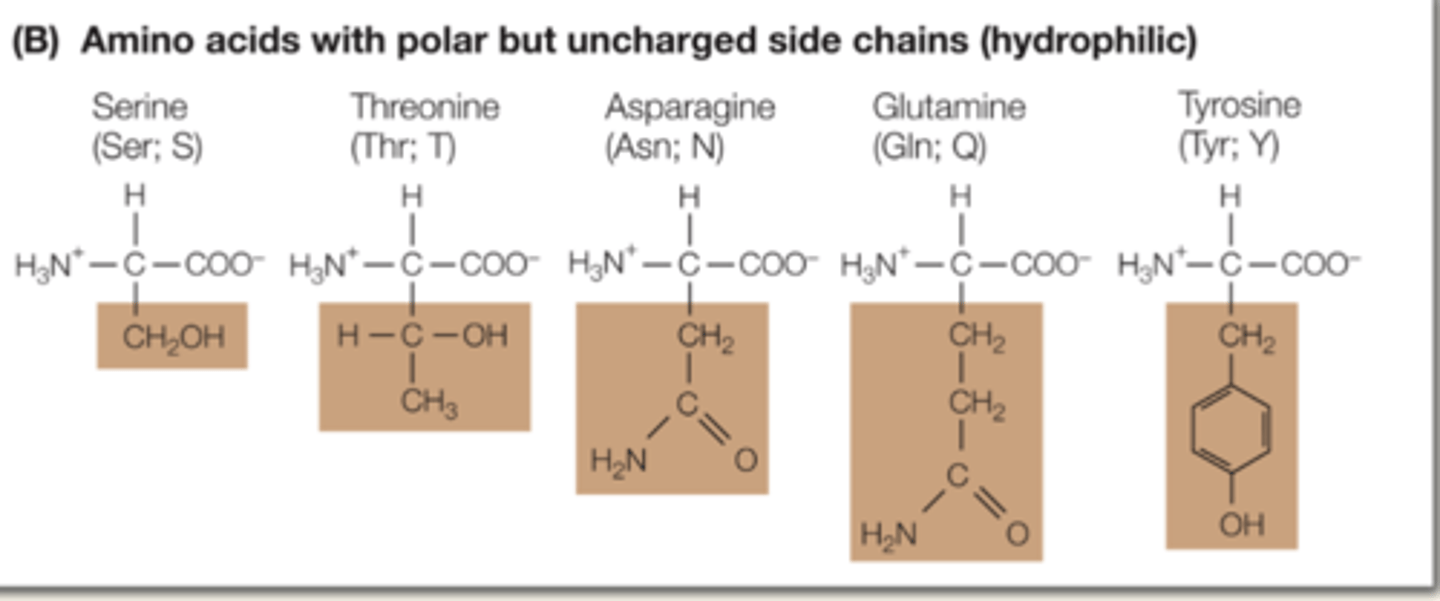
Explain characteristics of mildly hydrophilic amino acids.
they contain polar covalent bonds, and have no charges. they can interact with water.
Serine, Threonine, Asparagine, Glutamine, Tyrosine. (have O)
(do not need to memorize amino acids)
Explain characteristics of generally hydrophobic amino acids.
non-polar amino acids. they all have special cases:
- Cysteine: has polar covalent bonds but overall non-polar, but hides within proteins (protected from water). Can form disulfide bonds.
- Glycine: small side chain, one hydrogen. When folded, small amino acids can fit into small spaces.
- Proline: side chain forms ring structure.
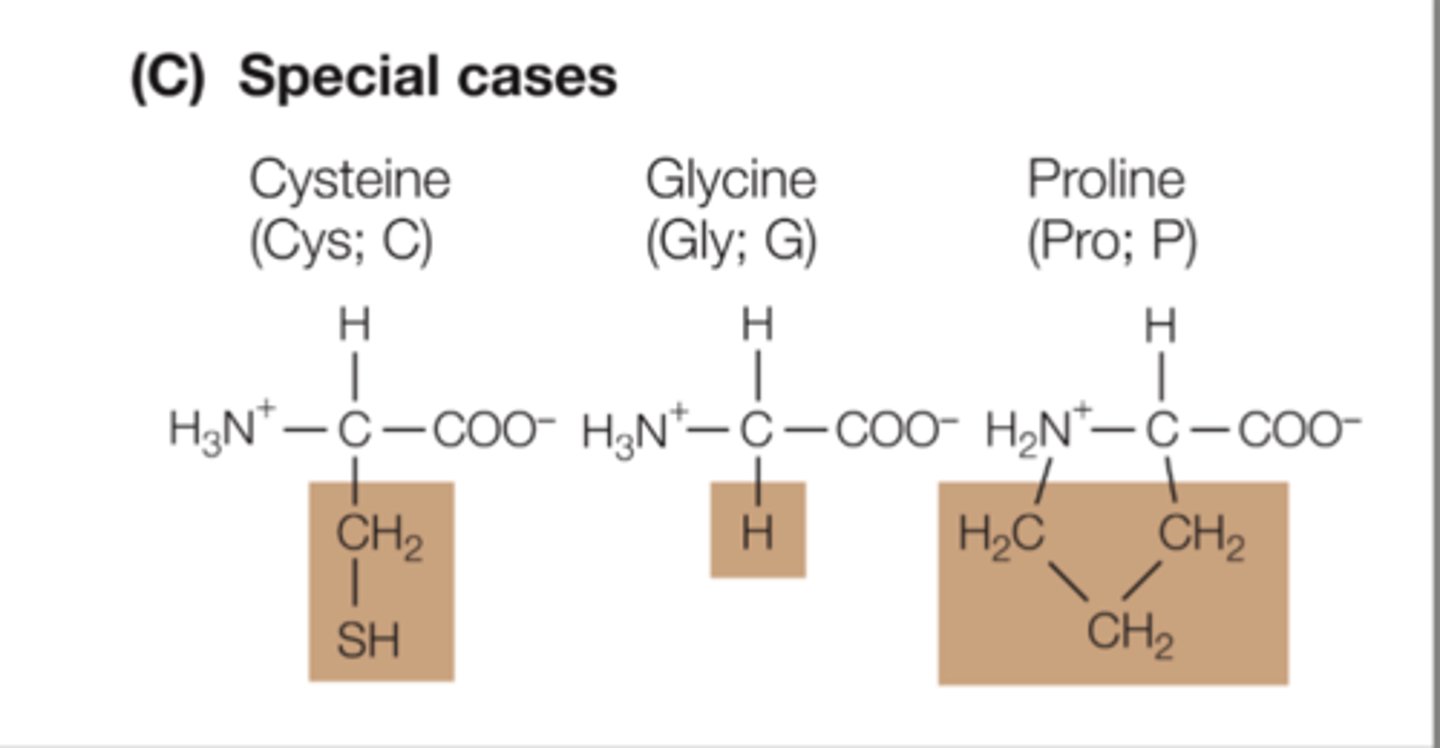
Explain characteristics of hydrophobic amino acids.
non-polar amino acids and bonds. must be sequestered from water (would go in the interior of the protein in contact with non-polar side chains).
Alanine, Isoleucine, Leucine, Methionine, Phenylalanine, Tryptophan, Valine
(don't need to memorize amino acids)
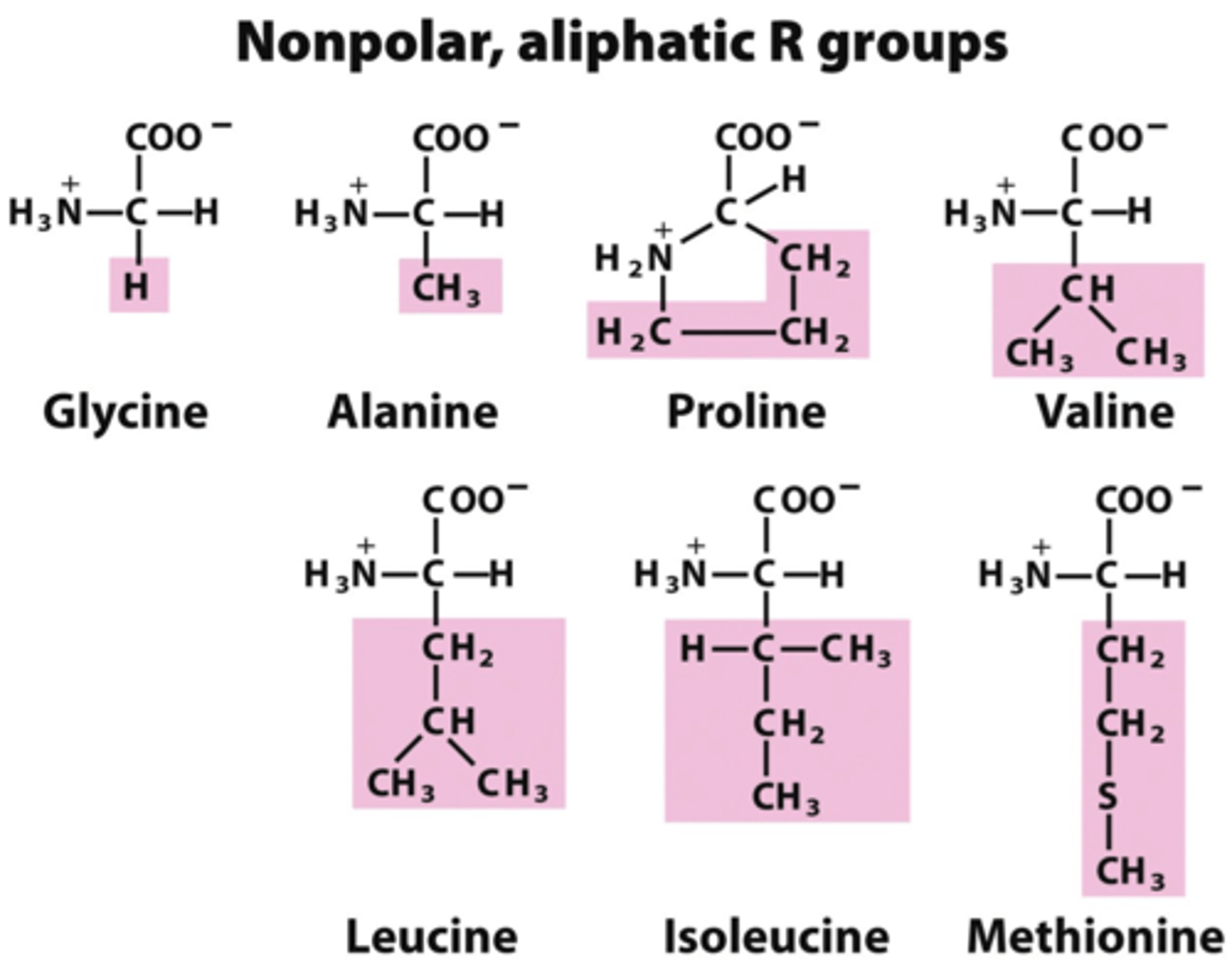
How to tell if an amino acid is polar (hydrophilic) or non-polar (hydrophobic)?
non-polar (hydrophobic) amino acids DO NOT contain oxygens in their r-groups.
polar (hydrophilic) amino acids either have a charged r-group or the r-group contains oxygen.
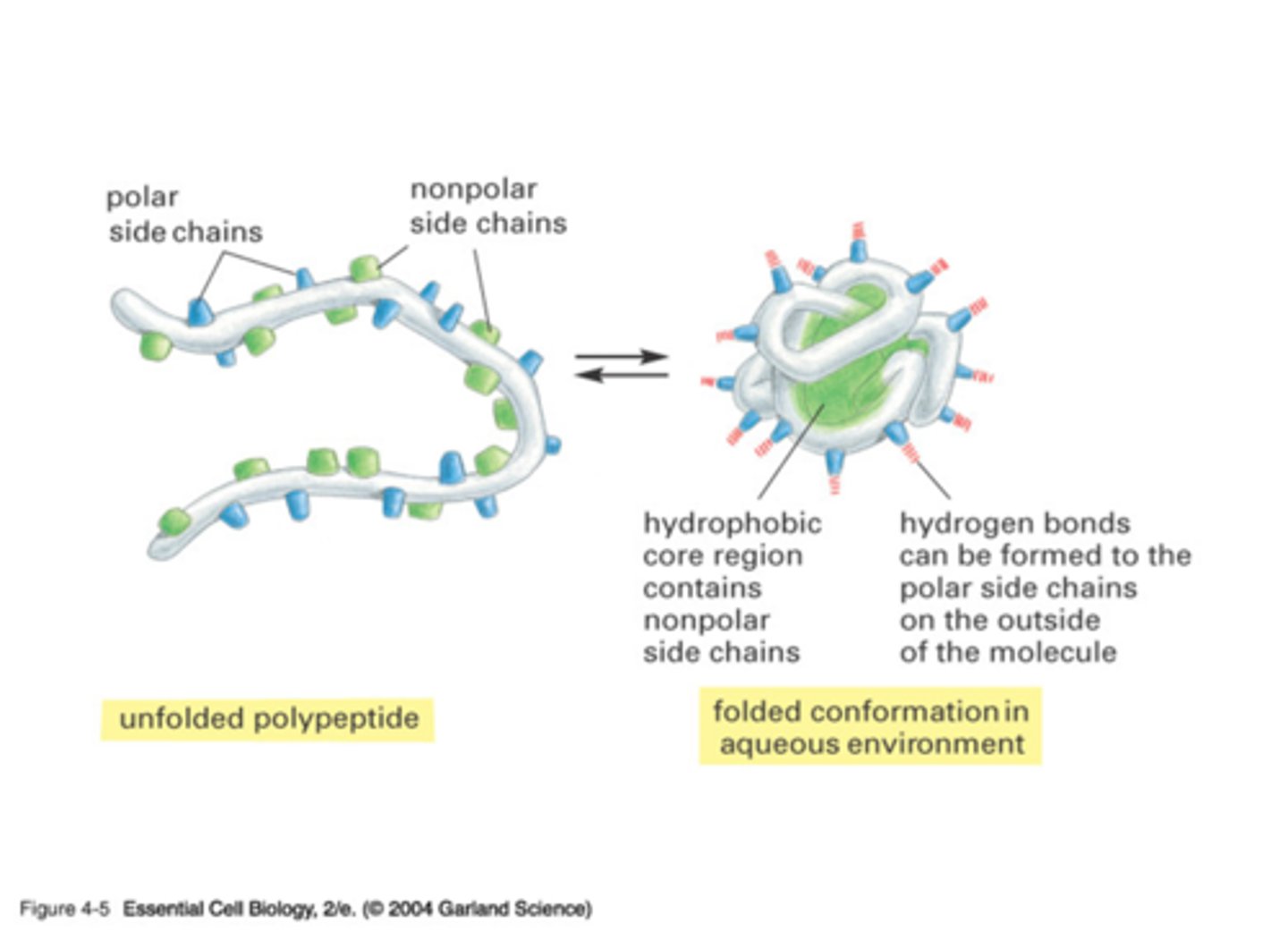
The Sequence of Amino Acids Dictates Protein Shape
the amino acid sequence -- drives the interactions with water and intramolecular bonding of the linear amino acid chain -> determines the protein's unique 3D shape
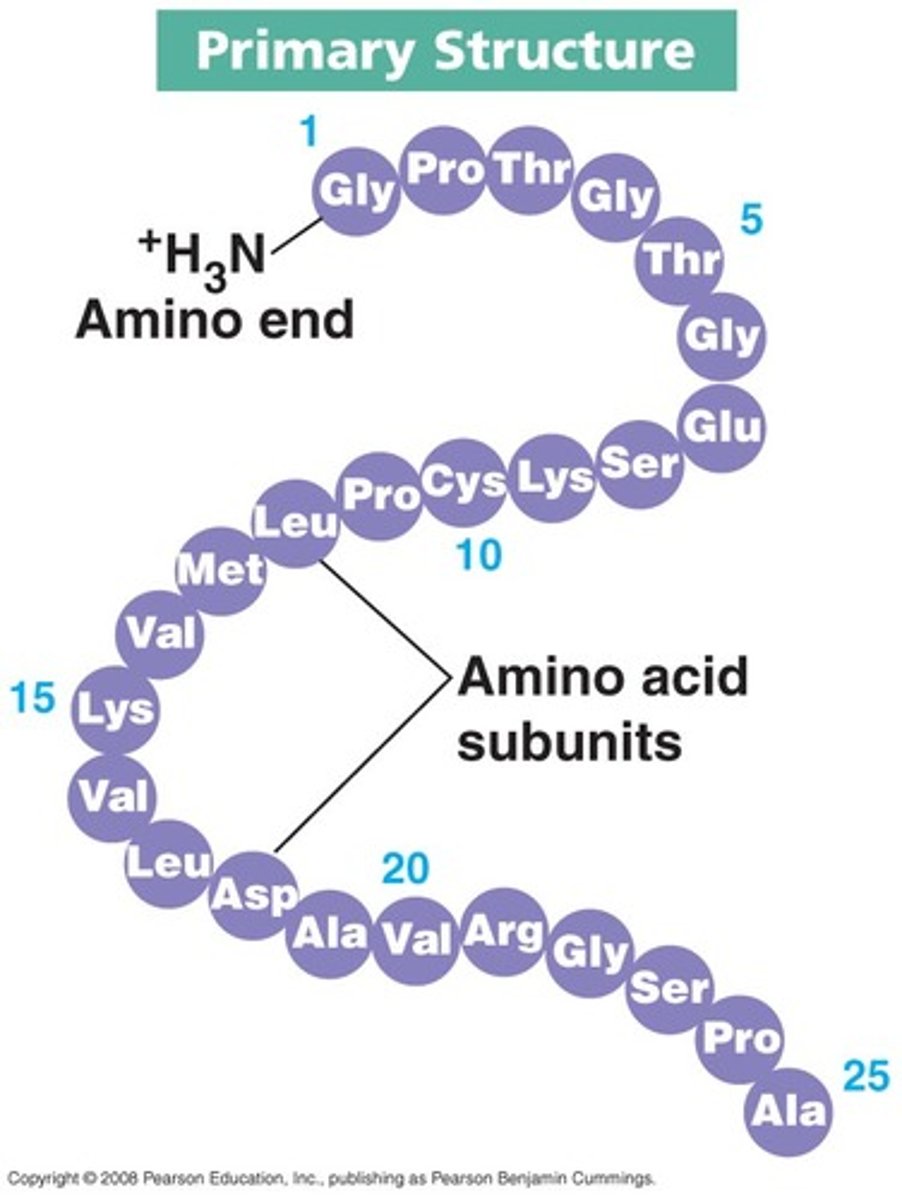
Explain characteristics of primary structure. (description, bonds)
- sequence of amino acids (monomers) joined by peptide bonds. creates polypeptide chains.
- order of amino acids dictate secondary and tertiary levels (shape)
Explain characteristics of secondary structure. (description, bonds)
- polypeptide chains may form alpha helices or beta pleated sheets.
- stabilized by hydrogen bonds and noncovalent bonds between atoms in peptide backbone, results in repeating pattern of helices or pleated sheets
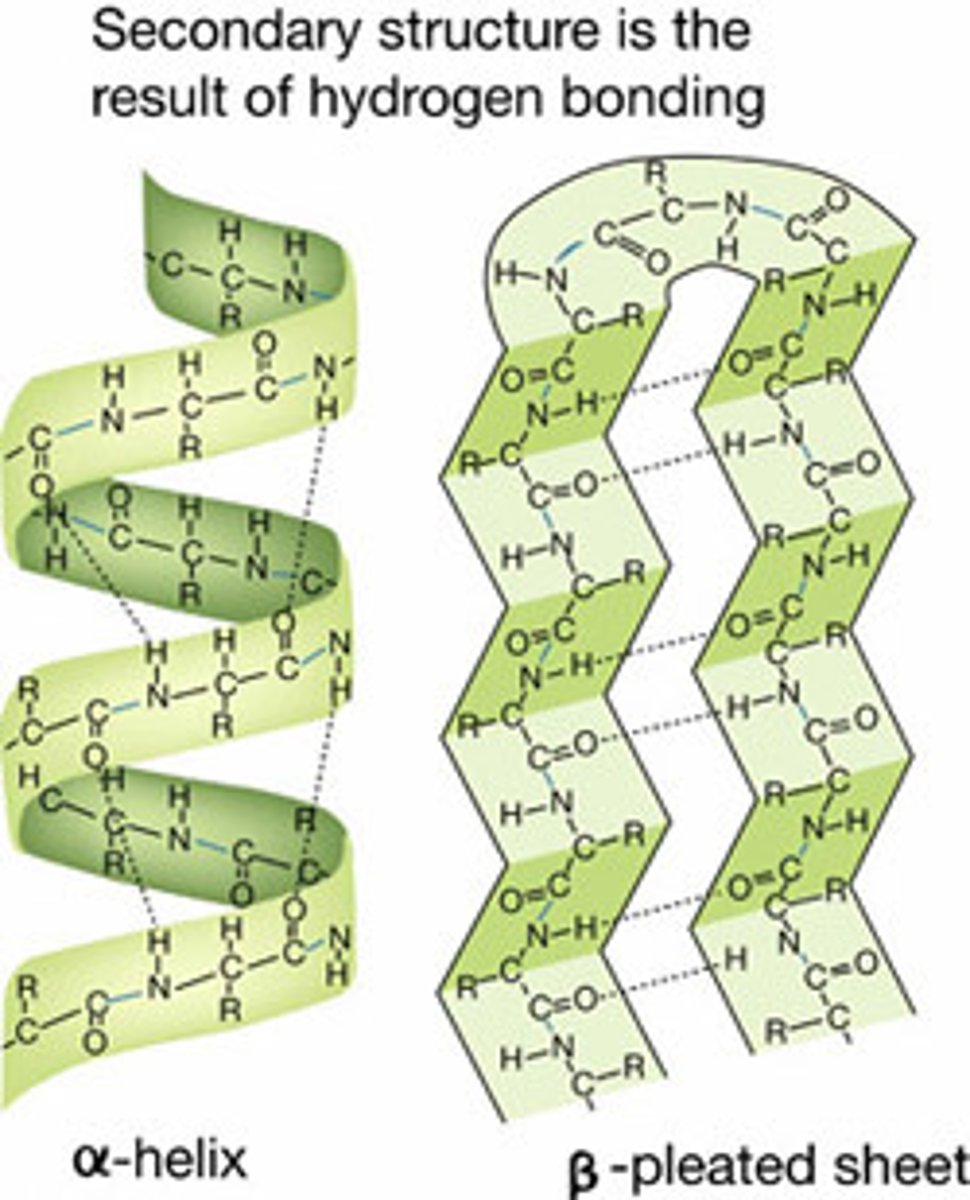
Where is the hydrogen bond in an alpha helix?
the N-H of every peptide bond is hydrogen-bonded to the C=O of a neighboring peptide bond 4 amino acids away from the same chain.
How are Beta pleated sheets made?
several strands of an individual polypeptide chain are held together by hydrogen-bonding in adjacent strands. amino acid strands project alternately above and below the plane of the sheet.
Explain characteristics of tertiary structure. (description, bonds)
- polypeptides fold, forming specific 3D shapes.
- stabilized by hydrogen bonds, disulfide bridges, van der waals - hydrophobic interactions (in the hydrophobic regions), and ionic interactions (salt bridges)
- determined largely by interactions between R-groups (side-chains)
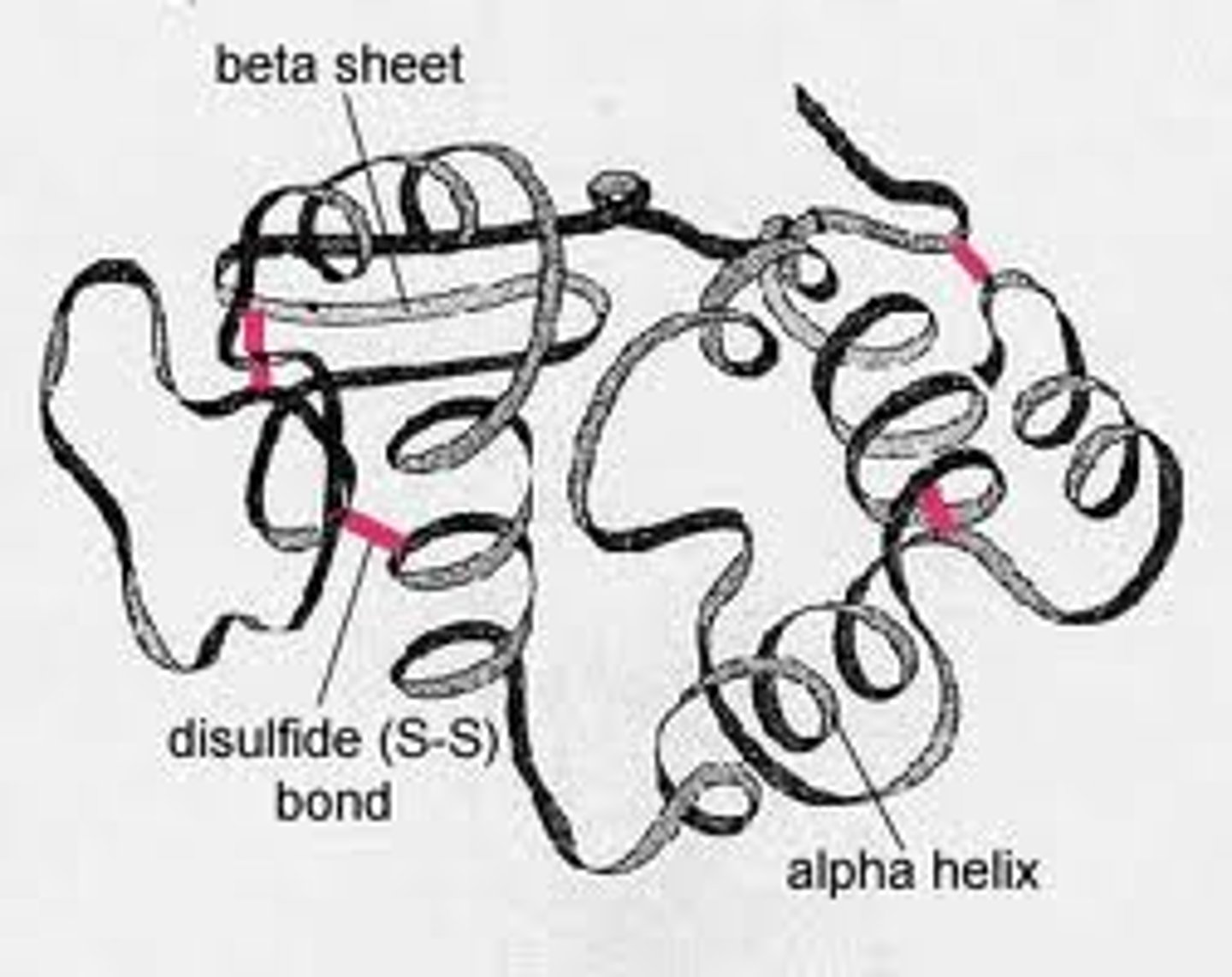
What type of bond is a disulfide bond? Where does it occur?
covalent. between two cysteines. stabilizes the tertiary and quaternary structures.
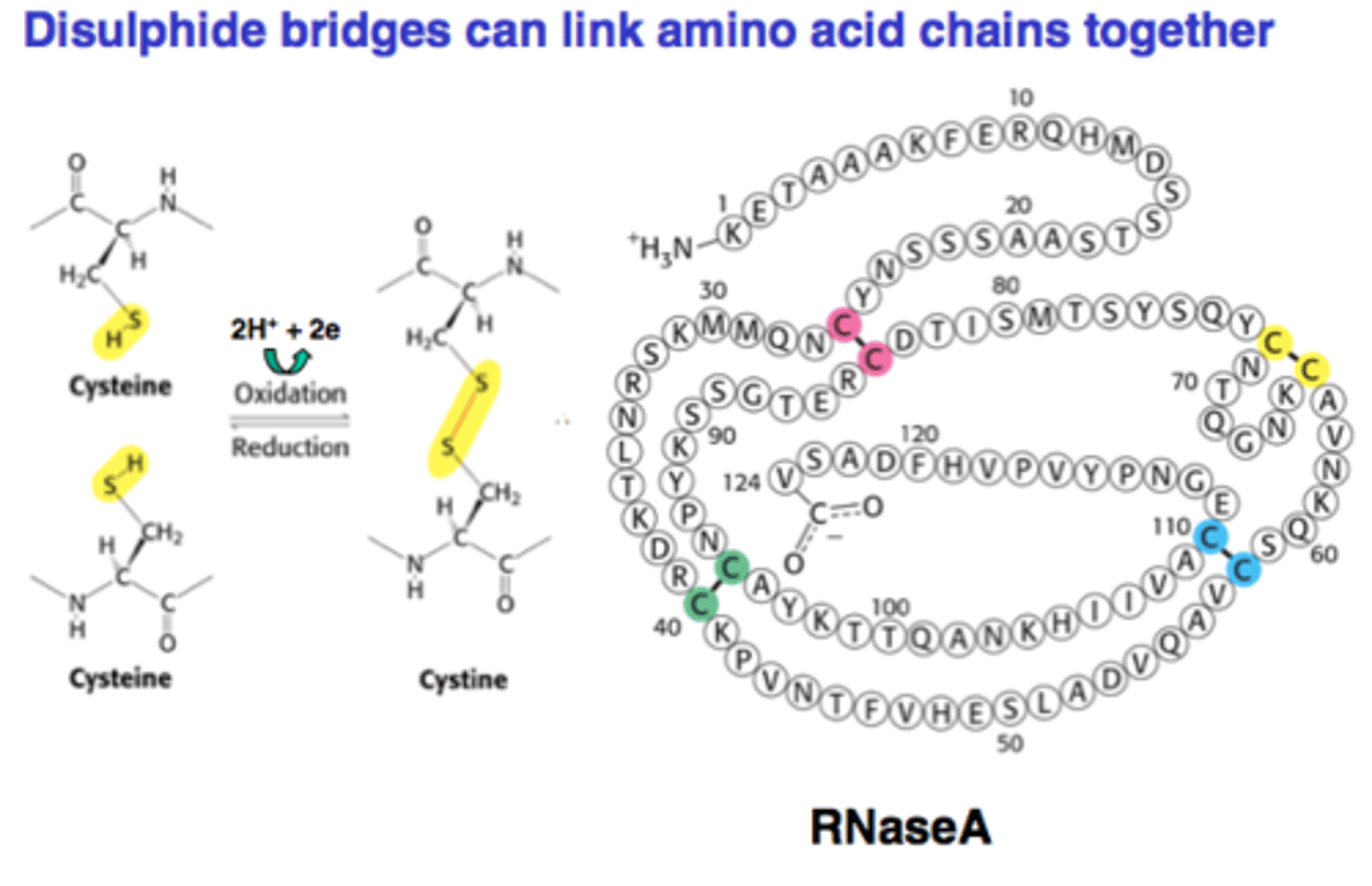
Explain interactions between R Groups in tertiary structures of multiple proteins.
- ionic interactions/salt bridges can form between charged amino acid side chains.
- hydrophilic side chains/R groups can form hydrogen bonds with peptide backbone and with other side chains/R groups.
- hydrophobic amino acids will be clustered inward to the center of the protein structure through Van Der Waals interactions (other than membrane proteins)
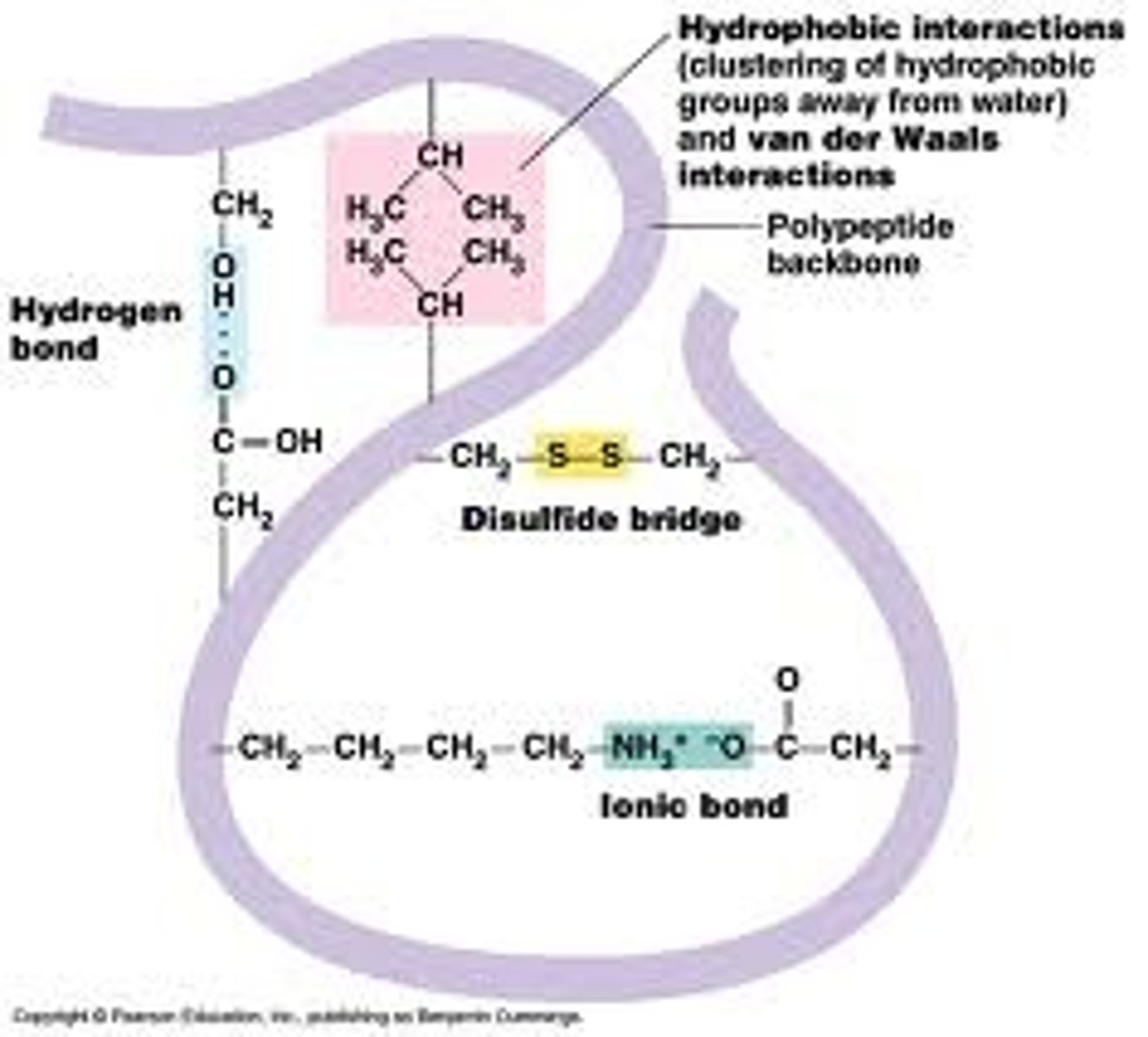
Where are hydrogen bonds found in protein levels?
- secondary structure: supports helices and pleated sheets. (between backbone and backbone)
- tertiary structure: supports final chain structure
(between backbone and side chain/R group, between side chain/R group and side chain/R group)
- quaternary structure
Explain characteristics of quaternary structure.
- two or more polypeptides assemble to form larger protein molecules.
- hydrogen bonds, disulfide bridges, hydrophobic interactions, ionic interactions
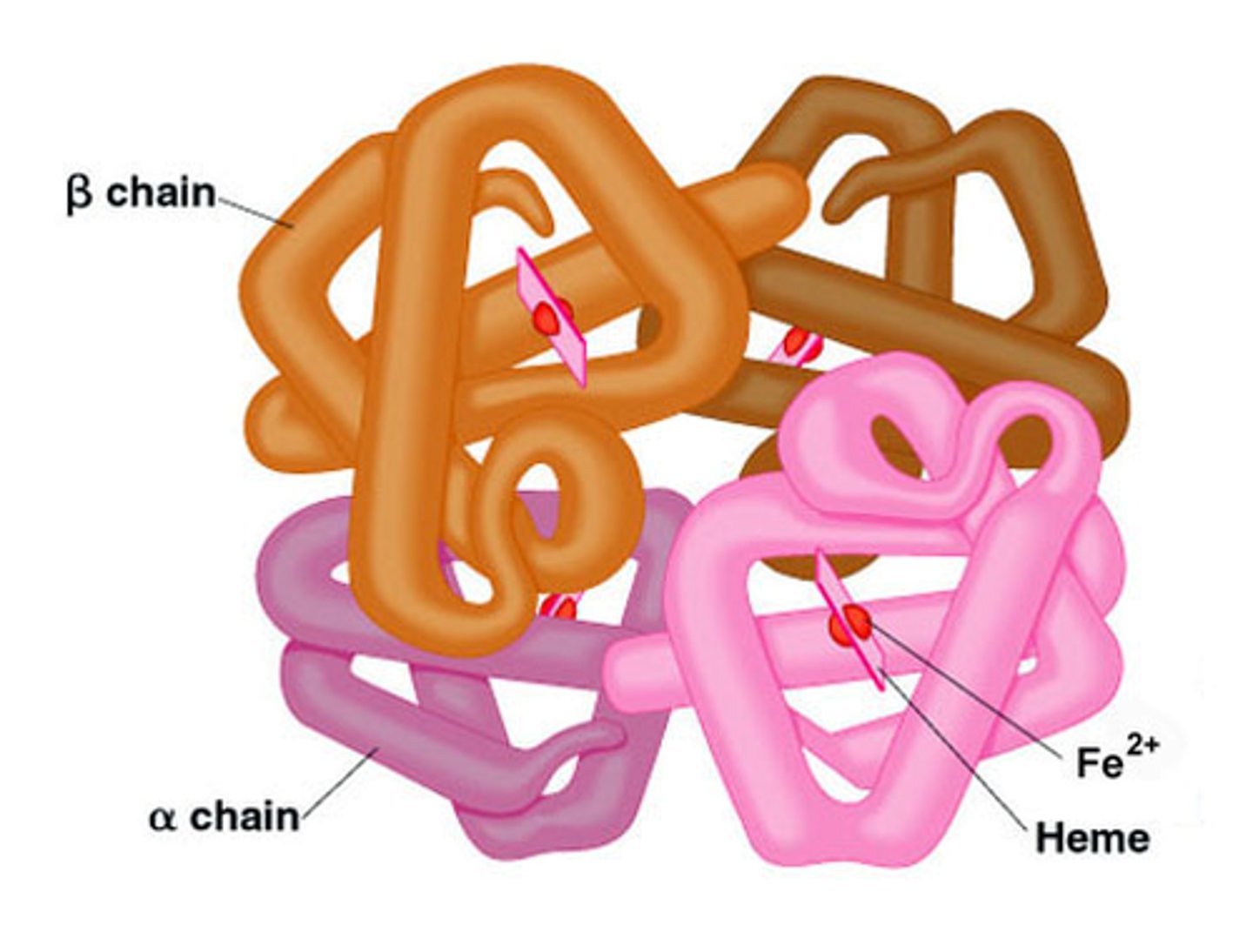
Explain hemoglobin in terms of quaternary structure.
- made up of 4 polypeptide chains.
- each chain cradles a molecule of heme, which is where oxygen is bound.
- multiple subunits come together to form a functional protein
What do chaperone proteins do?
help proteins fold properly.
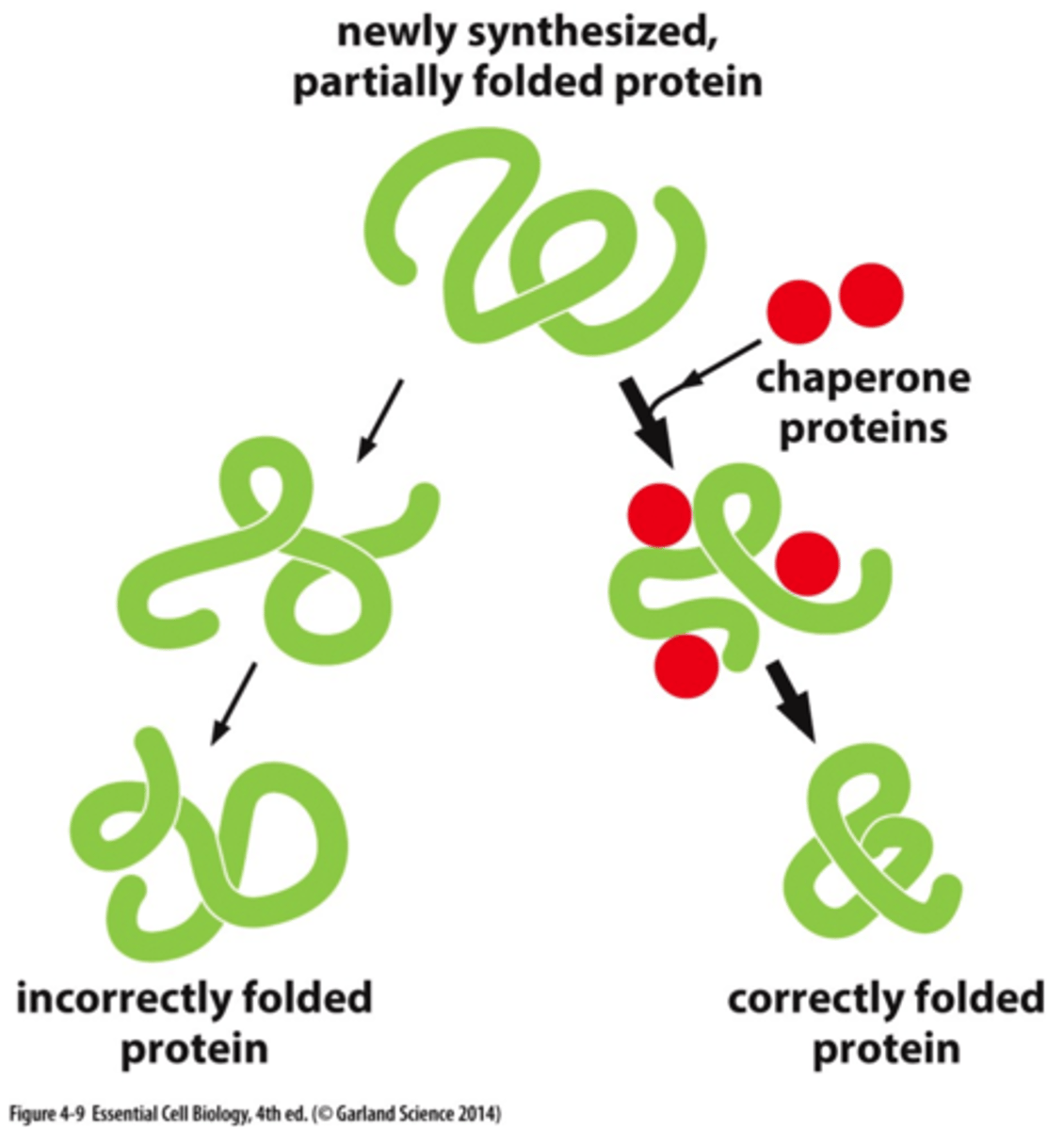
Why is it important for proteins to fold properly?
misfolded proteins are the primary cause of many human degenerative and neurodegenerative diseases.
Example of diseases caused by misfolded proteins.
- alzheimer's
- parkinson's
- huntington's
- ALS
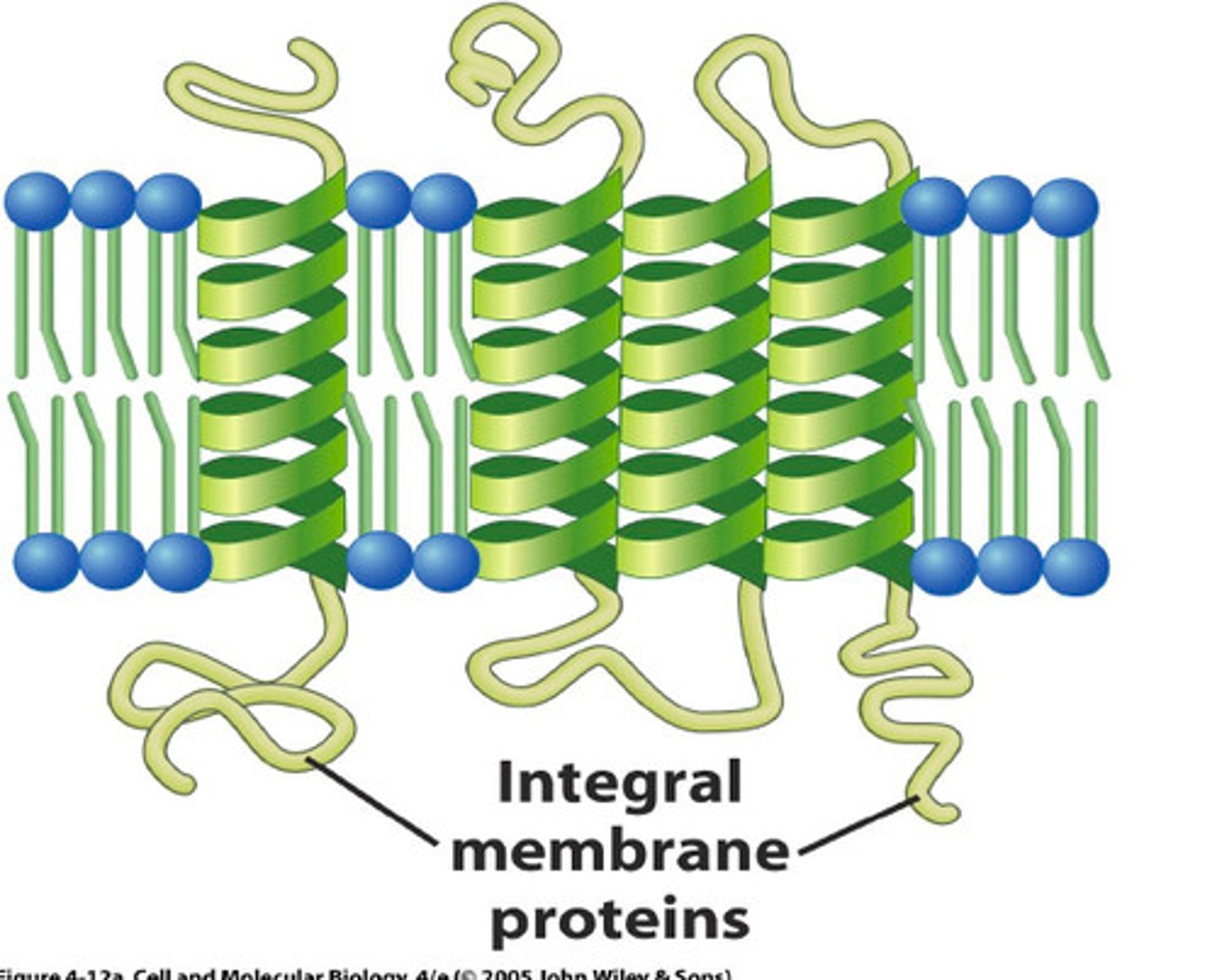
How are membrane proteins structured?
the hydrophobic side chains of amino acids that form the alpha helix make contact with the hydrophobic hydrocarbon tails of the phospholipid molecules. The hydrophilic parts of the polypeptide backbone form hydrogen bonds with one another along the inside of the helix.