o chem exam 2
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
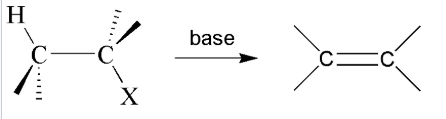
dehydrohalogenation
Reactant: alkyl halide
Reagent: strong base
product: alkene
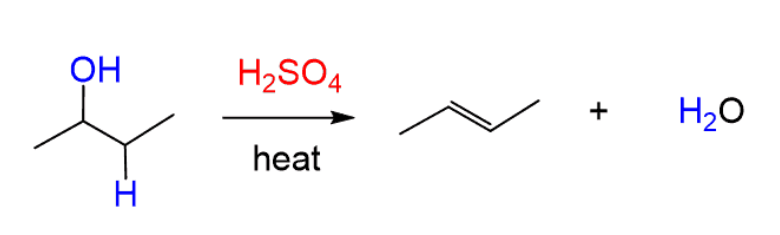
dehydration
reactant: alcohol
reagent: H2SO4
product: alkene
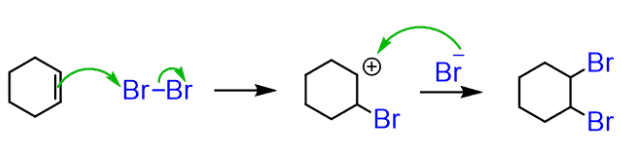
halogenation
reactant: alkene
reagent: Br2 or Cl2
product: each carbon gets 1 halogen
halohydrin reactant: alkene
reagent: Br2 or Cl2 with H2O
product: halohydrin (adding a halogen and a hydroxyl group to adjacent carbons)
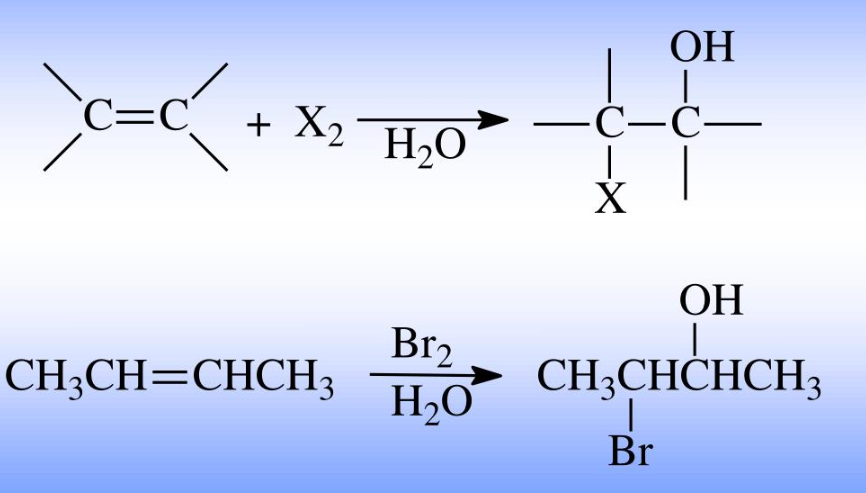
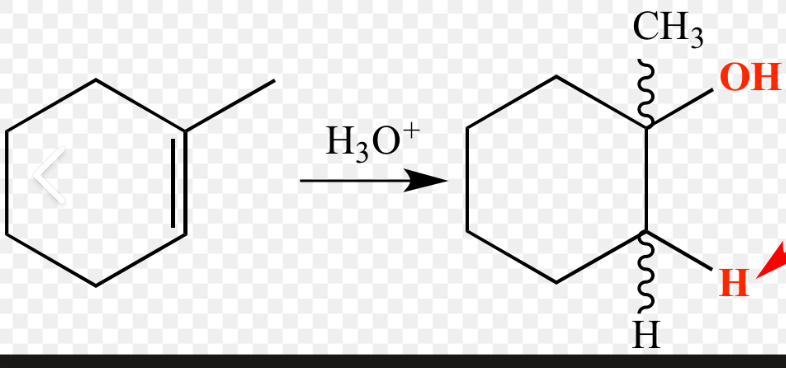
hydration (acid catalyzed)
reactant: alkene
reagent: H3O+
product: alcohol
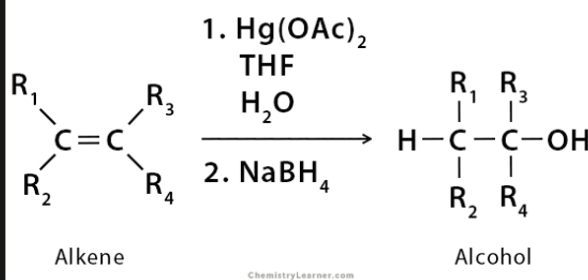
oxymercuration-demurcuration
reactant: alkene
reagent: Hg(OAc)2/THF/H2O then NaBH4
product: alcohol
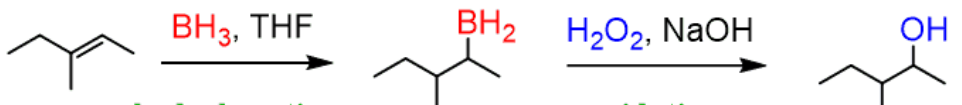
hydroboration-oxidation
reactant: alkene
reagent: 1. BH3 THF 2. H2O2, OH-
product: alcohol
hydrogenation (reduction)
reactant: alkene
reagent: H2 and metal (PtO2 or Pd/C)
product: alkane
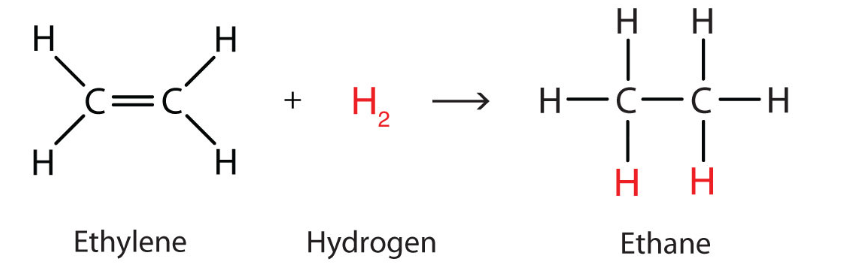
oxidation (mCPBA)
reactant: alkene
reagent: mCPBA
product: Epoxide (has 3-membered ring with O)
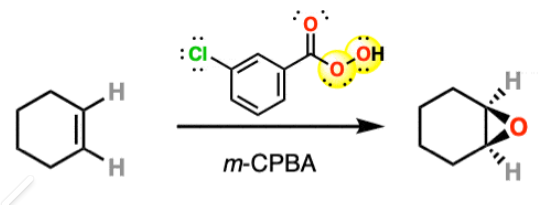
epoxide ring opening
reactant: epoxide
reagent: acid or base (probably H3O+)
]
product: trans-diol
![<p>]</p><p>product: trans-diol</p>](https://assets.knowt.com/user-attachments/4bbfe922-1716-437d-9b16-7eb58cf8f7f8.png)
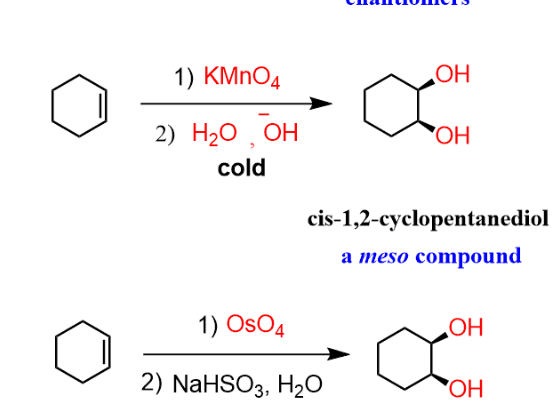
synthesis of cis-diols
reactant: alkene
reagent: 1. OsO₄ or KMnO₄(NMO) 2. NaHSO3
product: 2 OH (same side)
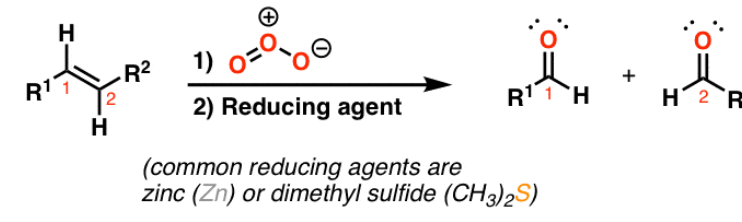
ozonolysis
reactant: alkene
reagent: O₃ → Zn or H3O+
product: carbonyl compounds
KMnO4
reactant: alkene
reagent: KMnO4
product: cis-diol
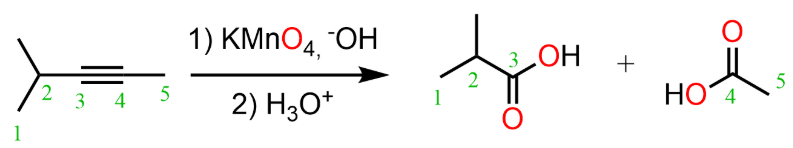
addition of alkynes (HX)
reactant: alkyne
reagent: H-X (x can be Br)
product: alkene
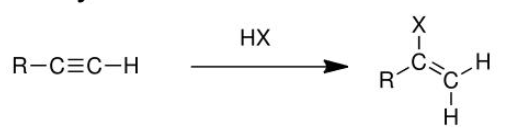
addition of alkynes (X2)
reactant: alkyne
reagent: X2 (Br2)
product: alkane
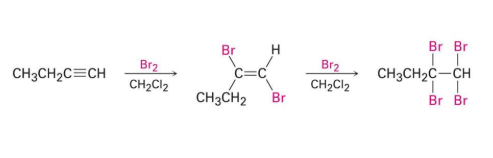
lindlar’s catalyst
reactant: alkyne
reagent: H2 and Lindlar’s Catalyst
product: cis-alkene
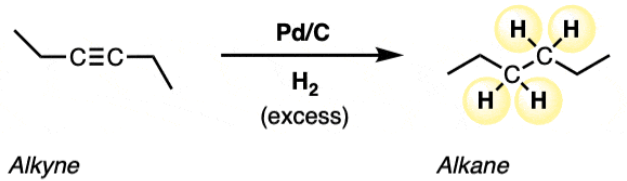
H2 , Pd/C reduction for alkynes
reactant: alkyne
reagent: H2 , Pd/C
product: alkane
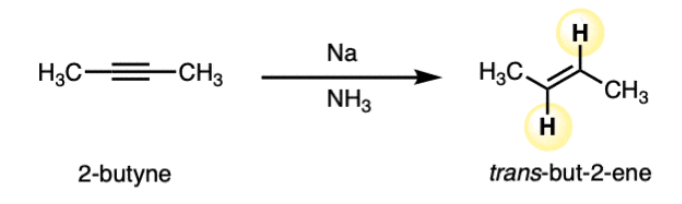
Na or Li in NH3
reactant: alkyne
reagent: Na or Li, NH3
product: trans-alkene
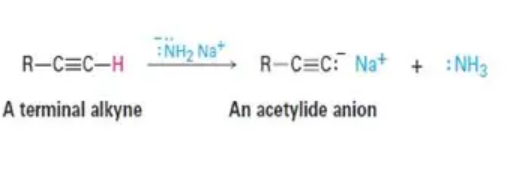
acidity for alkynes
reactant: terminal alkyne (triple bond at end)
reagent: strong base (NaNH2
product: acetylide anion (nucleophile)
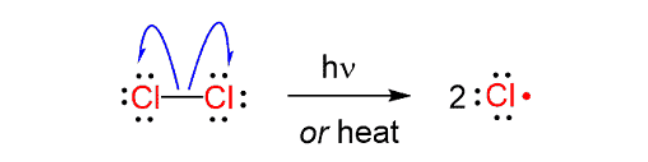
initiation (free radical halogenation for organohalides)
reactant: X2 (Cl2 or Br2)
reagent: hV(light)
product: 2X•
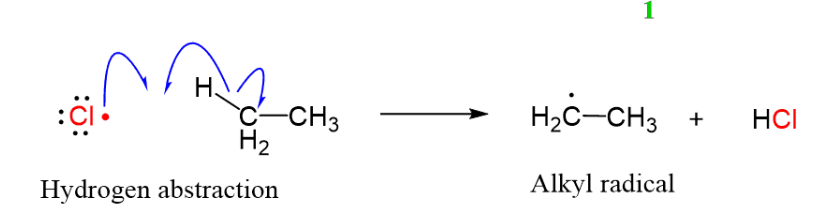
propagation (free radical halogenation for organohalides)
reactant: Alkane + X• (Br or Cl)
product: haloalkane and HCl as side product
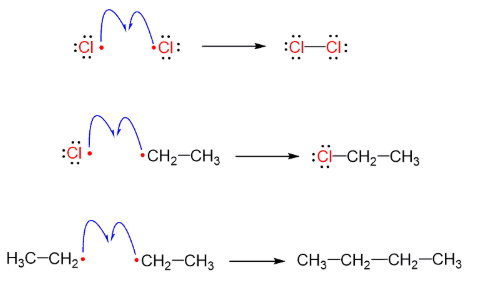
termination (free radial halogenation or organohalides)
reactant: radicals
products: stable product (halogenated)
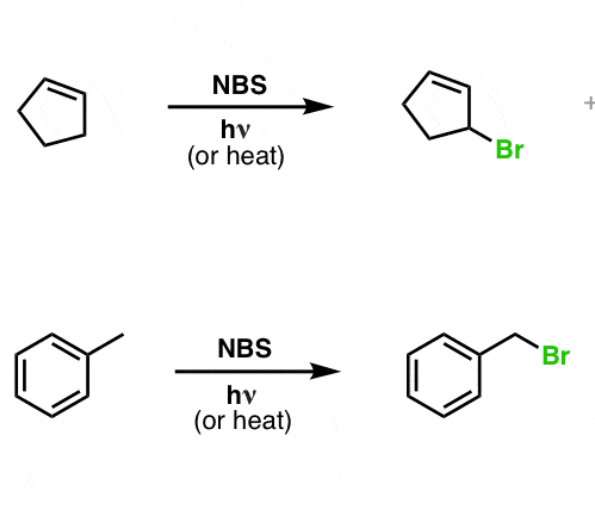
synthesis of alkyl halides from alkenes
reactant: alkene
reagent: NBS, hJ(light)
product: allylic bromide
allylic means C next to double bond
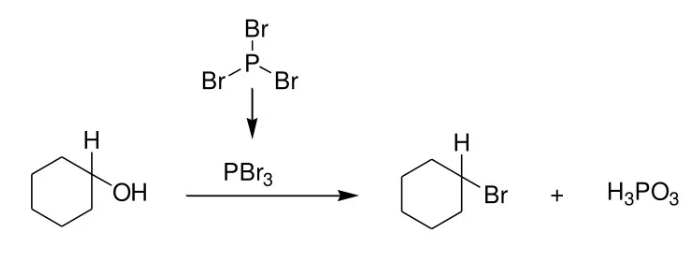
prepare alkyl halides from alcohols
reactant: alcohol
reagent: PBr3, X, HX (X=Br or Cl)
product: alkyl halide