Electron configuration
1/3
Earn XP
Description and Tags
https://www.youtube.com/watch?v=9ogq50CBgCg
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
4 Terms
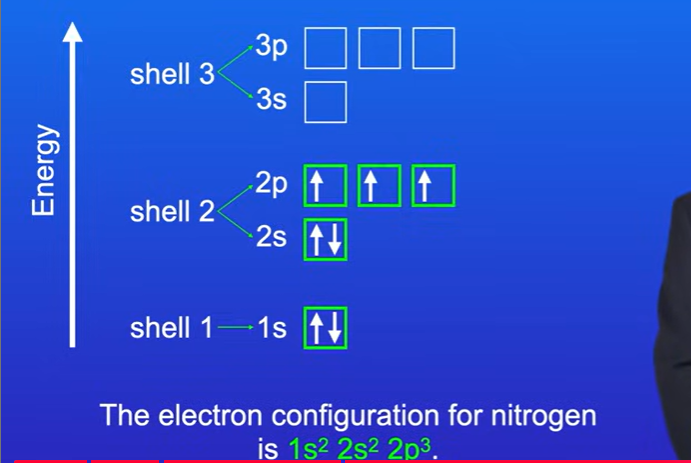
Rules for filling atomic orbitals
Orbitals with the lowest energy are filled first
We can have up to two electrons in the same orbtials, must have opposite spins.
If we have orbitals with the same energy, then we can put electrons into individual orbitals before we pair them. That is because electrons in the same orbital repel.
Whats the electron configuration for nitrogen
The first 2 electrons go into 1s subshell, The next 2 electrons go into the 2s subshells.
All of the p orbitals in the 2p subshell have the same energy . So each electron goes into individual orbitals in the 2p subshell. Electron configuration for nitrogen is 1s² 2s² 2p³
a
a
a
a