ATI Teas 7 Science Study Guide (Nurse Cheung)
1/288
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
289 Terms
changes between states of matter
condensation = gas to liquid; molecules of the gas slow down and become closer together
evaporation = liquid to gas; molecules of the liquid gain enough energy to break away from the surface of the liquid
sublimation = solid to gas; molecules of the solid gain enough energy to break away from the surface of the solid
deposition = gas to solid; molecules of the gas slow down and become closer together
melting = solid to liquid; molecules of the solid gain enough energy to break away from each other.
freezing = liquid to solid; molecules of the liquid lose enough energy to stay together
empirical evidence
information that is gathered through observation and experimentation
qualitative evidence
described in terms of quality or characteristics
quantitative evidence
described in terms of quantity or amount
reliable evidence
information that can be trusted and is consistent
valid evidence
information that accurately represents what it is supposed to represent
placebo
harmless substance that has no therapeutic effect
independent variable
variable that is being tested and is not affected by other variables
controlled variable
variable that is not being tested and is held constant
scientific method
First, identify a problem or question.
Second, gather information about the problem or question.
Third, form a hypothesis, which is a possible answer to the problem or question.
Fourth, design and conduct an experiment to test the hypothesis.
Fifth, analyze the data from the experiment and draw conclusions.
Sixth, communicate the results of the experiment.
proton
particle with a single positive charge
neutron
neutron is a particle with no electric charge
electron
particle with a single negative charge
isotopes
atoms of the same element that have a different number of neutrons
atomic mass
atom is the total number of protons and neutrons; Carbon-12 (number of 6 protons + the number of 6 neutrons)
atomic number
number of protons in an atom
atoms charge
more electrons than protons = negative charge
more protons than electrons = positive charge
protons and electrons are equal = neutral
ions
atoms that have gained or lost protons, and as a result, have a charge either positive or negative
cations
atoms that have lost one or more electrons; positively charged ions
anions
atoms that have gained one or more electrons; negatively charged ions
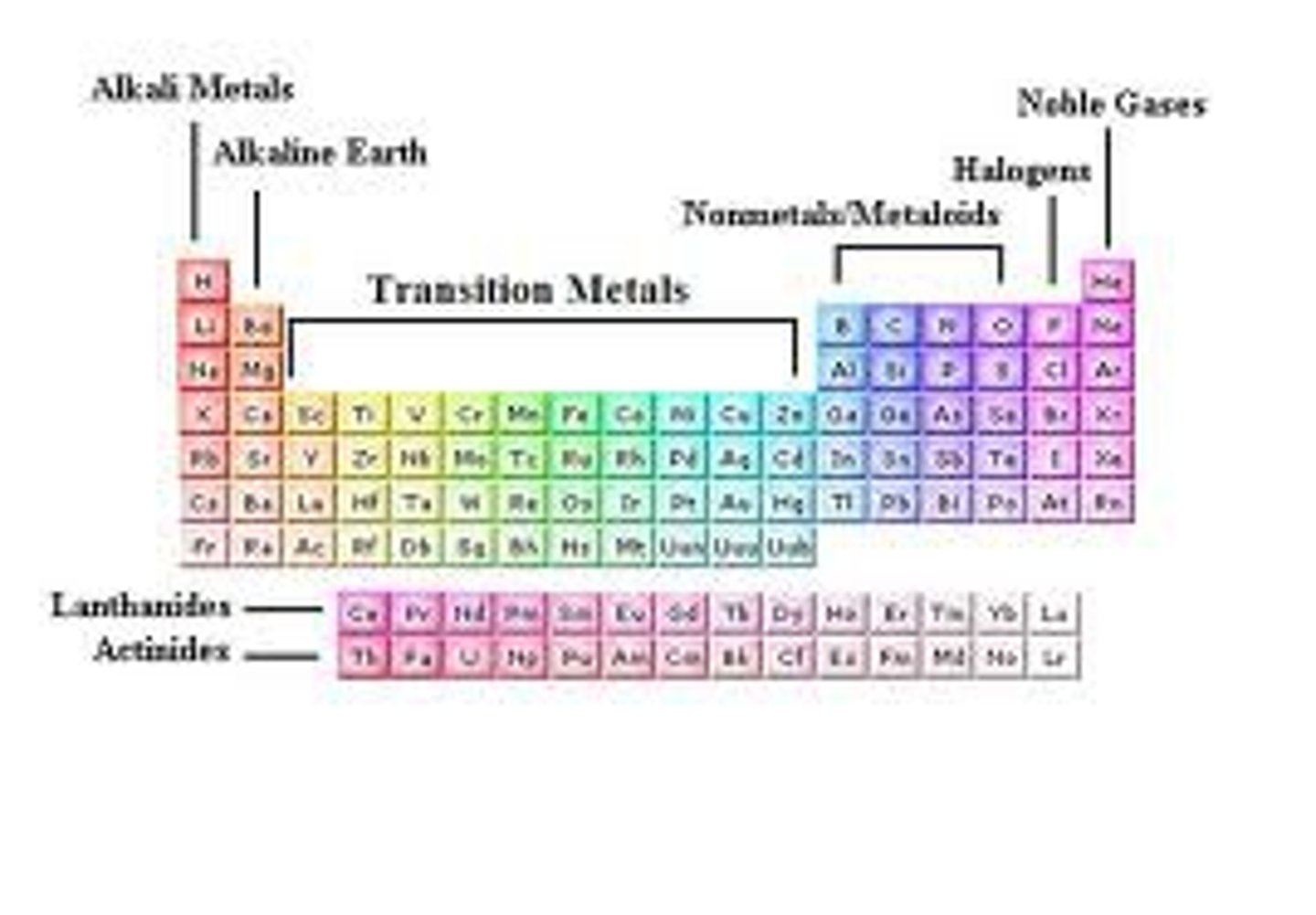
periodic table
columns = groups
rows = periods
orbitals
s = 2
p = 6
d = 10
f = 14
ionic bonds
atoms trade electrons in order to achieve stability
covalent bonds
atoms share electrons in order to achieve stability
electronegativity
matter
anything that has mass and occupies space
physical properties
different properties of a substance that can change their state without changing the identity of the substance
states of physical properties
mass = amount of matter in an object; grams (g)
volume = amount of space an object occupies; liters (L)
density = mass of an object divided by its volume; grams per liter (g/L)
phases of a substance
temperature = measure of the average kinetic energy of the molecules in a substance; higher temperature = more energy and more space they take up
pressure = measure of the force exerted on an object by the surrounding atmosphere; higher pressure = molecules are forced together
states of matter
solid = definite shape and volume; molecules packed together in a tight pattern
liquids = definite volume but take the shape of their container; molecules are not packed together as tightly
gases = no definite shape or volume; molecules are far apart from each other
plasma = state of matter found in the stars; electrons have been stripped from the atoms
valence electrons
outermost energy level of an atom
reactants
substances involved in the reaction
products
new substances formed by the reaction
combustion reaction
type of reaction that happens when a substance reacts with oxygen (O) to form carbon dioxide (CO) and water (H)
coefficient
number that is placed in front of a symbol or formula in order to multiply it
mole
unit of measurement of amount of a substance
catalysts
speeds up the reaction; lower the activation energy by providing an alternative pathway for the reaction to occur
endothermic reactions
absorb heat
exothermic reactions
release heat
dynamic equilibrium
forward and reverse reactions are occurring at the same time
static equilibrium
concentrations of the reactants and products are not changing
activation energy
minimum amount of energy that is needed for a chemical reaction to occur
cohesion
similar molecule surrounding and binding to another molecule; water with water
adhesion
dissimilar molecules binding to another molecule; water with glass
solvent
substance that dissolves in another substance
solute
substance that dissolves in a solvent
hydrophilic
attracts water molecules
hydrophobic
repels water molecules
solubility
ability of a solute to dissolve in a solvent
concentration of a solution
amount of solute that is dissolved in a given amount of solvent
dilution
adding solvent to a solution to decrease the concentration of the solution
molarity
unit of measurement that describes the concentration of a solute in a solution;
mole of solute/liters of solution (mol/L)
osmosis
water molecules moving from an area of high water concentration to an area of low water concentration through a semi-permeable membrane
hypertonic
net flow of water out of cell
hypotonic
net flow of water into cell
semi-permeable membrane
barrier that allows some molecules to pass through but not others
diffusion
molecules moving from an area of high concentration to an area of low concentration
passive transport
process of molecules moving that do not require energy, only require that the molecules be in motion
active transport
process of molecules moving that requires energy (moving against the concentration gradient), from an area of low concentration to an area of high concentration
excessive hydrogen ions
CO2 + H2O ↔ H2CO3
HCO3− + H+ ↔ H2CO3
acid
molecule that increases the concentration of hydrogen ions in a solution; pH = 0-6.9
base
molecule that decreases the concentration of hydrogen ions in a solution; pH = 7.1-14
neutral
pH = 7
buffer
resists change in pH
neutralization reactions
chemical reaction between an acid and a base
chemicals
help build cells
macromolecules
chemicals that are essential to life; carbohydrates, proteins, lipids, and nucleic acids
cell
basic unit of life; all living things are made up of cells
tissues (4 types and functions)
made up of cells that have a similar structure and function;
types = epithelial, connective, muscle, nervous
functions = protection, support, movement, communication
organs
made up of tissues that work together to carry out a specific function
organ systems (11 systems)
group of organs that work together to carry out a specific function;
integumentary
skeletal
muscular
nervous
endocrine
cardiovascular
lymphatic
respiratory
digestive
urinary
reproductive
organisms
made up of one or more organ systems
cell is composed of 10 parts:
cell membrane
cytoplasm
Golgi Apparatus
lysosome
mitochondrion
nucleus, ribosomes
rough endoplasmic reticulum
smooth endoplasmic reticulum
vacuole
cell membrane
layer of protein and fat that surrounds the cell;
selectively permeable
cytoplasm
jelly-like substance that contains the cell's organelles;
supports and suspends the structures inside the cell membrane;
transfers material required for other cellular processes
Golgi apparatus
organelle that helps to package and transport molecules within the cell;
helps process proteins and lipid molecules
lysosomes
organelles that contain enzymes that break down food and other molecules;
aid in digestion and recycle old cell materials;
destroy invading bacteria and viruses
mitochondria
organelles that produce energy for the cell;
convert nutrients into ATP (cell's energy source)
nucleus
organelle that contains the cell's hereditary information
deoxyribonucleic acid (DNA)
responsible for the cell's growth, reproduction, and function;
genes that are coded with instructions to produce proteins in the cell;
made up of two long chains of nucleotides that twist to create a double helix
ribosomes
organelles that help synthesize proteins = essential for the cell to carry out its functions;
round on either the rough-endoplasmic reticulum or floating in the cytoplasm
rough endoplasmic reticulum (RER)
organelle that helps to package and transport molecules within the cell;
involved in the synthesis of proteins
smooth endoplasmic reticulum (SER)
organelle that helps to package and transport molecules specifically lipids within the cell but does not contain ribosomes;
involved in carbohydrate metabolism and inactivates toxins along with harmful metabolic products
vacuole
organelle that stores food, water, and other materials;
helps to maintain the cell's shape
mitosis
process of cell division that results in two genetically identical daughter cells.
mitosis (4 cell cycle stages)
interphase
prophase
metaphase
anaphase
telophase
interphase (mitosis)
first stage of the cell cycle;
cell grows and carries out its normal functions;
DNA starts to replicate
prophase (mitosis)
second stage of the cell cycle;
chromosomes condense and become visible;
nuclear envelope breaks down
metaphase (mitosis)
third stage of the cell cycle;
chromosomes line up in the middle of the cell
anaphase (mitosis)
fourth stage of the cell cycle;
chromosomes are pulled apart to opposite sides of the cell;
cell division begins
telophase (mitosis)
fifth and final stage of the cell cycle;
new nuclear envelope forms around the chromosomes;
chromosomes uncoil and become less visible;
cell divides into two daughter cells
meiosis
cell division that results in four genetically diverse daughter cells
meiosis (2 main stages)
meiosis I:
interphase
prophase I
metaphase I
anaphase I
telophase I
meiosis II:
prophase II
metaphase II
anaphase II
telophase II
meiosis I:
interphase
prophase I
metaphase I
anaphase I
telophase I
Interphase = first stage of meiosis; cell grows and carries out its normal functions; DNA starts to replicate.
Prophase I = second stage of meiosis; homologous chromosomes pair and start to crossover.
Metaphase I = third stage of meiosis; homologous chromosomes line up in the middle of the cell in pairs
Anaphase I = fourth stage of meiosis; one chromosome from each homologous pair is pulled apart to opposite sides of the cell
Telophase I = fifth stage of meiosis; two daughter cells start to form, each cell has half the number of chromosomes as the original cell (mixture of genetic information)
meiosis II:
prophase II
metaphase II
anaphase II
telophase II
Prophase II = sixth stage of meiosis; daughter cells contain half of the chromosomes from the original cells
Metaphase II = seventh stage of meiosis; chromosomes line up in the middle of the cell again
Anaphase II = eighth stage of meiosis; sister chromatids are pulled apart to opposite sides of the cell
Telophase II = ninth and final stage of meiosis; cells divide into four genetically diverse daughter cells (haploids)
chromosomes
long, thread-like structures found in the nucleus of cells made up of DNA and histone proteins; winding structure condenses DNA and allows regulation
prokaryote = one chromosome
eukaryote = multiple chromosomes
genes
basic unit of heredity; made up of DNA and are responsible for the characteristics of an organism; passed down from parent to offspring
structural genes
responsible for the physical traits of an organism
regulatory genes
control the activity of other genes
complementary bases
hydrogen bonds pair up to hold the two strands of DNA together;
G to C and T to A (DNA);
G to C and U to A (RNA)