EPIB301: Exam 2 Prep
1/86
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
87 Terms
What are the characteristics of experimental studies?
Manipulations of variables; assigning participants to exposure group, only use when feasible and ethical
What are the examples of experimental studies?
Quasi-Experimental and Randomized Control trial (RCT)
What are the characteristics of observational studies?
Investigator watches, and there is no manipulation or exposure. Used when an experimental study is not feasible
What are the examples of observational studies?
Cohort, case control, and cross-sectional
What are the types of intervention designs?
Clinical trials and community trials
What is the focus of clinical trials?
Individuals
What is the focus of community trials?
Group or community
What is a community trial (community intervention)?
Intervention that is allocated to entire communities
What are the characteristics of randomization (Randomized Control Trial/RCT)?
Each participant has the same probability of being put into the treatment group
Eliminate bias in treatment assignment
Confounding factors will be balanced between the treatment and control groups, making these groups directly comparable
What is the ultimate goal of randomization (RCT)?
Ensure comparability between study groups by balancing known and unknown confounding factors
What does binding/masking mean?
Participants and/or researchers are unaware which subjects is getting the treatmment or placebo
What are the phases of a clinical trial summarized?
Start with small and specific samples, then gradually grow larger and broader. Continue monitoring after FDA approval
What is phase 1 of clinical trials?
Tests drug on healthy individuals, tests for safety dosage and side effects
What is phase 2 of clincal trials?
Tests on larger group of effected individuals, tests for efficacy and side effects
What is phase 3 of clinical trials?
Tests on new and wider demographic, tests for long term effectiveness and comparisons with other medications
What is the FDA approval phase of clinical trials?
Treatment determined effective and safe for public use
What is phase 4 of clinical trials?
Continues to test for effectiveness and safety, can be taken off the market if necessary
What is similar to clinical trials?
Cohort studies
What is the formula of RR (Relative risk)?
A/(A+B) divided by C/(C+D)
If RR = 1, what does that indicate?
no association
If RR > 1, what does that indicate?
treatment group more likely to develop the outcome
If RR < 1, what does that indicate?
treatment group less likely to develop outcome
If the outcome is improvement, a RR > 1 would mean what?
Treatment group is more likely to improve;
If the outcome is improvement, a RR < 1 would mean what?
treatment group is less likely to improve
What is efficacy in terms of epidemiology?
The proportion of individuals in the control group who experience an unfavorable outcome who could have been expected to have a favorable outcome had they been in the active group instead
What indicates that an intervention was successful: Low efficacy or high efficacy?
High efficacy
What does NNT stand for?
Number Needed to Treat
What does NNT mean?
The expected number of people who would have received a treatment to prevent an unfavorable outcome in one person
What indicates a more effective intervention: a small NNT or a big NNT?
Small NNT
A participant is given a “fake” intervention.
Participant believes this intervention will change or improve his/her condition.
This belief can have a therapeutic effect.
Sometimes this belief improves their condition in a measurable way
What is this phenomenon known as?
Placebo effect
Why do experimental studies involve a high level of ethical risk?
Participants do not choose whether they get assigned to exposures (treatments)
What is the list of ethical considerations for experimental studies?
Informed consent (plan language, multiple languages)
Withholding treatment known to be effective
Protecting the interests of the individual patient
Monitoring for side effects
Deciding when to withdraw a patient
What are these the strengths of?
Provides the strongest evidence of association
Manipulation means having control over:
Amount of exposure
Timing and requency of exposure
Period of observation
Ability to randomize reduces the likelihood that group will differ significantly
Experimental designs
What are these the strengths of?
They represent the only way to estimate the impact of change in behavior or modifiable exposure on the incidence of disease.
Manipulation of intervention and period of observation
Ability to randomize at community level
Community trials
What are these the limitations of?
They are inferior to clinical trials with respect to the ability to control entrance into the study, delivery of the intervention, and monitoring of outcomes
Fewer study units are capable of being randomized, which affects comparability
They are affected by population dynamics, secular trends, and nonintervention influences
Community trials
What are these the limitations of?
Artificial setting
Limited scope of potential impact
Adherence to protocol is difficult to enforce
Expensive
Ethical concerns
Experimental designs
Name this study design:
To test the efficacy of vitamin C in preventing colds, army recruits are randomly assigned to two groups: one given 500 mg of vitamin C daily, and one given a placebo. Both groups are followed to determine the number and severity of subsequent colds.
Randomized control trial
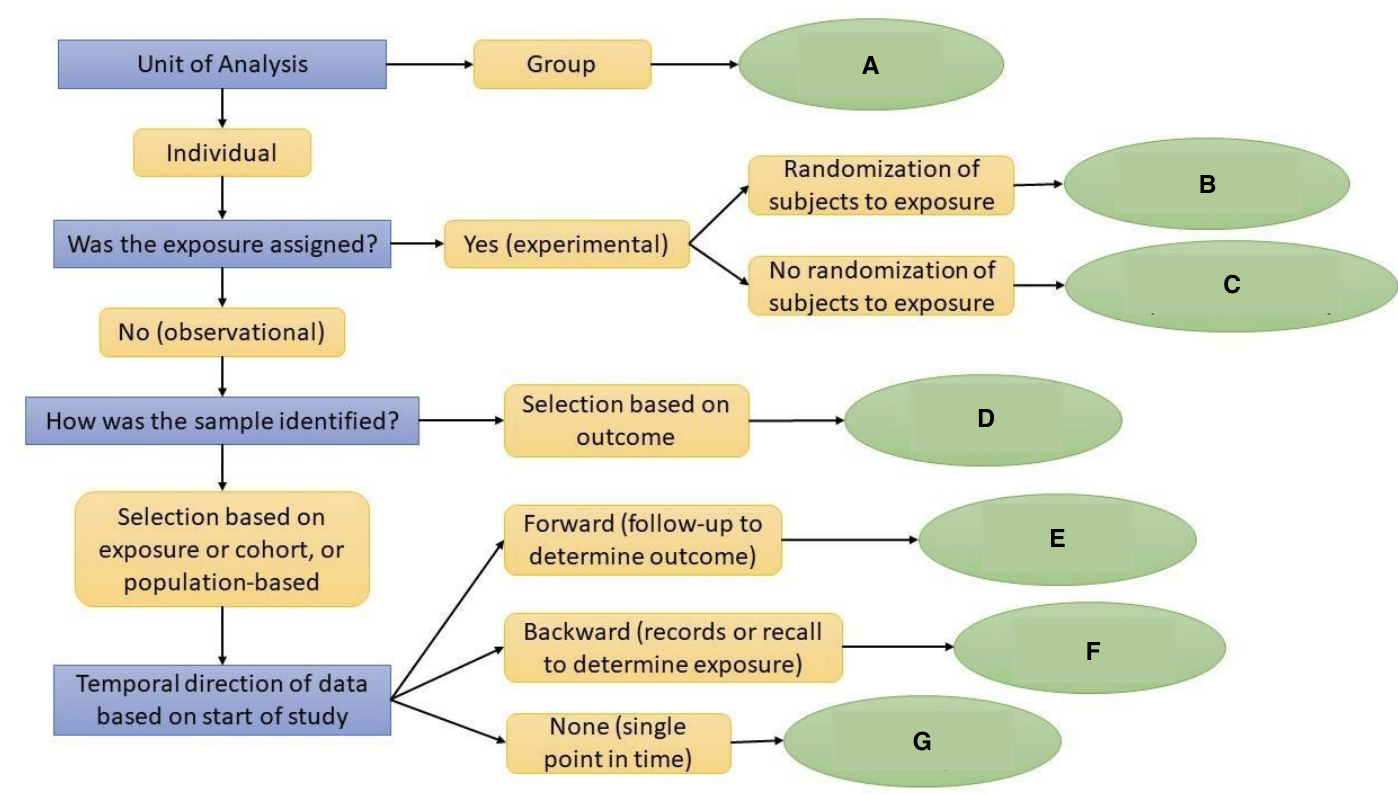
What is A?
Ecological Study
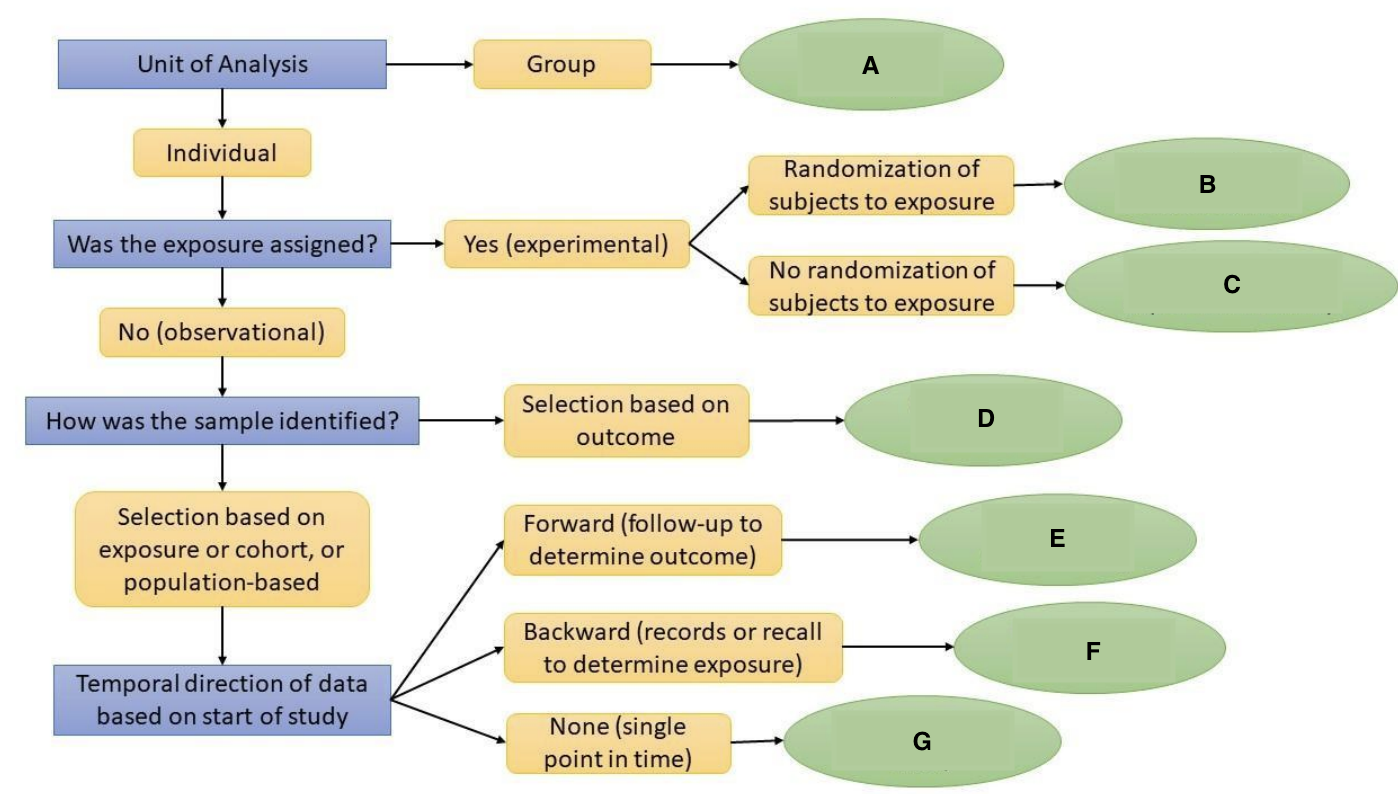
What is B?
Randomized Controlled Trial
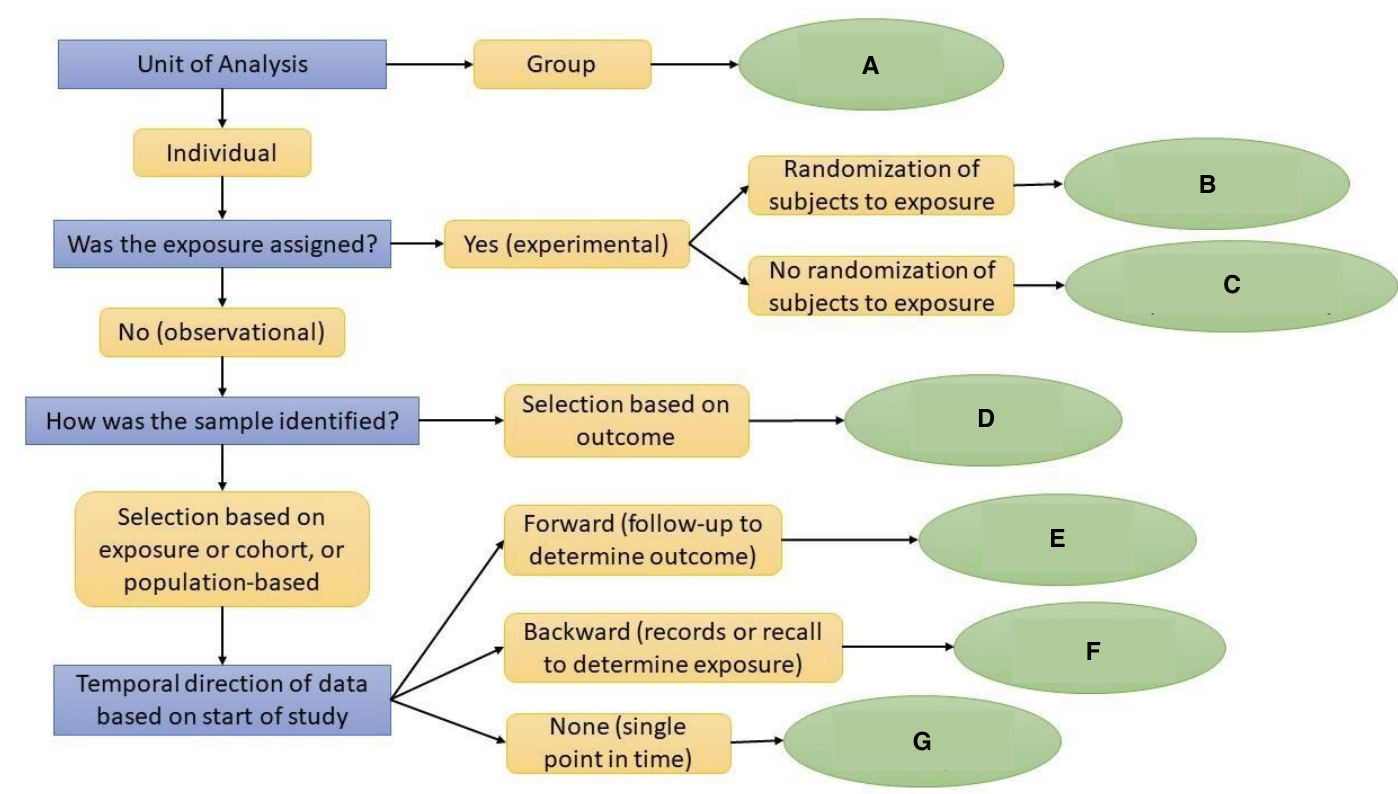
What is C?
Quasi-experimental Study
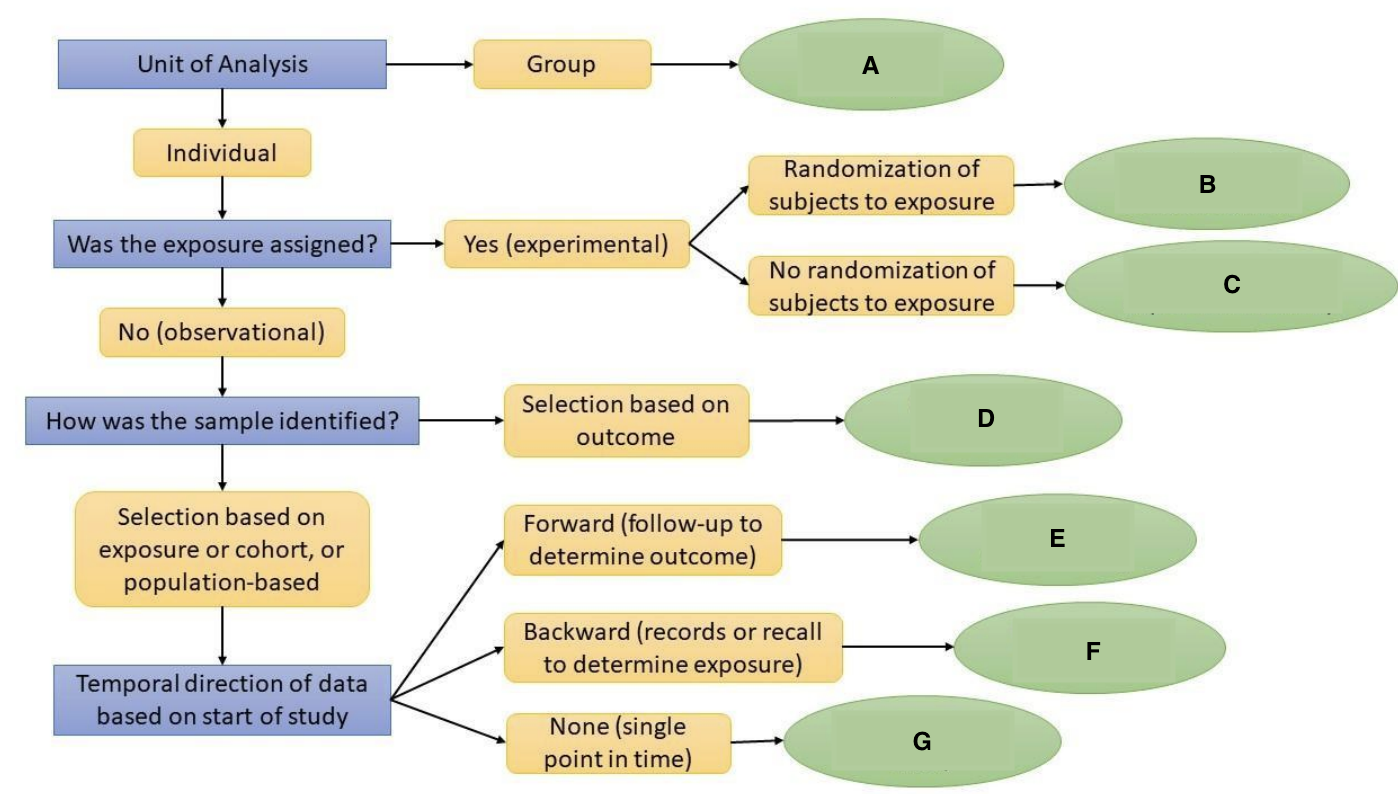
What is D?
Case-Control Study
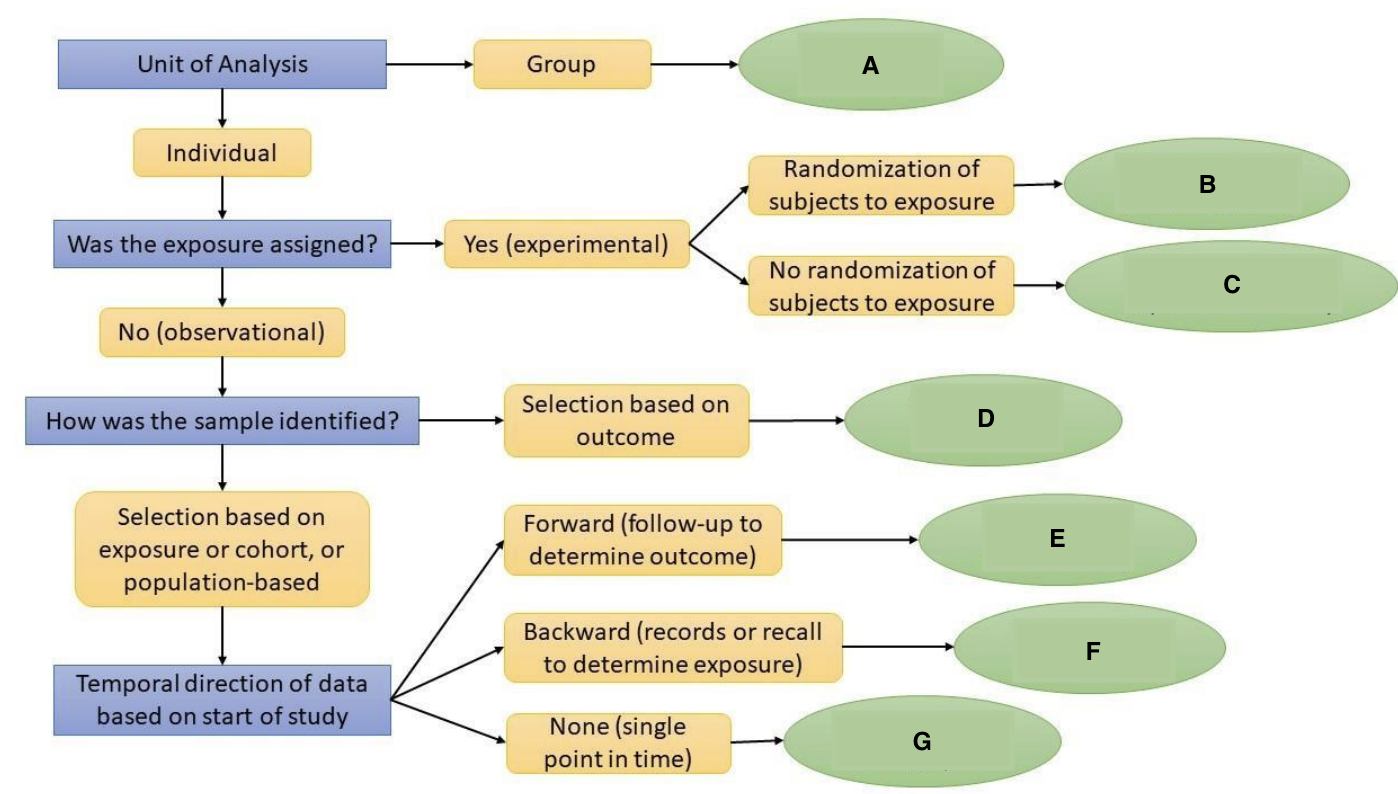
What is E?
Prospective Cohort Study
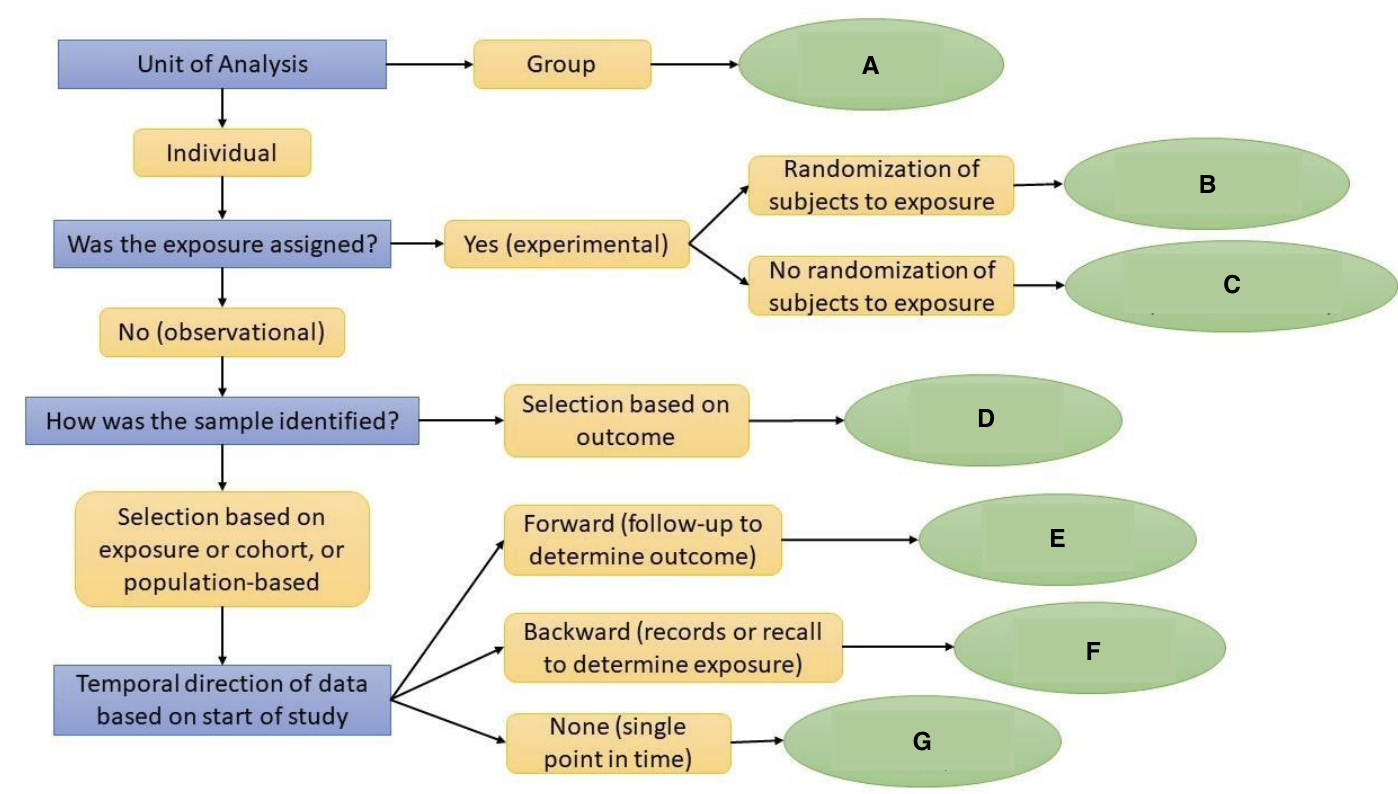
What is F?
Restrospective Cohort Study
What is G?
Cross-Sectional Study
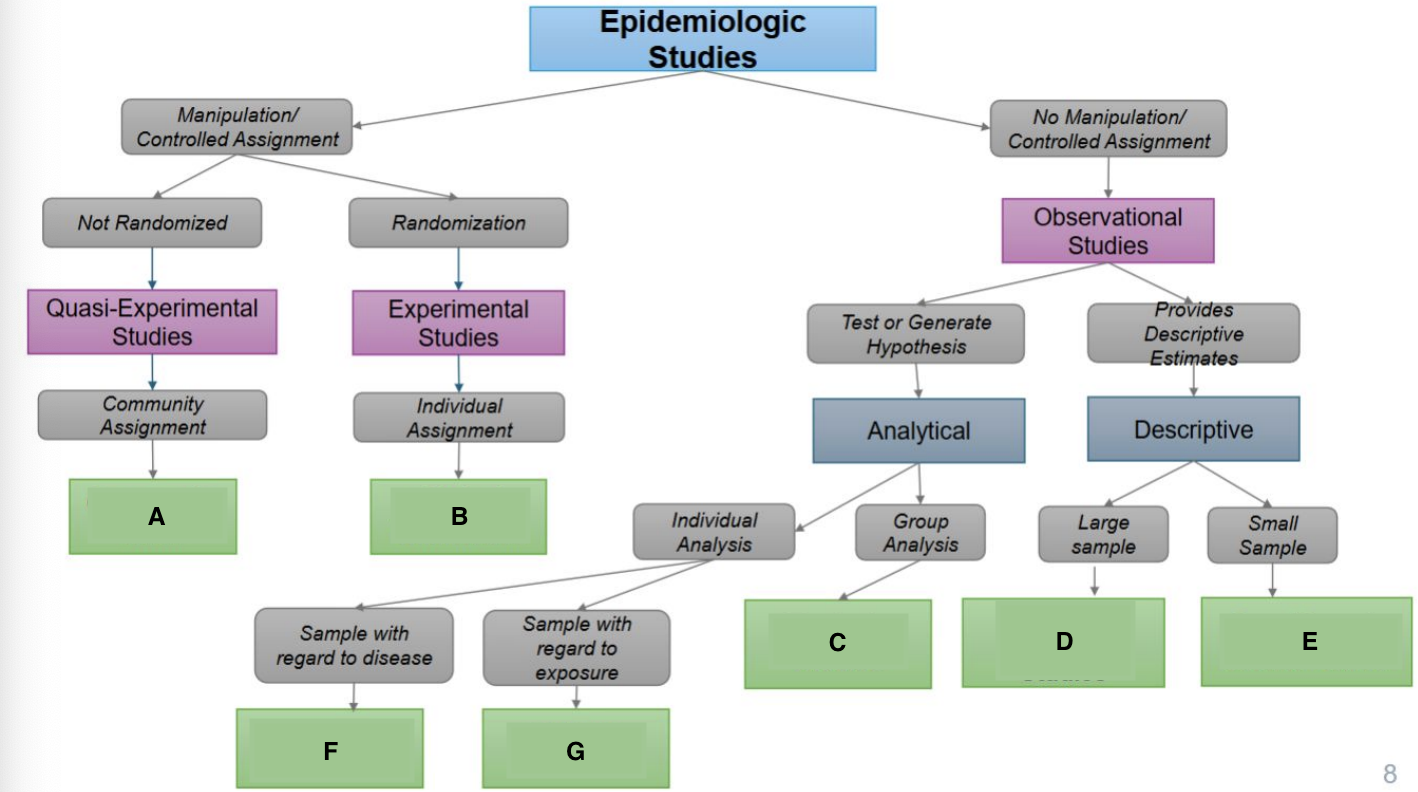
What is A?
Community trials
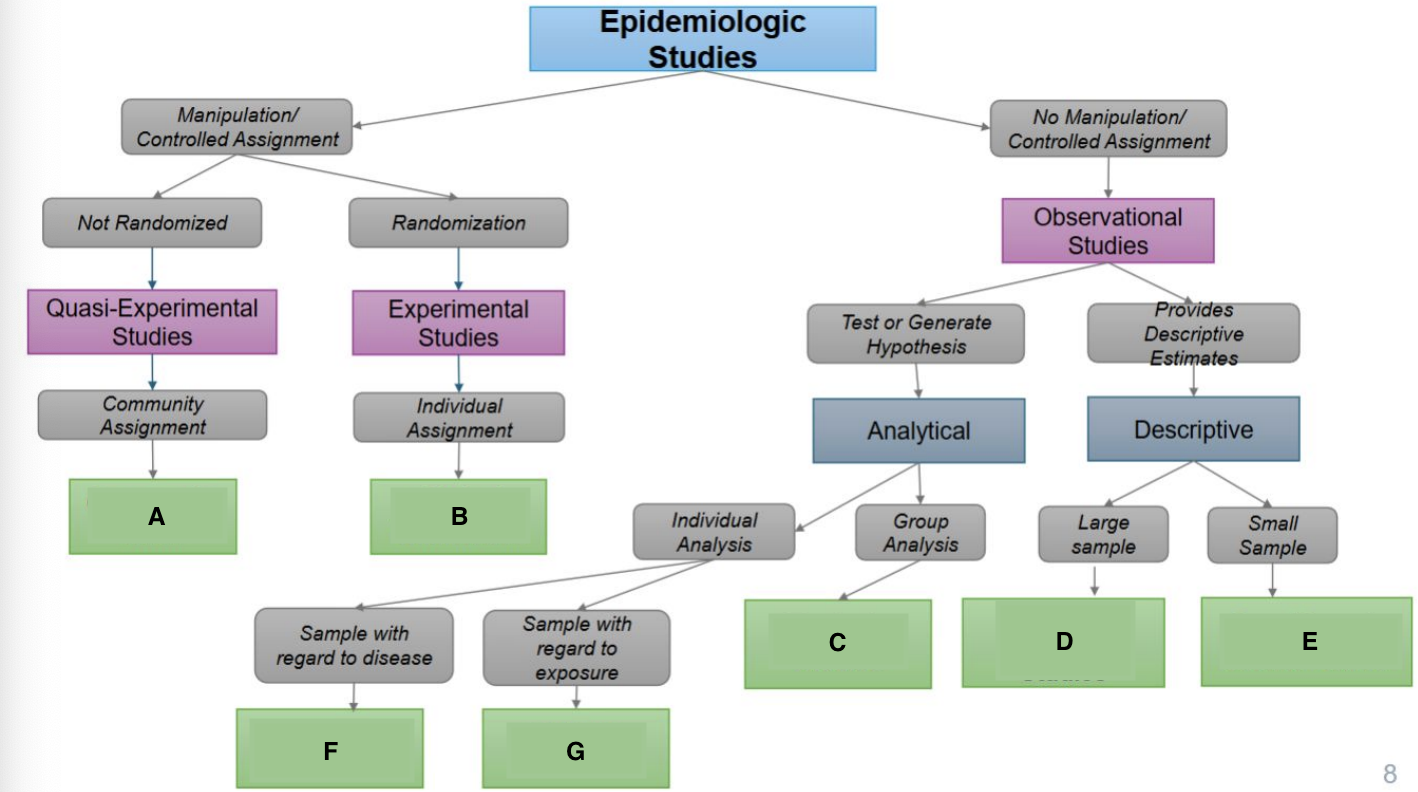
What is B?
Clinical Trials/RCTS
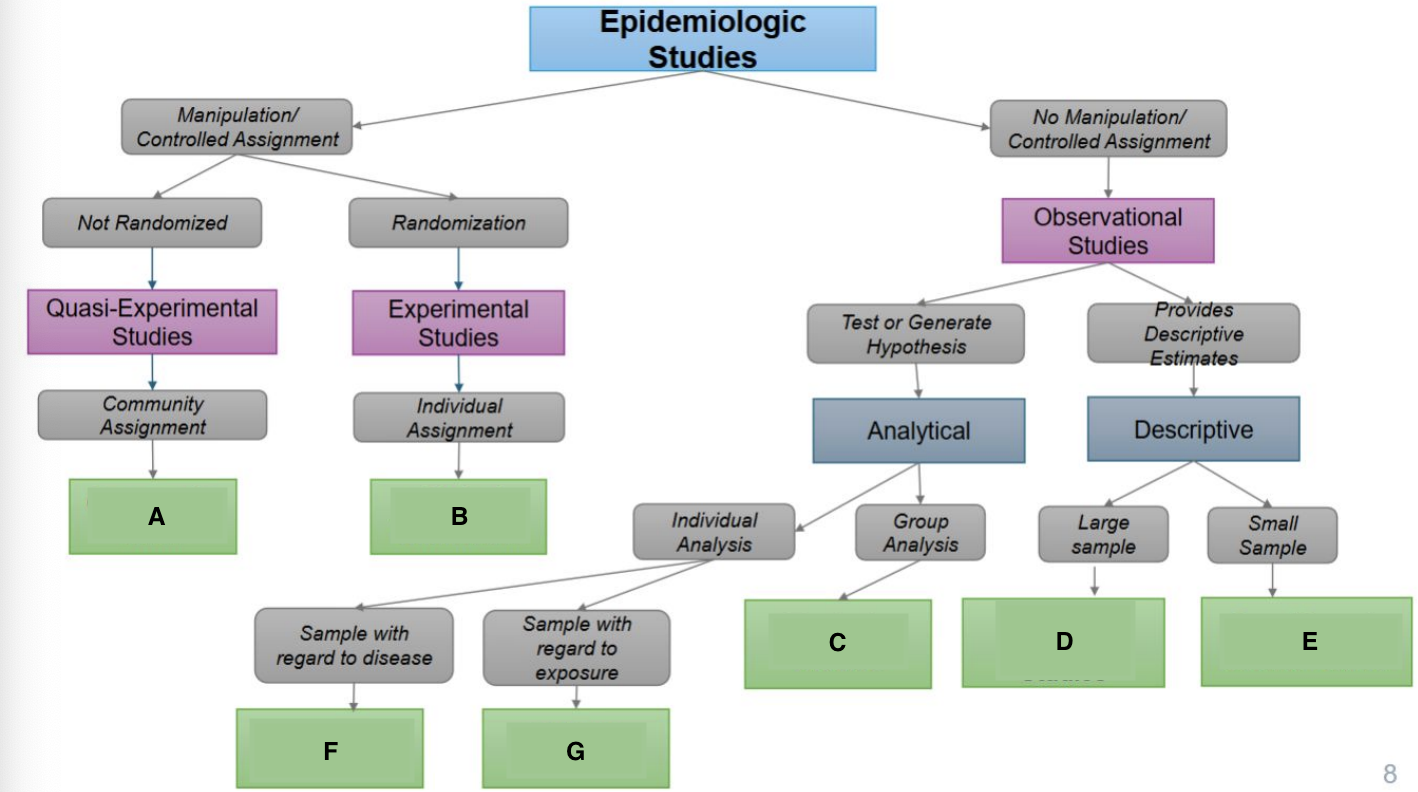
What is C?
Ecological Studies
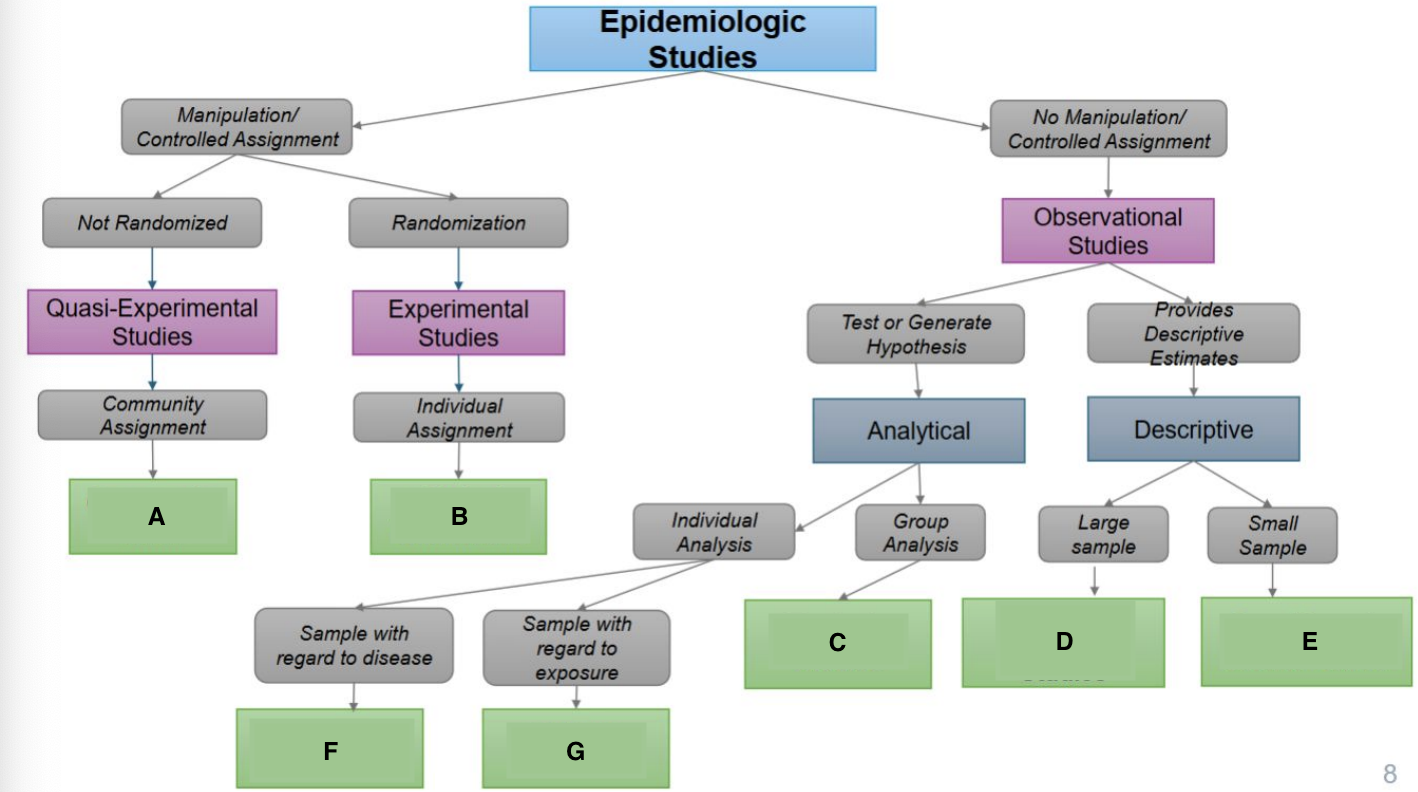
What is D?
Cross-sectional Studies
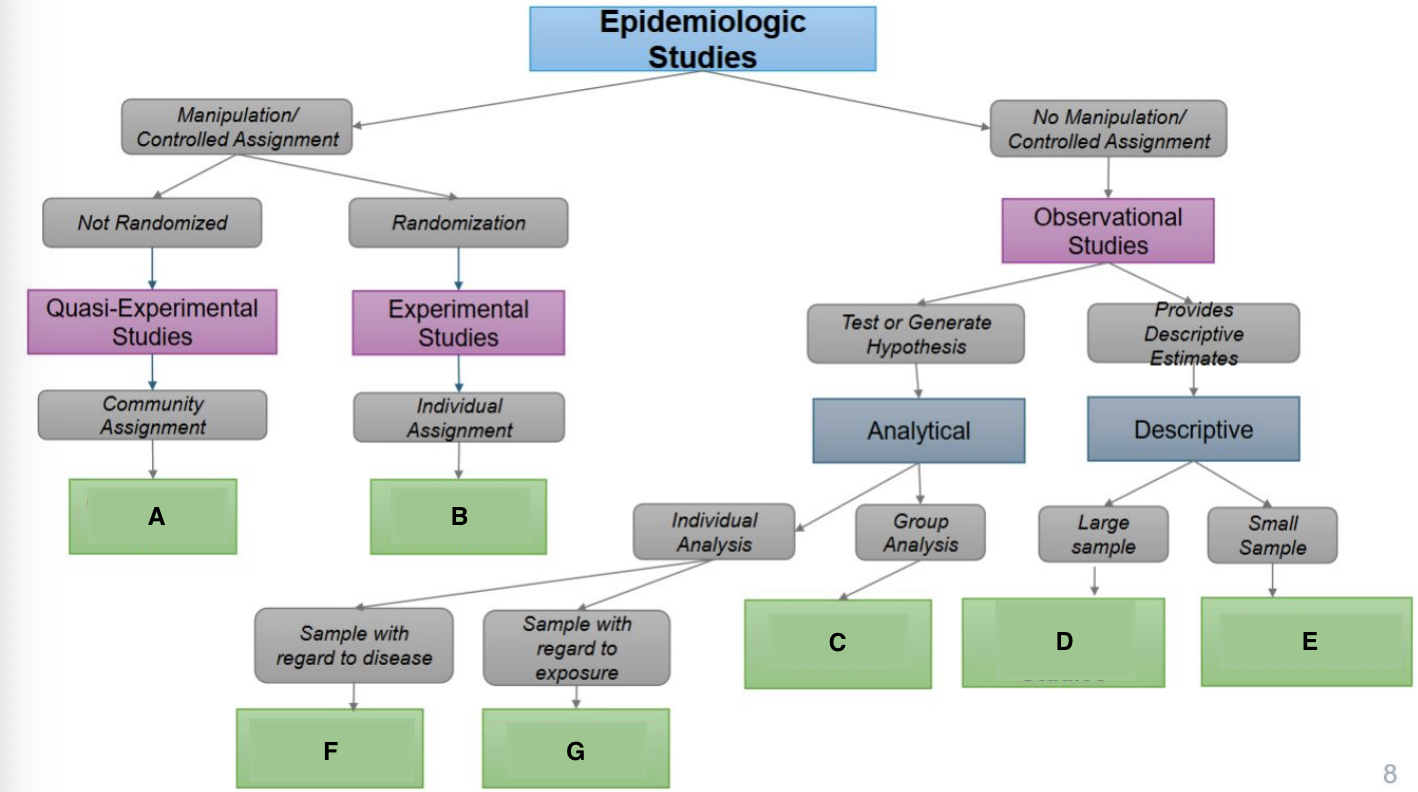
What is E?
Case Reports & Case Series
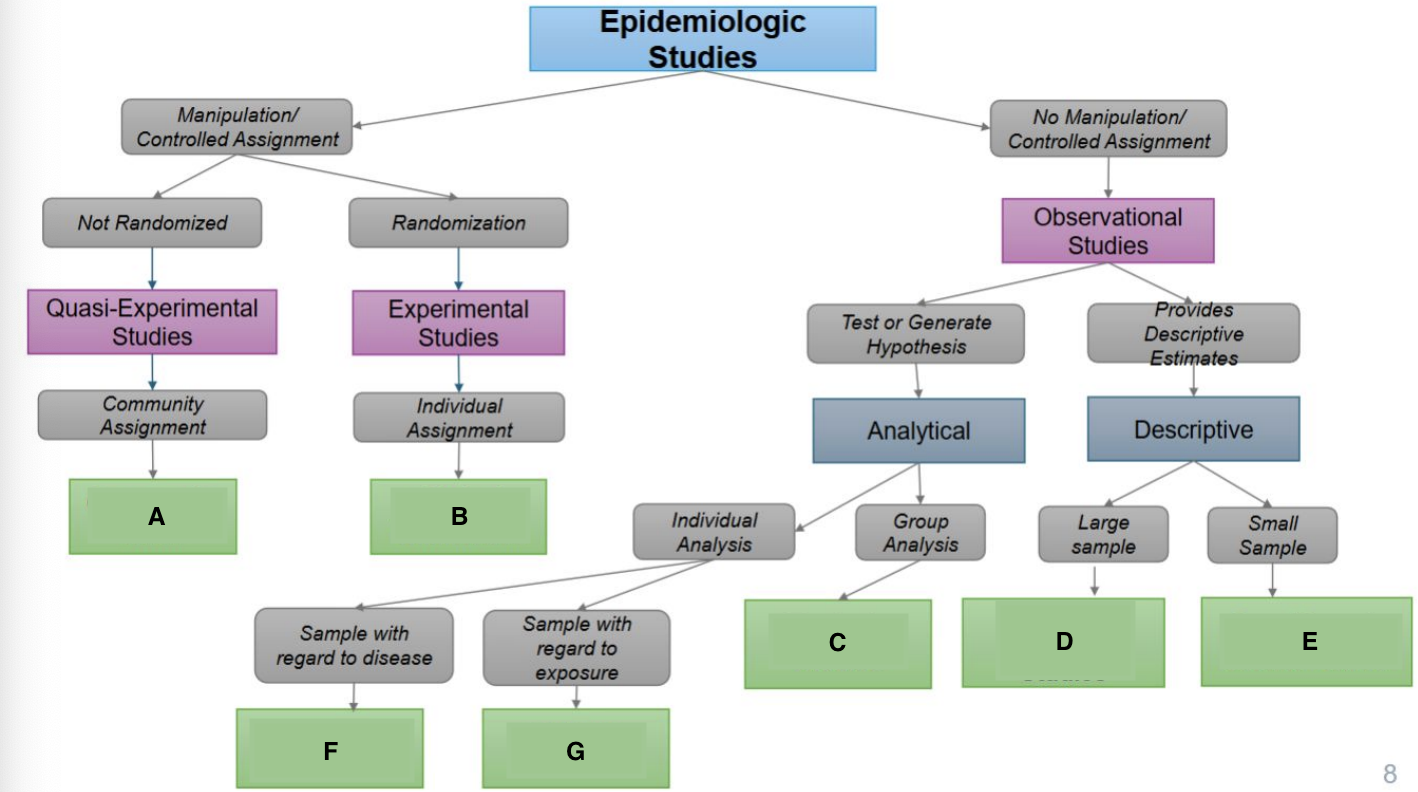
What is F?
Case Cohort Studies
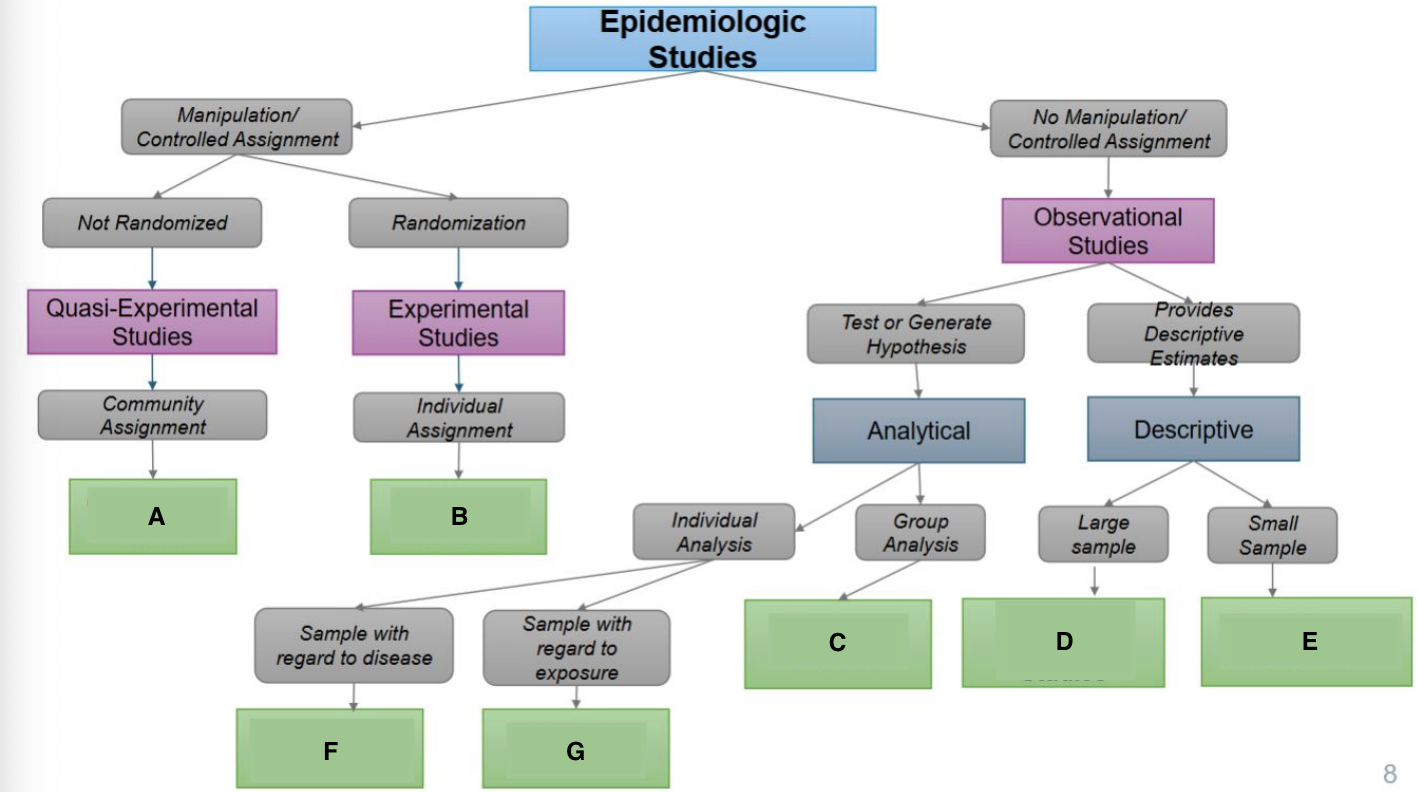
What is G?
Cohort Studies
What fits experimental trials but not a prospective cohort study?
Exposure status is assigned by the researcher(s) into treatment and control groups (experimental)
What is the similary between experimental trials and prospective cohort study?
Outcome has not yet occured at the start of the study for any of the participants
What is the similarity between a prospective cohort study and a retrospective cohort study?
Participants selected based on exposure status/shared characteristics known as cohort (other than outcome)
What is the similarty between restrospective cohort study and case-control study?
Outcome status of participants is known at the start of the study
What fits case-control study but not a retrospective cohort study?
Participants selected based on outcome/disease status
What is a prospective cohort study?
A study of healthy participants who are followed forward in time to observe the development of an outcome
What is a retrospective cohort study?
Uses historical records or recall to determine exposure status at a specific point in the past, assess the already existing outcome
What are these the strengths of?
Collect information on multiple exposures and outcomes
Efficient for rare exposures
Temporality
Prospective Cohort Study
What are these the strengths of?
Efficient for disease with long latency periods
Temporality (when using good historical records, not guaranteed)
Low cost and quick
Retrospective Cohort Study
What are these the limitations of?
Expensive and time-consuming
Not practical for diseases with long latent periods
Prospective Cohort Study
What are these the limitations of?
May have poor information on exposures or other key variables
Only as good as the data (records) available
Retrospective Cohort Study
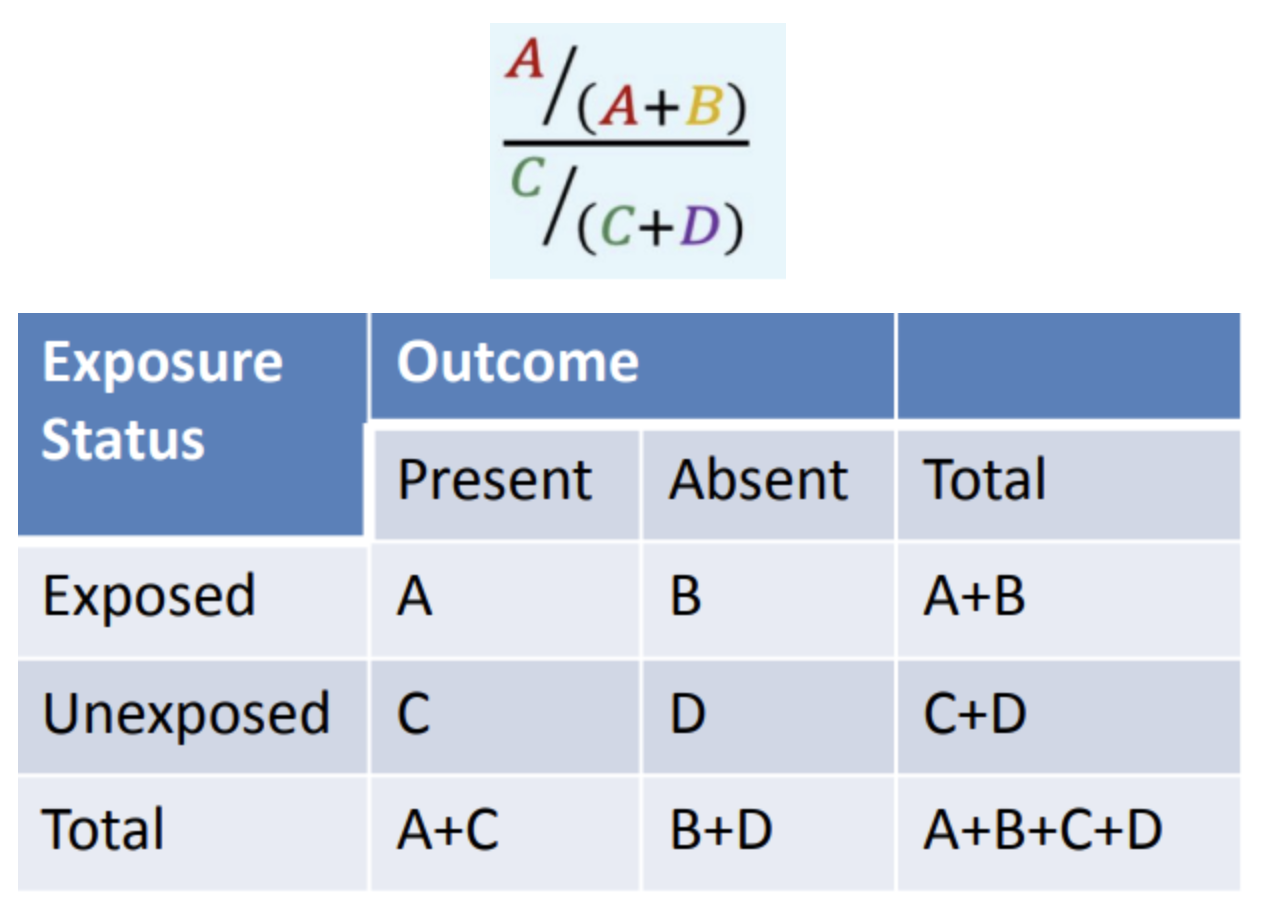
What is this formula for?
Relative Risk (RR)
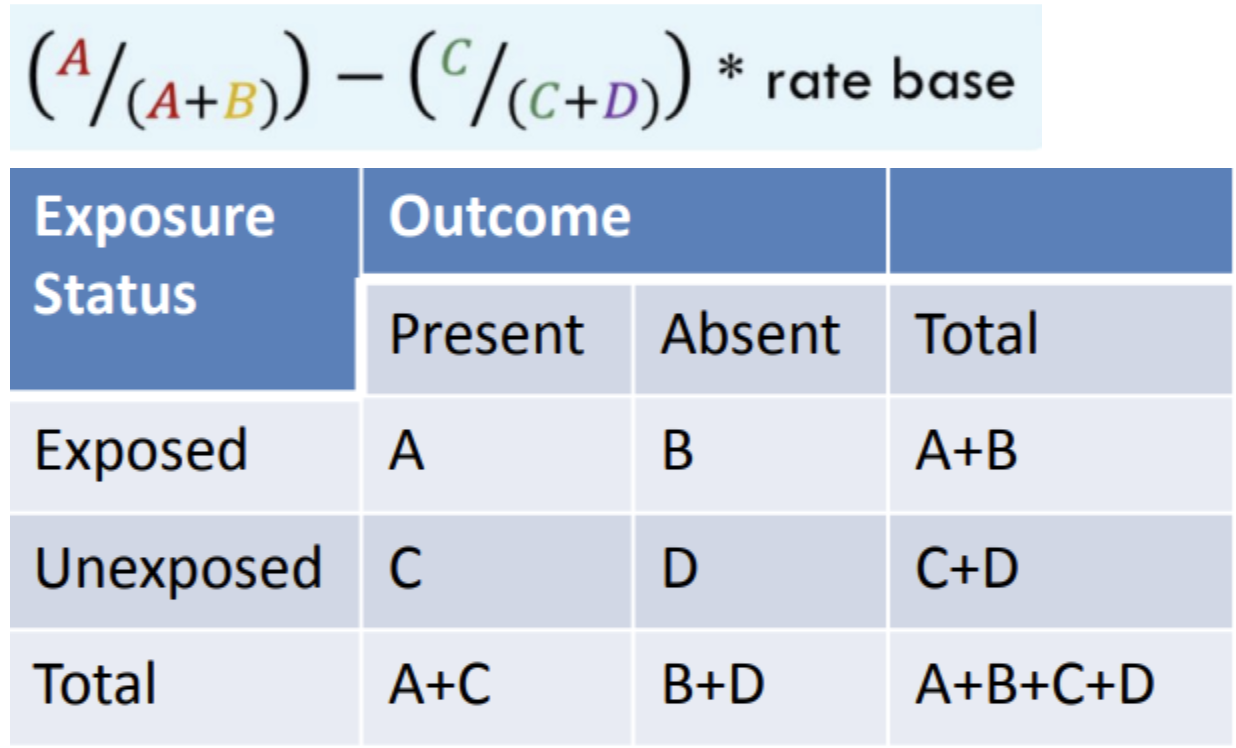
What is this formula for?
Risk Difference (Attributable risk)
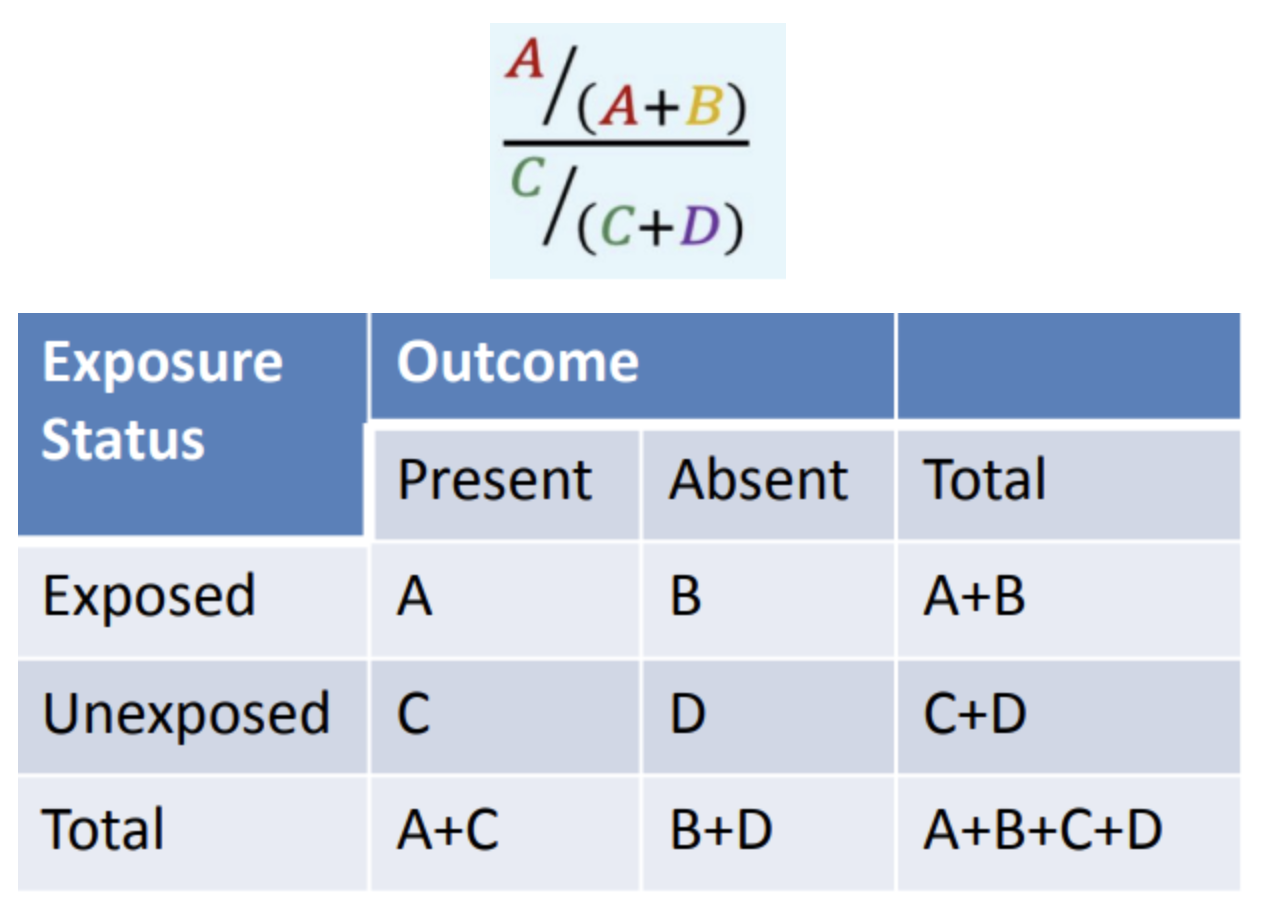
In the equation given, what does A/(A+B) mean?
Risk exposed
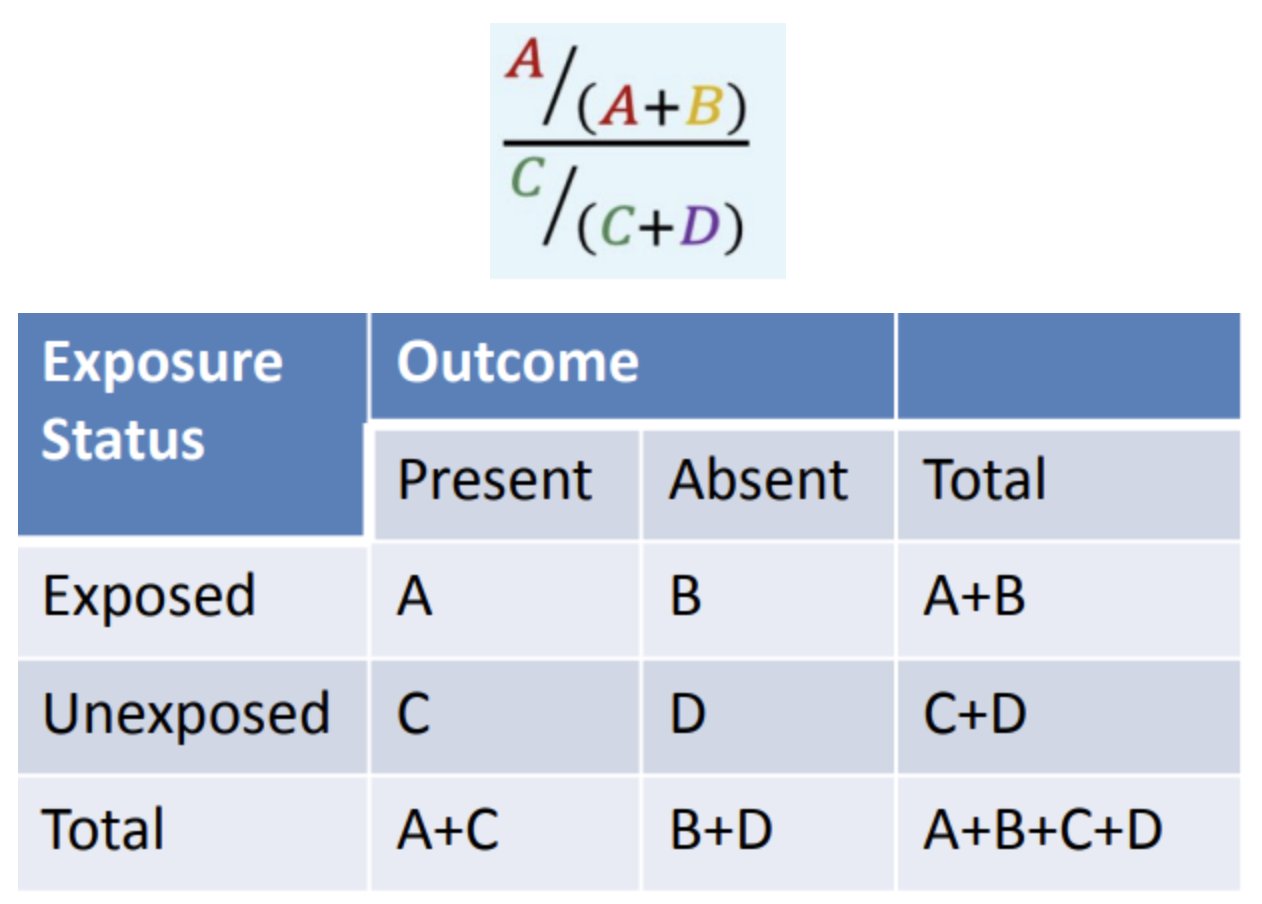
In the equation given, what does C/(C+D) mean?
Risk unexposed
What is the interpretation of relative risk > 1?
The risk of [outcome] in the [exposed group] is [RR] times higher than in the [unexposed group].
What is the interpretation of relative risk = 1?
The risk of [outcome] in the [exposed group] is the same as the risk of [outcome] in the [unexposed].
What is the interpretation of relative risk < 1?
The risk of [outcome] in the [exposed group] is [(1-RR)x100%] lower than in the [unexposed group].
What is the interpretation of risk difference?
[The exposed group] had [value] times higher risk of developing [outcome] compared to [the unexposed group].
What are these the strengths of?
Efficient for rare diseases and diseases with long induction or latent periods
Can evaluate multiple risk factors for the same disease so useful for poorly understood diseases
Typically inexpensive because they use a smaller number of subjects
Case-control Study
What are these the limitations of?
Inefficient for rare exposures
Vulnerable to recall bias because of the retrospective nature
Can only study one outcome
Case-control Study
What is odds ratio?
Odds of exposure among cases divided by the odds of exposure among controls
What is odds ratio used for?
Case-control Study
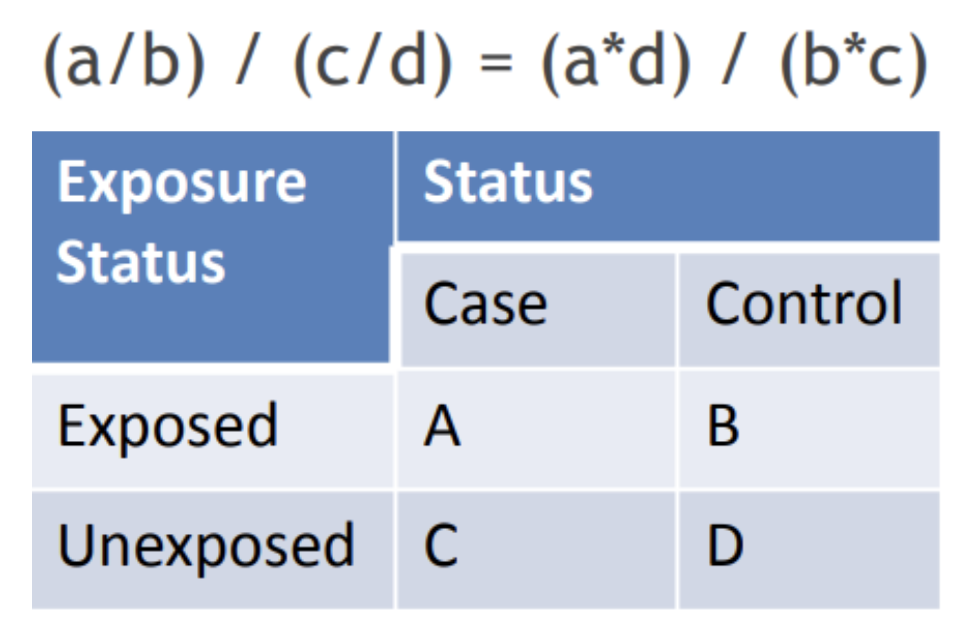
What is this equation?
Odds ratio
What is the interpretation of the odds ratio?
The odds of [exposure] among the [cases] is [#] times that of the [controls].
What are these the strengths of?
Measures prevalence (disease frequency)
Single point in time, so investigators do not have to follow up with individuals over time
Useful for establishing preliminary evidence for future planning of studies
Population-based so it helps investigators understand determinants of health
Cross-sectional Study
What are these the limitations of?
Cannot follow up or establish a causal relationship
Only 1 time measurement of exposure and no follow-up
Prone to bias
Selection bias: when study participants’ characteristics are systematically different compared to eligible participants not selected for the study
Cross-sectional Study
What are these the strengths of?
Low cost (typically using data that was already collected)
Useful to examine the rate of disease in relation to a population-level factor
Ecological Study
What are these the limitations of?
Ecological fallacy
Ecological Study
What is an ecological fallacy?
Drawing conclusions about indivduals based on data collected at the GROUP level
What is this an example of?
A specific neighborhood has a high crime rate, assume that a resident living in that area is more likely to commit a crime.
Ecological fallacy
What is this an example of?
Richer cities have higher rates of heart disease, assume that richer individuals have higher rates of heart disease than poorer individuals.
Ecological fallacy
Which of the following represents the main strength of case-control studies?
Efficiency
What is the study design?
In order to determine if vitamin D (the “sunshine” vitamin) is associated with reduced levels of ovarian cancer, an epidemiologist identified 12 geographic regions in Sweden and obtained data on the average annual number of days of sunlight and ovarian cancer rates for each region. The findings indicated that those regions with the highest average days of sunlight per year had significantly lower rates of ovarian cancer than those regions with the fewest average days of sunlight per year. The epidemiologist reported her findings at the Scandinavian Conference on Cancer.
Ecological Study
What is this study design?
Smoking histories are obtained from all patients entering a hospital who have lip cancer and are compared with smoking histories of other patients who enter the same hospital.
Case control Study
What is this study design?