``TOPIC 5 ERRORS
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
Rb Cl O4 name
rubidium chlorate (VII)
What numbers do we need for delta h
.4.18 x temperature change x volume of FINAL SOLUTION
What does iodine look like as a solid
I2
Which compound requires the most energy to convert one mole into its gaseous ions?
A NaF
B Na2O
C MgF2
D MgO
D
If a reaction gives out heat, its…
exothermic, so delta (x) is negative
Write down the electron configuration of Zn2+
1s2 2s2 2p6 3s2 3p6 3d10
No electrons in 4s sub shell
Define the term ligand
Molecule / ion that has a lone electron pair that can form a dative / coordinate bond with a metal ion, forming a metal comp,ex, where both electrons in the electron pair are donated from the ligand
What bond angle do tetrahedral molecules have
109.5
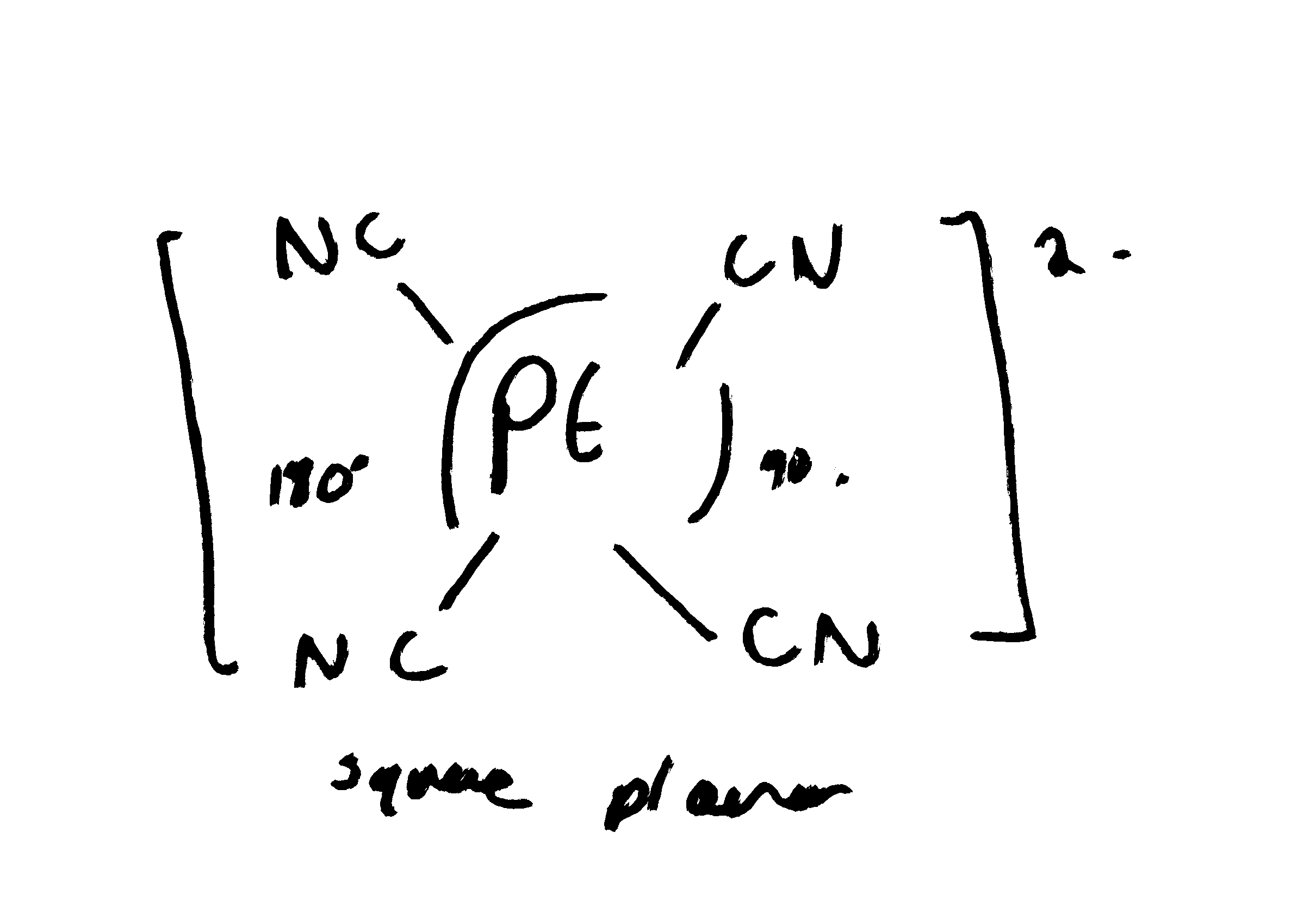
Draw (Pt(CN)4)2- structure
(a) When concentrated hydrochloric acid i s added to aqueous copper (I!) sulfate, the solution colour
changes from blue, first t o green and then t o yellow.
Explain these colour changes. Include a n equation for the reaction involved.
[Cu(H2O)6]2+ (blue) + 4Cl- → reversibly [Cu(Cl)4]2- (yellow) + H2O
Cl- concentration increases as HCl is added
Equillibrium shifts to the right to oppose the change
Solution goes from blue to yellow
Bidentate ligand meaning
Two coordinate / dative bonds are formed by 2 lone electron pairs from the same ligand, with the same central transition metal ion
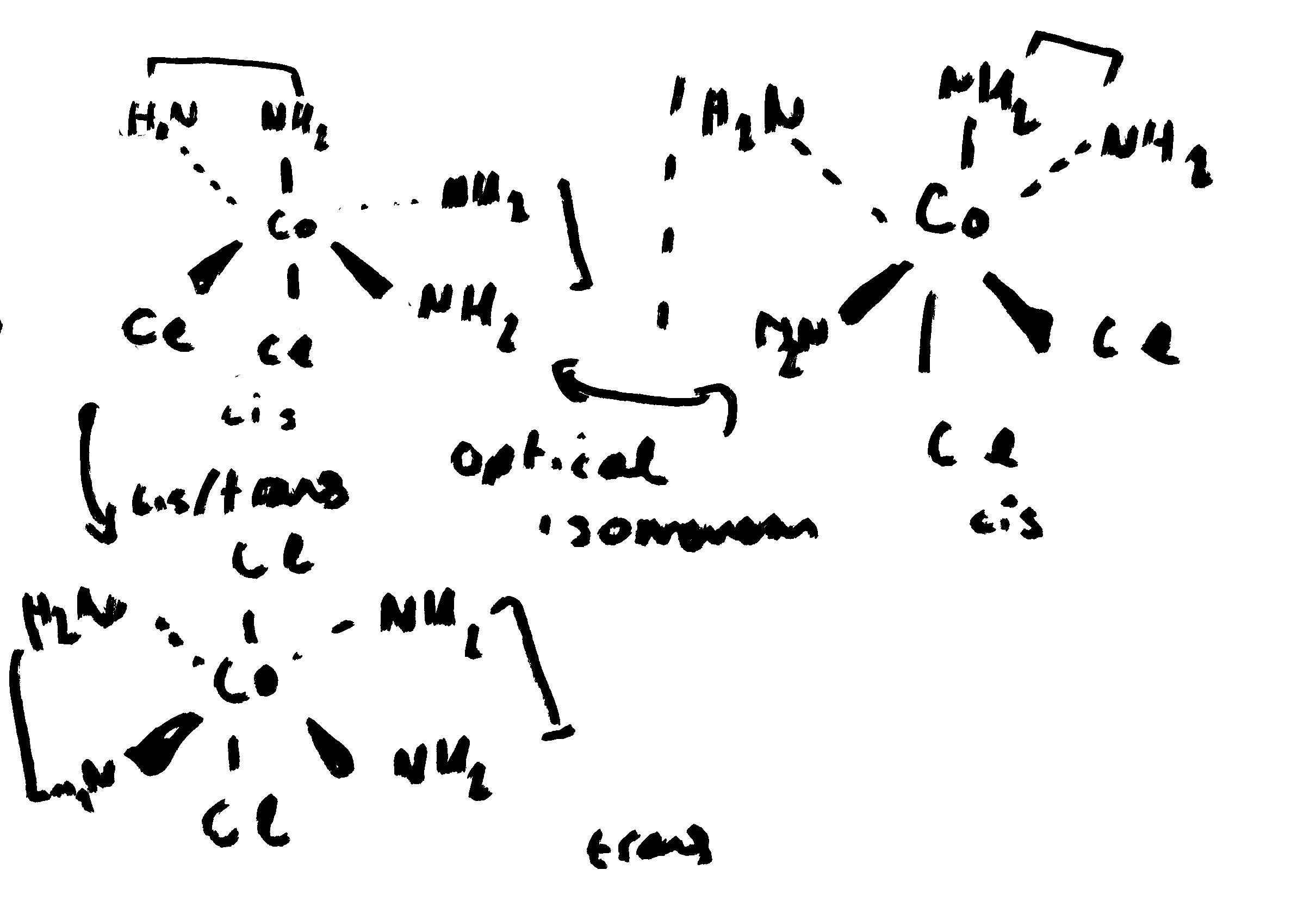
Cis vs trans isomer of complex metal ions in octahedral and square planar
Cis:
octahedral: same group are on same side of metal ion complex (90 degrees)
square planar: same group are adjacent
Trans:
Oct: same group on opposite sides (180 degrees)
Square palanar: same group on opposite sides
Draw and name both types of isomerism in [CoCl2(1,2 - diaminoethane)2]+
Co-ordination number
Number of coordinate bonds formed by ligands to the metal ion in the metal ion complex
How do square planar platinum metal complexes help with the treatment of cancer (2)
Cis isomer is cis platiin, only one isomer (cis) will work
Binds to DNA, preventing it from replication so cancer cells cannot divide
Coordinate bond definition
Covalent dative bond that is dodged between metal ion and ligand, where both electrons come from the same atom
Covalent bond definition
Attraction between a shared pair of electrons and the nuclei of two atoms
Explain the biological role of haemoglobin as an oxygen carrier in terms of ligand substitution (4)
Hb contains Fe2+ which forms an octahedral complex ion with O2 which can reversible substitute with H2O
HbO2 + H2O reversible to HbH2O + O2
In lungs at high o2 concentration, equilibrium shifts to left so o2 is picked up
At low o2 concentration at respiring tissues, equilibrium shifts to right so O2 is released
Colours of Fe3+ and Fe2+ precipitate with NaOH
Yellow and green
How to form and properly check colour of precipitate a (3)
Filter off precipitate
Wash with water to see colour clearly
Leave to dry
Why is COOH a worse ligand than COO-
Uncharged, less attracted to metal ion
How to reduce K2Cr2O7 2- to Cr3+
Add HCl (aq) and Zn (s)