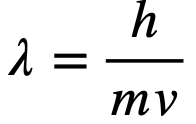Quantum Phenomena
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
What is the Photoelectric Effect?
If light is shone onto a metal surface so that the frequency of the light is greater than the threshold frequency of the metal, electrons from the surface of the metal will be released.
Explain why the photoelectric effect is evidence of the particle nature of light.
One electron absorbs one photon of light.
The energy of the photons = plank’s constant x frequency
If the energy is less than the work function of the metal no electrons will be released.
Define the Threshold Frequency
The lowest frequency of light that can cause electrons to be emitted from the surface of a metal.
Define the Work Function (ϕ)
The minimum quantity of energy which is required to remove an electron to infinity from the surface of a metal.
What is observed if the photon energy < ϕ
The electron cannot escape
What is observed if the photon energy = ϕ
The electron can just leave the surface and f (the frequency of the the em radiation) is the threshold frequency f0.
What is observed if the photon energy > ϕ
There is energy to spare and this becomes the kinetic energy of the electron
What is the effect of increasing the intensity of the photons incident on the metal
Only increases number of electrons released if the radiation is of sufficient energy (frequency). Will not change the maximum kinetic energy of the photoelectrons.
What is the effect of increasing the frequency of the photons incident on the metal
If already above the threshold frequency —> kinetic energy of the emitted electrons increases.
If below the threshold frequency —> electrons will instantly be emitted when the frequency reaches the threshold frequency
What is the Stopping Potential?
The potential difference required to stop the fastest moving electrons crossing a gap.
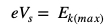
What is an Electron Volt?
The kinetic energy carried by an electron after it has been accelerated from rest to a pd of 1 V. 1eV = 1.6 x10-19 J
What are; a, b, & c on the graph?
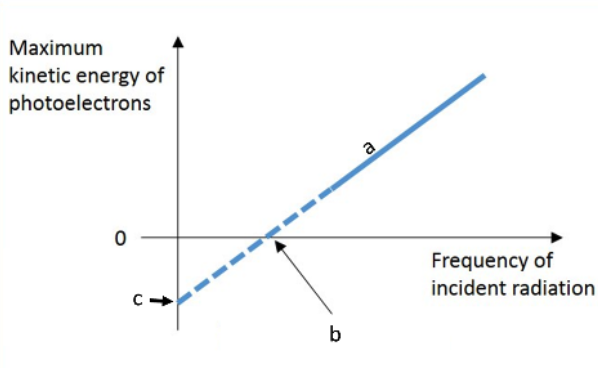
a= planck’s constant
b= threshold frequency
c= work function
When is an atom in the Ground State?
When all the electrons in an atom are in their lowest possible energy level
What happens when an atom is excited?
One or more electrons absorb a photon (with energy equal to the difference between the energy levels) and move into a higher energy level in the atom.
What happens when an atom is ionised?
An electron absorbs a photon and is removed from the atom
What happens when an atom de-excites?
An electron moves towards ground state releasing a photon with energy equal to the difference between the energy levels the electron has moved between.
They can de-excite directly to the ground state, or via other levels
How does a fluorescent Tube work?
High voltage applied - accelerates the free electrons
Ionises some mercury atoms, producing more free electrons.
When these electrons collide with the electrons in the mercury atoms, the atomic mercury electrons are excited to a higher energy level
When the electrons drop back down they give out photons which are in the UV range.
Phosphorous coating on the inside of the tube absorbs the UV photons, exciting its electrons to a much higher energy level.
When these electrons cascade down the energy levels, they lose energy by emitting many lower energy photons of visible light
Line Emission Spectra
A series of bright lines against a black background, with each line corresponding to a wavelength of light.
Line Absorption Spectra
When light with a continuous spectrum of energy (white light) pass through a cool gas. Some electrons will absorb photons of light and be excited into higher states. These photons are then missing from the spectrum causing black lines on the continuous spectrum.
Diffraction
When a wave passes through a narrow gap and spreads out.
Wave Particle Duality
An entity behaving with both particle and wave-like behaviour.
The wavelength of a wave particle can be found from the De Broglie's Wavelength: