L5- Vitamins and Coenzymes
1/79
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
80 Terms
What defines a vitamin?
An essential organic molecule required in small amounts in the diet to produce coenzymes or regulate metabolism.
Which two major categories are vitamins divided into?
Water-soluble and fat-soluble.
Why are water-soluble vitamins not stored extensively in the body?
They are hydrophilic and rapidly cleared in urine.
Which vitamins are fat-soluble?
Vitamins A, D, E, and K.
Why are fat-soluble vitamins cleared more slowly?
They are hydrophobic and stored in fatty tissue and liver.
What is a cofactor?
A nonprotein molecule required for enzyme activity.
Difference between apoenzyme and holoenzyme?
Apoenzyme lacks cofactor; holoenzyme = apoenzyme + cofactor (active form).
Two major types of cofactors?
Metal ions and coenzymes.
How do metal ions assist catalysis?
They stabilize substrates and transition states, often via positive charge.
Metal in carbonic anhydrase II?
Zn²⁺.
What is a coenzyme?
A small organic molecule assisting enzyme catalysis.
Prosthetic group vs loosely associated coenzyme?
Prosthetic group is tightly bound; loosely associated acts as second substrate.
Why must loosely associated coenzymes be present in stoichiometric amounts?
They are consumed and regenerated during reactions.
Which vitamins produce most metabolic coenzymes?
B vitamins.
Coenzyme derived from thiamine (B1)?
Thiamine pyrophosphate (TPP).
Pyruvate dehydrogenase complex links?
glycolysis to the TCA cycle
Coenzyme derived from niacin (B3)?
NAD⁺.
Coenzyme derived from pantothenic acid (B5)?
Coenzyme A (CoA).
What does CoA transfer?
Acyl groups.
What is the most abundant protein in mammals?
Collagen
Collagen accounts for how much of total protein mass
25-30%
collagen is extremely rich in
glycine, proline, hydroxyproline
The amino acid sequence of a part of a collegen chain reveals that glycine is every _____ residue.
third
Why are B vitamins rarely toxic compared to fat-soluble vitamins?
Why are B vitamins rarely toxic compared to fat-soluble vitamins?
Why must every third residue be glycine?
Only glycine fits inside the crowded triple helix core.
Structural level of collagen triple helix?
Superhelical quaternary structure.
What stabilizes collagen superhelix?
Interchain hydrogen bonds.
Are there hydrogen bonds within a strand of collagen?
no
What is hydroxyproline?
Hydroxylated form of proline in collagen.
When is proline hydroxylated?
After procollagen synthesis.
Enzyme that hydroxylates proline?
Prolyl hydroxylase.
Cofactors for proline hydroxylation?
O₂, α-ketoglutarate, Fe²⁺, ascorbate.
Role of vitamin C in collagen synthesis?
Regenerates active prolyl hydroxylase.
What happens to ascorbate during hydroxylation?
It is oxidized.
What enzyme catalyzes hydroxylation of proline in collagen?
Prolyl-4-hydroxylase.
Where does proline hydroxylation reaction occur?
In the ER during collagen synthesis.
What happens to α-ketoglutarate during the reaction?
It is decarboxylated to succinate.
What metal ion is required in the active site?
Fe²⁺ (ferrous iron).
What happens to iron during the reaction?
It becomes oxidized from Fe²⁺ to Fe³⁺.
Why is Fe³⁺ a problem for the enzyme?
The enzyme becomes inactive if iron remains oxidized.
What restores iron to the active Fe²⁺ state?
Ascorbate (vitamin C).
what does peptide bound proline become
peptide bound hydroxyproline
Disease from vitamin C deficiency?
Scurvy. (common in sailors)
if theres no vitamin c what cant happen?
ascorbate cant regenerate active enzyme, leads to buildup of inactive enzyme, lack of enzyme decreases proline hydroxylation
Why does vitamin C deficiency weaken collagen?
Reduced hydroxylation → fewer hydrogen bonds → unstable triple helix.
Why do scurvy symptoms worsen over time?
Collagen turnover replaces stable collagen with defective collagen.
What does thiamine (B1) defiency cause
Beriberi
what is beri beri
thiamin deficiency disease, neurologic and cardiovascular disorder,that causes muscle weakness, loss of appetite, nerve degeneration, and edema. BERRIES ARE FAT AND DUMB (prabh did this not me)
TPP is critical for
central carbon metabolism
3 things TPP is used for
1. Glycolysis
2. TCA cycle
3. Aminoacid catabolism
TPP is the coenzyme for
pyruvate dehydrogenase and a-ketoglutarate
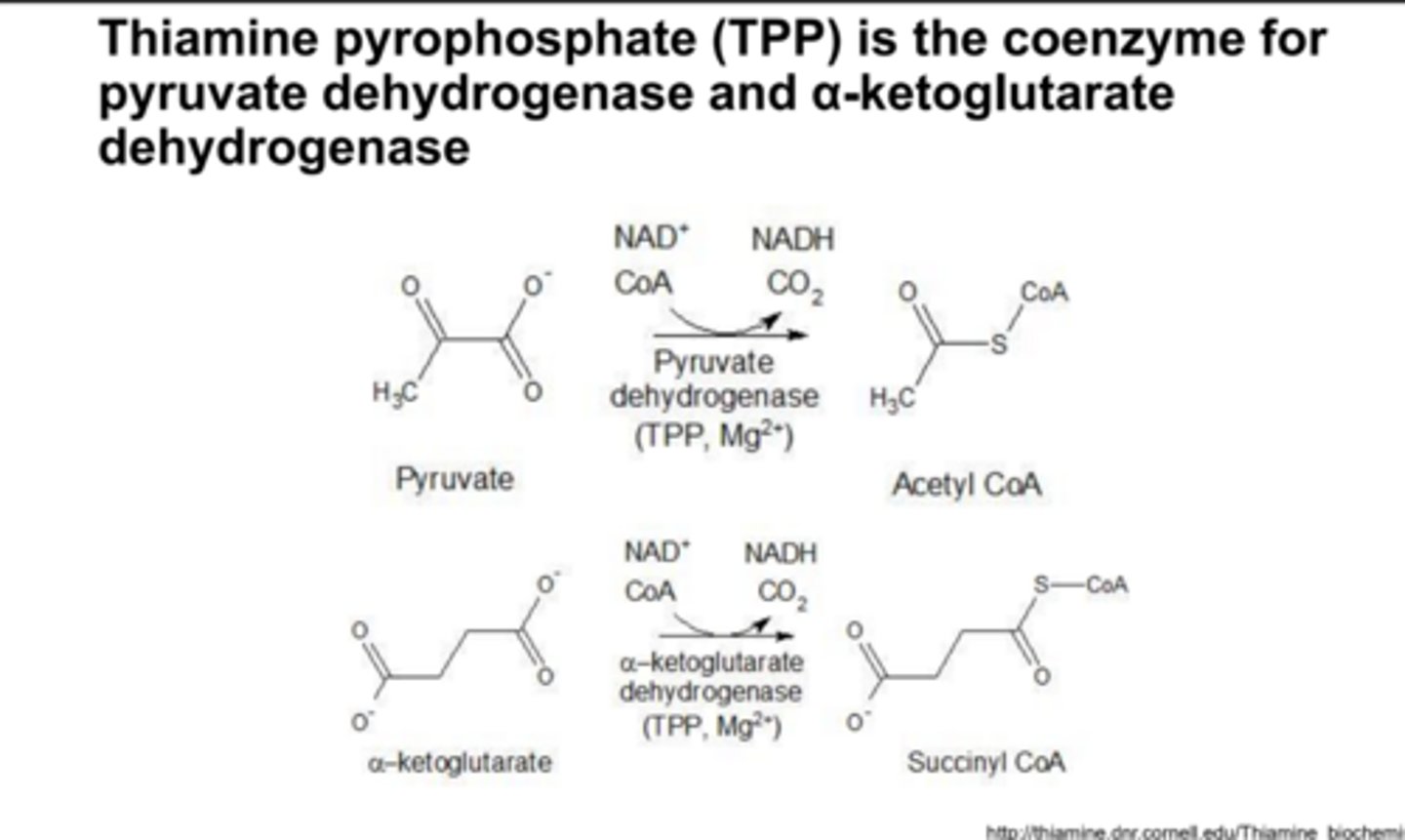
In patients with beriberi, the levels of pyruvate and α-ketoglutarate are ______ than normal
higher
symptoms of what are similar to beriberi
mercury and arsenic due to inhibiton of pyruvate dehydrogenase complex
Why does arsenite shut down pyruvate dehydrogenase?
Because they bind strongly to sulfhydryl (-SH) groups in active sites, disrupting essential catalytic chemistry.
What is the general strategy behind treating metal poisoning with sulfhydryl compounds?
Use a molecule with higher affinity thiol groups to outcompete the enzyme, bind the metal, and allow the enzyme to function again.
Cobalamin (b12) deficiency causes what? and why?
anemia, because blood cells require proliferation: so low cobalamin levels cause: 1: enlarged red blood cells
2: nuclear hypersegmentation of DNA in nucleophils
cobalamin has 3 similar coenzymes what are they
cyanobalamin- CN
methylcobalamin- CH3
Coenzyme B12
cobalamin catalyzes a
rearrangement reaction
What two reactions is cobalamin a coenzyme for?
Methylmalonyl-CoA mutase (fatty acid degrdation) and Methionine Synthase( remethylation of homocysteine)
What reaction does methylmalonyl-CoA mutase catalyze?
→ Methylmalonyl-CoA → succinyl-CoA. in the mitochondria
What molecule donates the methyl group in the methionine synthase reaction?
Methyl-H4-folate.
Where does the methyl thats donated go and what adds it and what product is formed
homocysteine, ---methionine synthase adds it also --- makes methionine
What cycle is directly connected to methionine synthase activity?
→ The folate cycle.
What cellular process depends on proper folate cycling?
DNA synthesis.
What nucleotide is specifically shown as dependent on folate metabolism?
Thymidine.
What molecule formed from methionine is used for methylation reactions?
S-adenosylmethionine (SAM).
Why do humans require dietary vitamins but E. coli do not?
Humans lack biosynthetic pathways for vitamins; E. coli have them.
What allows E. coli to grow on glucose and salts alone?
it can synthesize its own vitamins.
Why are vitamin biosynthetic pathways considered complex?
→ They require many enzymatic steps.
What evolutionary trade-off occurred in humans regarding vitamin synthesis?
→ Loss of synthesis pathways in exchange for dietary dependence.
Why is there debate about whether humans require 12 or 13 vitamins?
→ Because vitamin D can be synthesized in the body but still requires dietary intake. ( we go with 13)
What molecule is vitamin D3 synthesized from?
Cholesterol.
What environmental factor is required for vitamin D3 formation?
Ultraviolet (UV) light.
What structural change occurs during vitamin D synthesis?
→ Ring splitting of a cholesterol precursor.
In which organs is vitamin D3 converted to its active form?
→ Liver and kidneys.
What is the active hormone form of vitamin D?
→ Calcitriol.
What mineral absorption does calcitriol promote?
→ Calcium and phosphorus.
Why is vitamin D critical for bone health?
→ It enables calcium and phosphate absorption.
Why is dietary vitamin D still required even though we synthesize it?
→ Endogenous synthesis is insufficient for physiological demand.
What distinguishes essential from nonessential amino acids?
→ Essential amino acids must be obtained from diet.