Carboxylic Acids: Intro + Physical properties
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
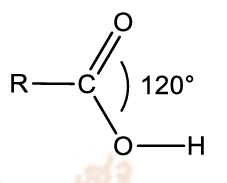
general structure of carboxylic acids
contains the carboxyl group (-CO2H)
trigonal planar about the carboxyl carbon (bond angle=120)
carbonyl carbon is sp2 hybridised
nomenclature for carboxylic acids
carbon bonded to
straight chain: suffix ‘-oic acid’ eg methanoic acid
-CO2H bonded to the ring: suffix ‘carboxylic acid’ eg cyclopentane carboxylic acid
-CO2H bonded to aromatic ring: suffix ‘benzoic acid’
carbon in ring attached to -CO2H is numbered C1
Arrange in increasing boiling point
carboxlic acids
hydrocarbons
aldehydes and ketones
alcohols and phenols
hydrocarbons> alcohols and phenols > aldehydes and ketones > carboxylic acids
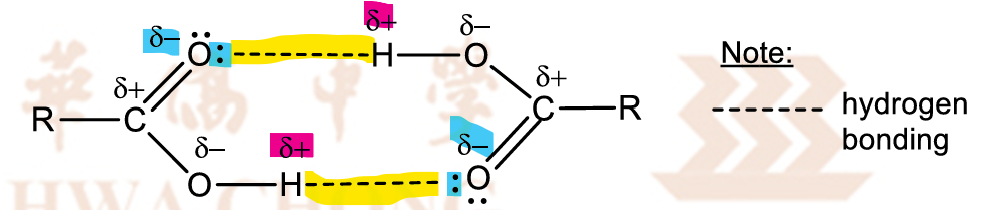
Why do carboxylic acids have higher bp than alcohols with similar electron clouds size?
Carboxylic acids dimerise with hydrogen bonding
effectively doubling their electron cloud size in the dimer
stronger dispersion forces between carboxylic acid molecules
electron-withdrawing C=O group intensifies the partial positive charge on the H atom,
stronger hydrogen bonding between carboxylic acid molecules
more energy required to overcome the stronger dispersion forces and hydrogen bonding than for alcohol
Which type of carboxylic acids are highly soluble in non-polar solvents and why?
carboxylic acids with long carbon chains
can form hydrogen-bonded dimers and increase size of e- cloud, therefore strength of dispersion forces
energy released from stronger dispersion forces between hydorcarbon chain of dimers and solvent molecules
is able to overcome the dispersion forces and hydrogen bonding in carboxylic acid and hydrogen bonding in water
Which type of carboxylic acids are highly soluble in water/polar solvents and why?
carboxylic acids with shorter hydrocarbon chains
cannot form hydrogen-bonded dimers
instead, form hydrogen bonds with water
energy released form forming of hydrogen bonds between carboxylic acid molecules and water moleculesbetween -CO2H group and water molecules
is more than energy required to overcome weaker dispersion forces between carboxylic acid molecules and hydrogen bonding in water
As length of hydrocarbon chain of carboxylic acid inceases, solubility in water increases/decreases?
as hydrocarbon chain length increases
stregnth of dispersion forces between carboxylic acid molecules inceases
energy released from hydrogen bonding between -CO2H group and water molecules
is less than energy needed to overcome stronger dispersion forces between carboxylic acid molecules and hydrogen bonding between water molecules