4. Mass and Heat transfer
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
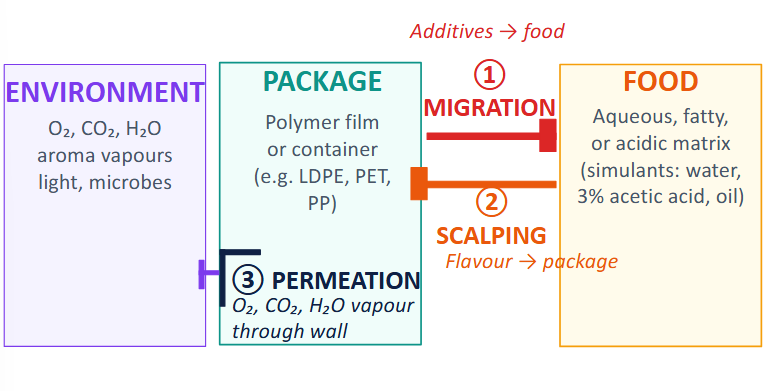
What are the three directions of mass transfer?
Migration (additives (in packaging) → food)
Scalping (flavour → package)
Permeation (O2, CO2, H2O vapor through wall)
What is Fick’s first law?
Describes the diffusion of a permeant through a polymer film
Diffusion flux J is the amount of permeant diffusing per area per time through a plane perpendicular to the flow direction.
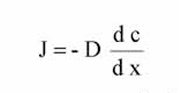
Fickian vs Non-fickian behavior
Fickian = concentration independent diffusion coefficients
Non-Fickian = Concentration dependent behavior
Three different steps of permeation through a polymer film
Sorption (Henry’s law)
Diffusion (Fick’s law)
Is typically 103 - 104 x slower than the surface steps
Therefore, D controls the overall transmission rate, not the surface equilibria
Desorption (Henry’s law)
Henry’s law formula
describes how much gas dissolves into a material at the surface.

Permeability coefficient formula
D = Diffusion coefficient
S = Solubility coefficient
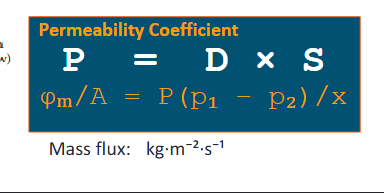
What are diffusivity and solubility?

Mass flux vs mass flow rate
Mass flux (Φm/A): the rate of mass transfer per unit area (kg.m-2.s-1)
Mass flow rate (Φm): the amount of mass passing over unit time (kg.s-1)
EVOH
Used as a gas barrier layer - has really low GTR
However it is humidity sensitive
Water plasticises the EVOH matrix, increasing free
volume and therefore diffusivity D, raising GTR
dramaticallyOnly works well in low humidity
Units thickness normalized flux, N

units transmission rate

Units permeance (R)

Units permeability coefficient (P)

What is GTR?
GTR stands for Gas Transmission Rate. It is a specific type of transmission rate (TR) used to measure the quantity of a gas (such as oxygen, nitrogen, or carbon dioxide) that passes through a packaging film
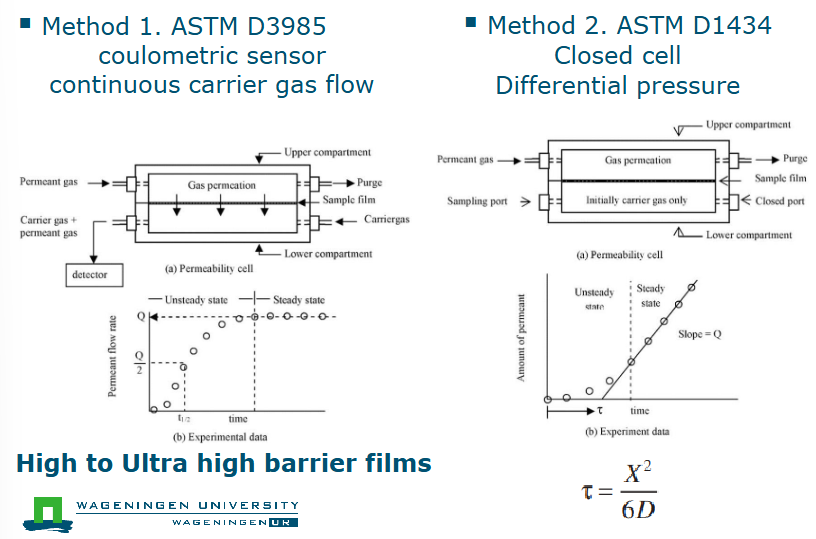
What are the two methods to measure GTR
Method 1: ASTM D3985 coulometric sensor
Continuous carrier gas flow
High to ultra high barrier films
Method 2: ASTM D1434
Closed cell
Differential pressure
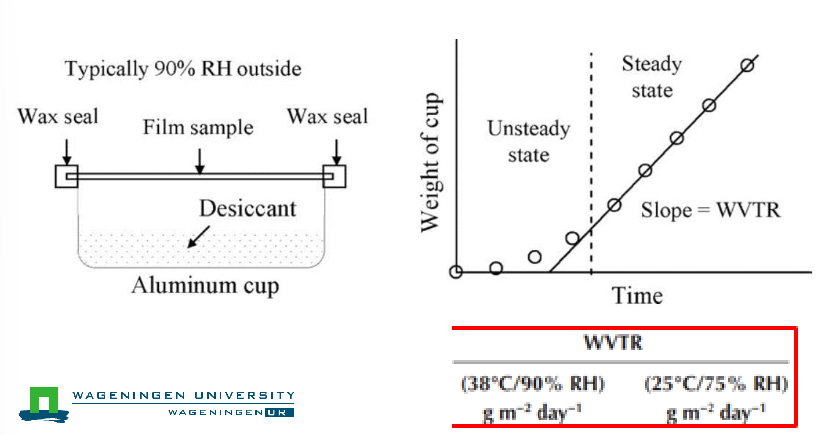
How do you measure WVTR?
Done using an ASTM E96 (gravimetric desiccant method)
EVOH (ethylene-vinyl alcohol) shows excellent O₂ barrier
at low humidity, but a poor O₂ barrier at high relative humidity. What is the physical reason?
EVOH is a crystalline copolymer containing polar hydroxyl (-OH) groups.
Under dry conditions, these -OH groups engage in intense hydrogen bonding between the polymer chains
Because EVOH is a hydrophilic polymer, it readily absorbs water vapor when exposed to high relative humidity
Once absorbed, the water molecules interact with the polar -OH groups and behave as a plasticizer within the polymer structure
How to calculate GTR?
Calculate flux: respiration rate * weight = total flux (ml/h)
Determine partial pressure: pout - pin = partial pressure
Rearrange: flux (ml/h) = GTR x partial pressure x area
How is GTR measured in laminate structures?
Using resistance in series
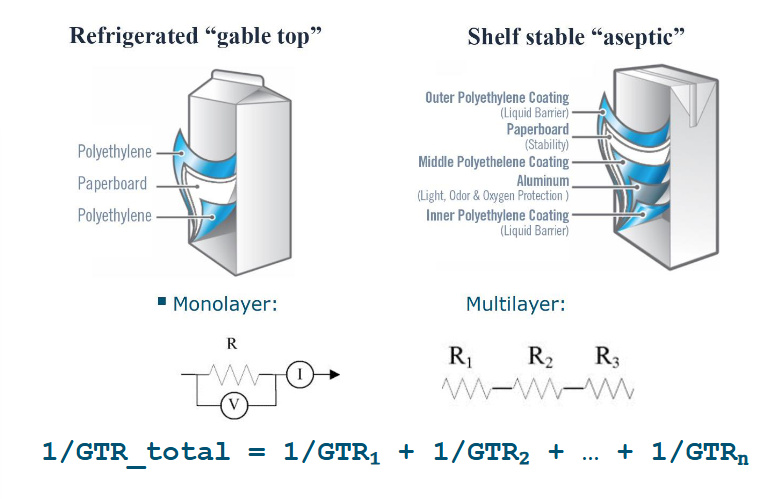
How do you calculate total GTR?
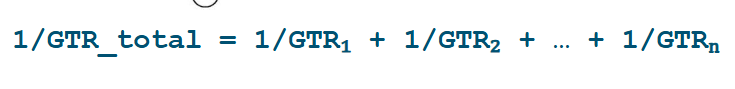
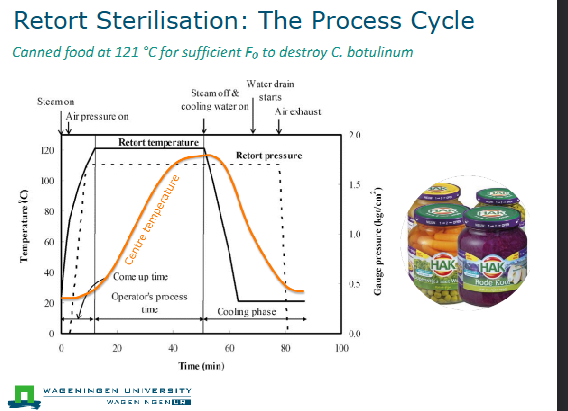
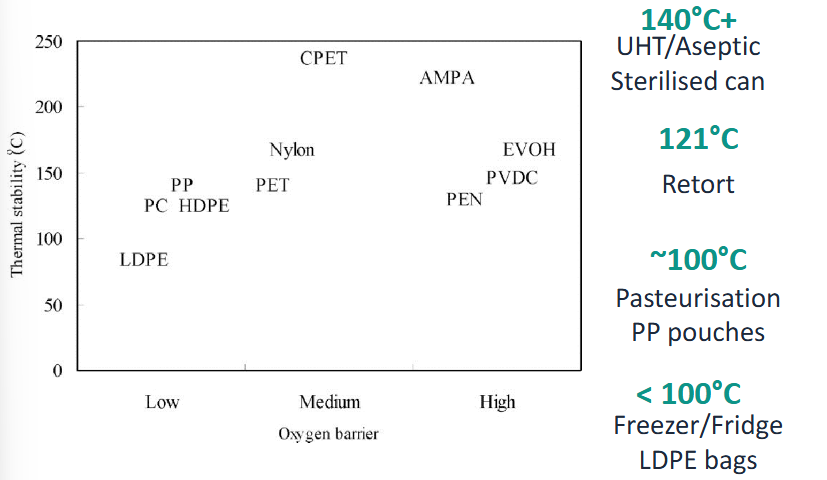
Retort sterilization
Heat stability of packaging plastics
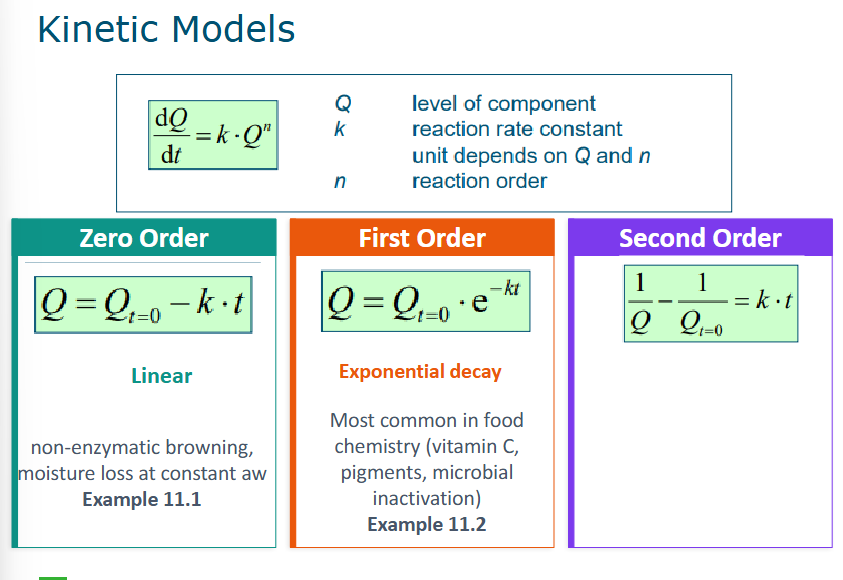
Zero, first and second order models
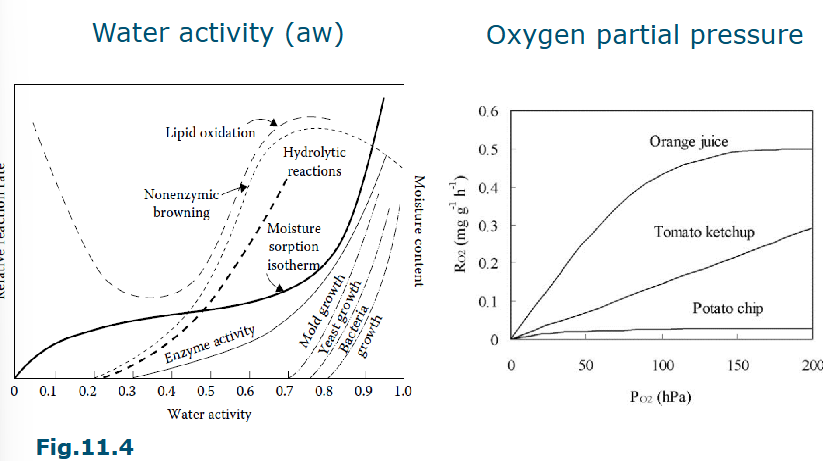
Factors affecting reactions
Temperature
Water activity
Oxygen partial pressure
What can cause deviations in the Arrhenius equation?
Changes in physical phase (e.g. freezing)
Other reaction becomes dominant
Water activity change with temperature
pH change with temperature
Solubility changes with temperature
Catalyst is inactivated
Temperature UHT/Aseptic sterilized can
140 C
Temperature Retort
121 C
Temperature Pasteurization of PP pouches
~100 C
Temperature of making freezer/fridge LDPE bag
<100 C
A package shows a pressure decay of ΔP = 2 mbar over 20 seconds in a chamber with volume 1000 cm³. Assuming atmospheric pressure is 1000 mbar, which of the following best describes the effect of doubling the diameter of a cylindrical leak (pinhole) on the leak flow rate?
