TMs 2 - Ch 12 - 14 - ligand effects, reaction types, catalysis
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
trans effect series
least substitution of trans ligand
F-, H2O, OH-
< NH3 < py
< Cl- < Br- < I-
< NO2-, Ph- < PR3
< H-, NO, CO, CN-, C2H4
most substitution of trans ligand
Linear vs. bent NO
Linear = 3 e- donor
Bent = 1 e- donor
Describe the bonding in alkyne ligands
2x π systems
Only 1 is of correct orientation to react
act same as alkenes - 2 e- donor (L type)
what metals do phosphines help stabilise?
Late TMs (Pd, Pt)
In 0 ox. state
Table of reactions between metals + ligands
ligand exchange reaction
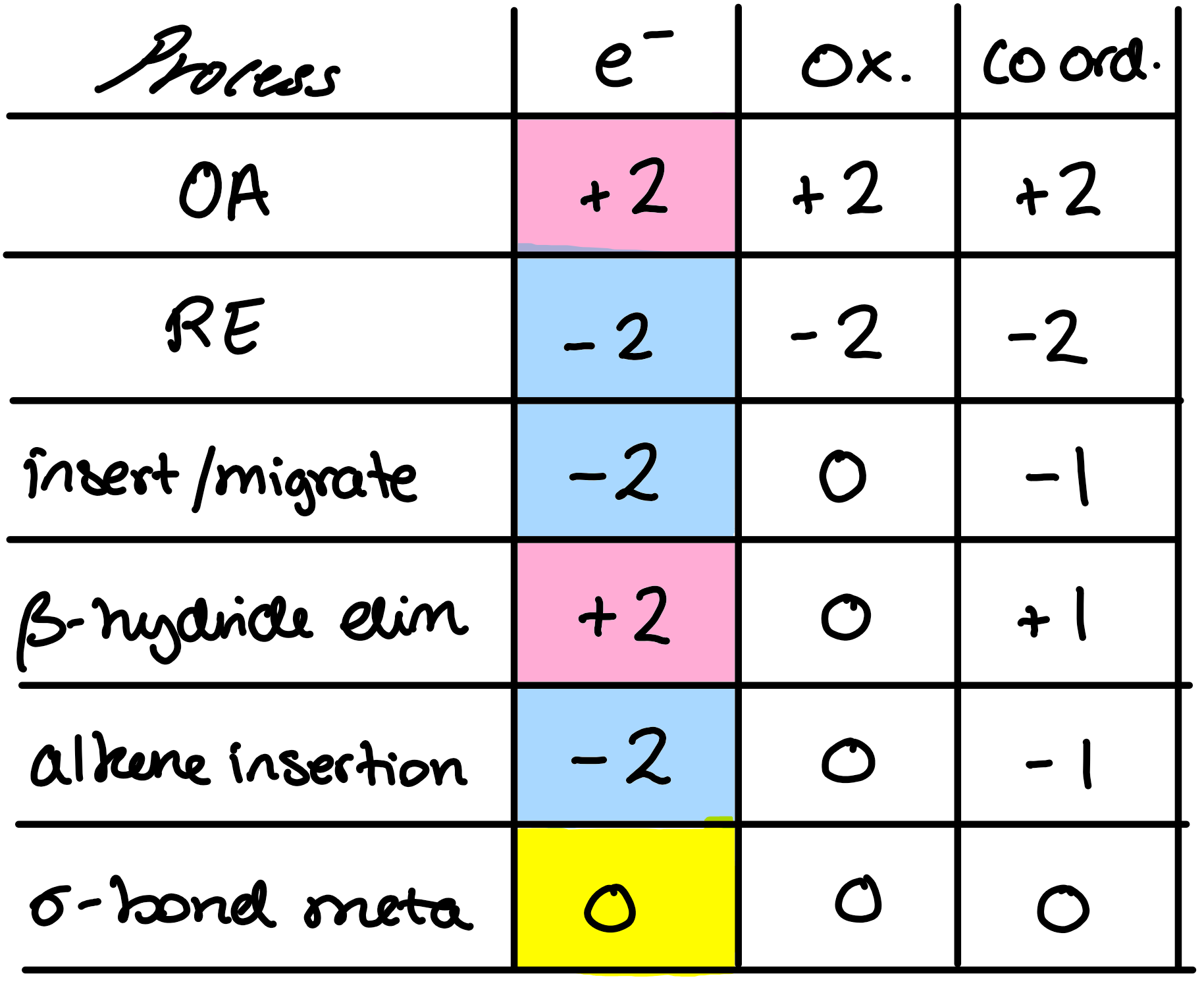
oxidation state change in metals
rearrangement of e- between metal + ligand
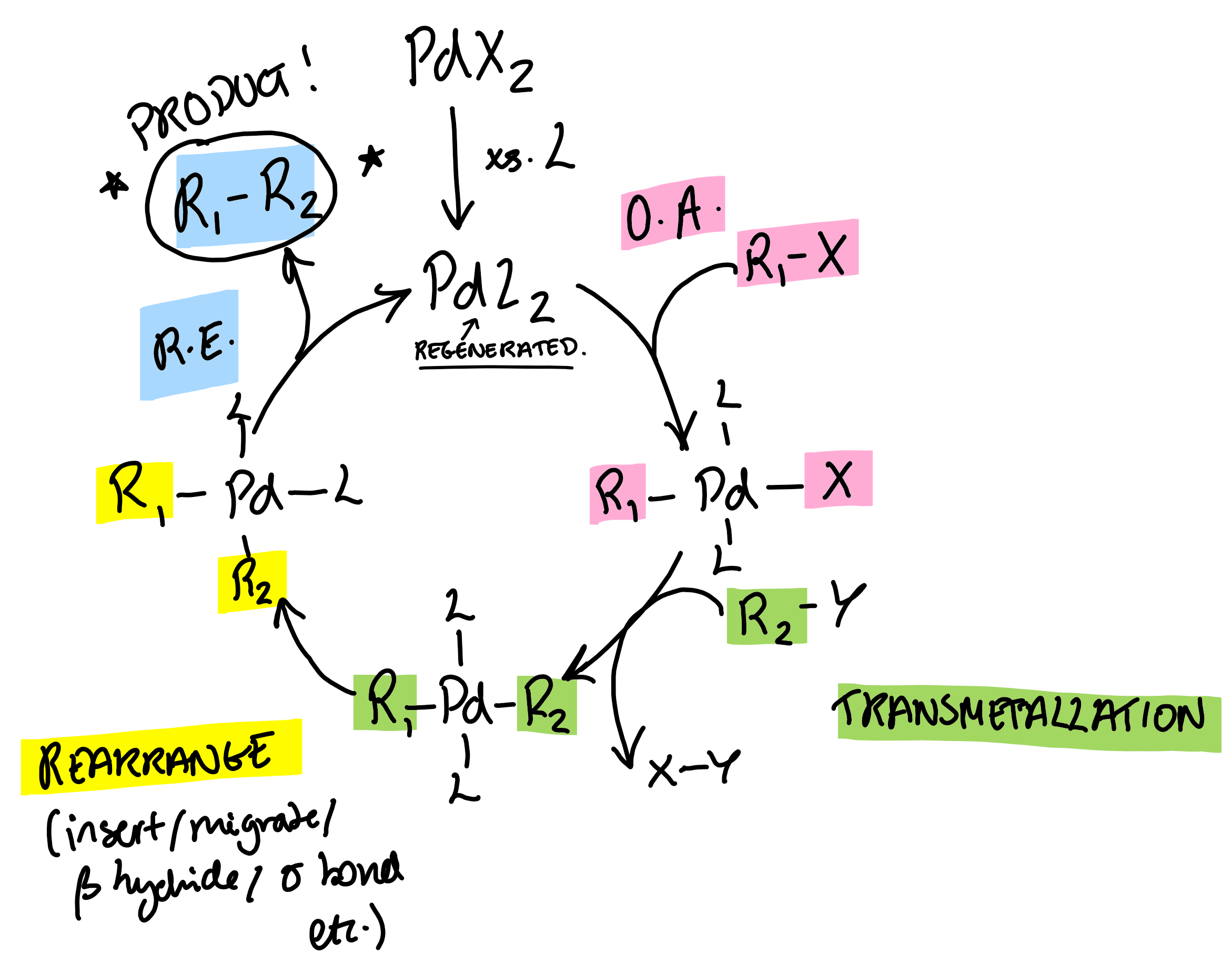
oxidative addition
reductive elimination
insertion/migration reaction
β-hydride elimination
eliminates FROM LIGAND - forms C=C
alkene insertion
GETS RID OF ALKENE!
σ- bond methathesis
d0 metals
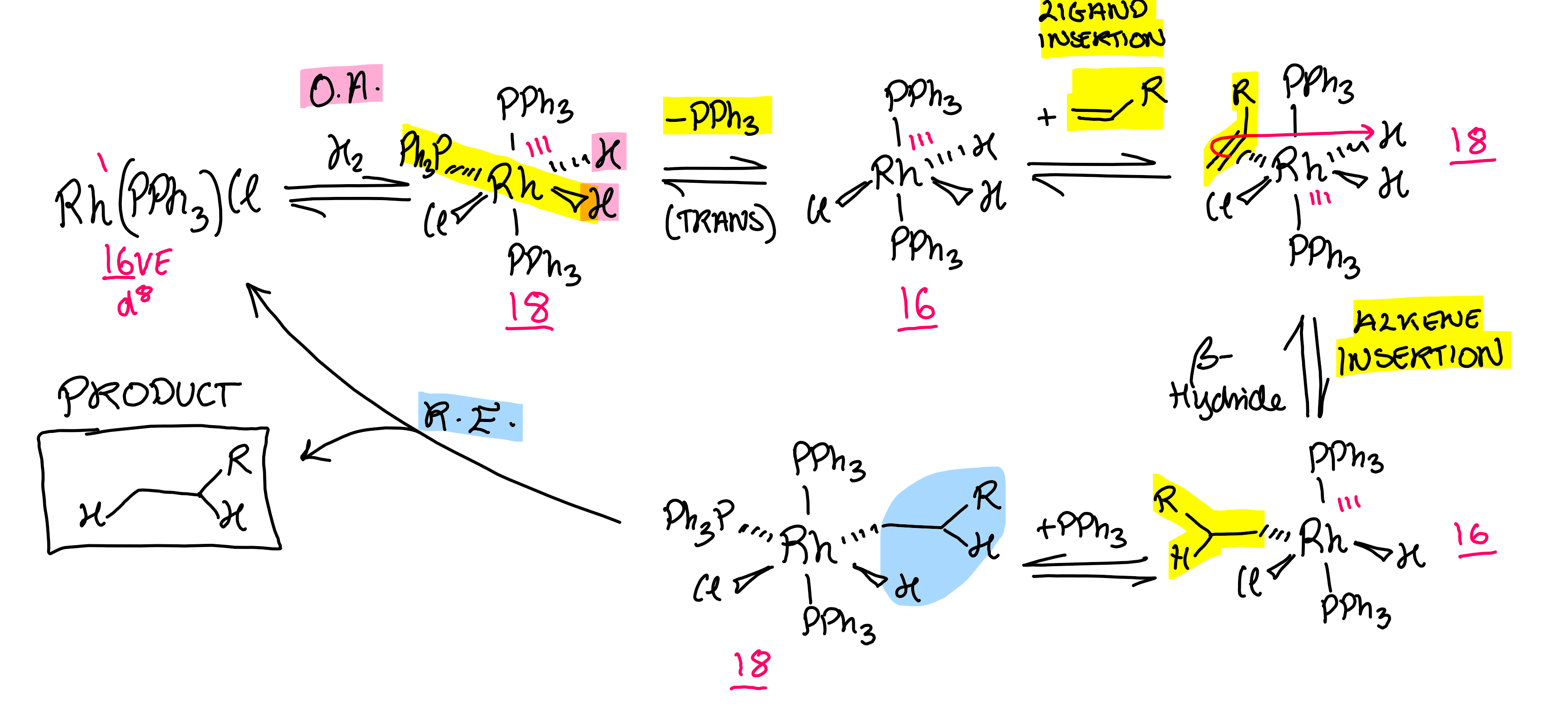
Formula of Wilkinson’s catalyst. What does it do?
Catalyses hydrogenation across C=C bond

Hydrogenation catalytic cycle
Focus: Wilkinson’s Catalyst, a Rh complex.
What is a hydroformylation rxn?
Add CO + H
to C=C
Form aldehyde
Hydroformylation catalysis
Focus: migration/insertion of CO
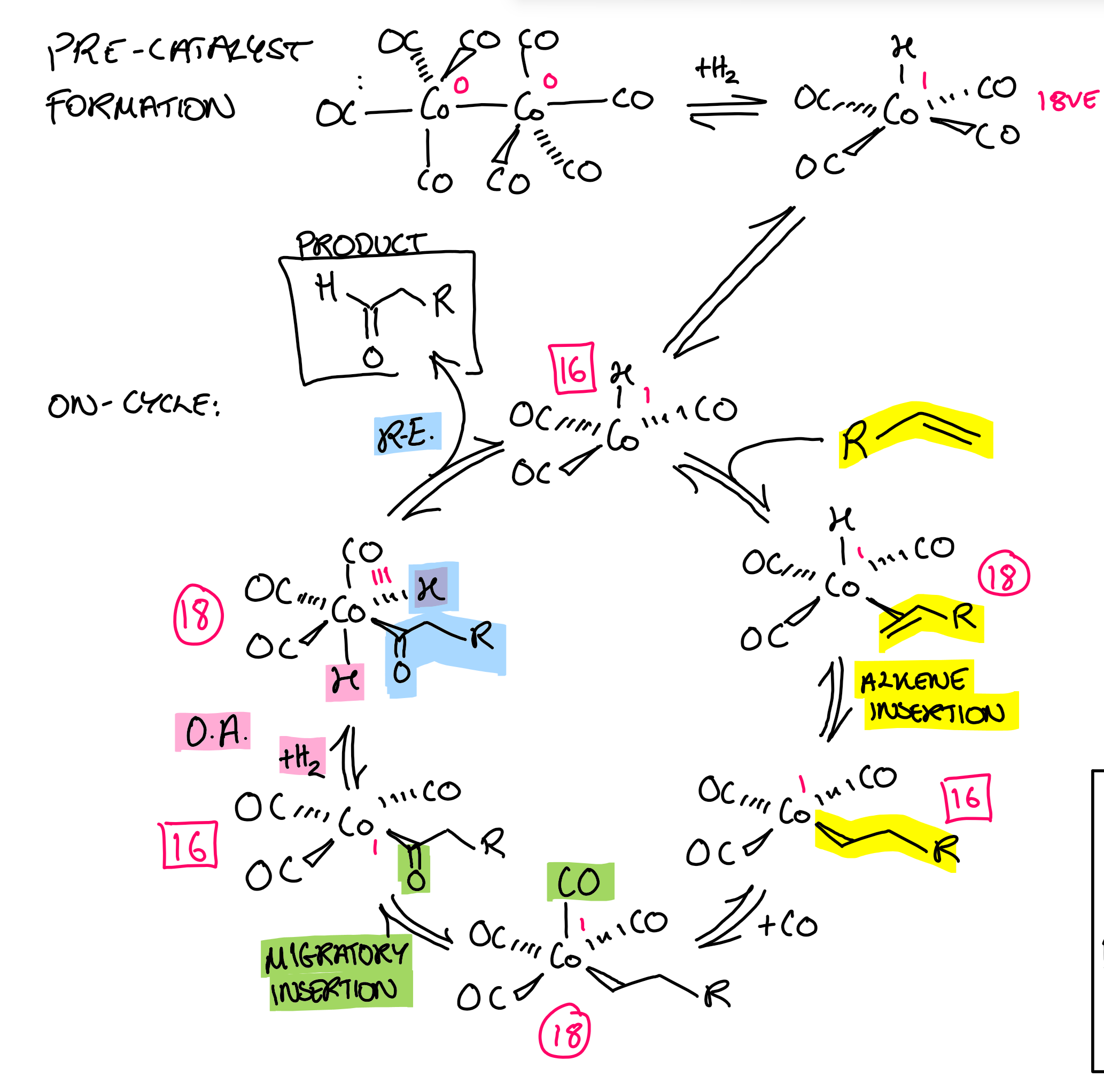
How do you dictate formation of a linear vs. branched aldehyde in hydroformylation?
+ bulky group on catalyst (e.g. PR3) = branched
CO (less bulky group) = linear
STERICS not electronics dominated
What reaction is Mizoroki-Heck used for?
extending ALKENE chain
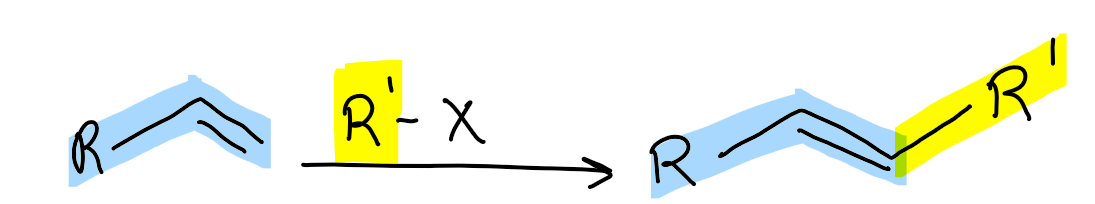
Mizoroki-Heck cross-coupling cycle
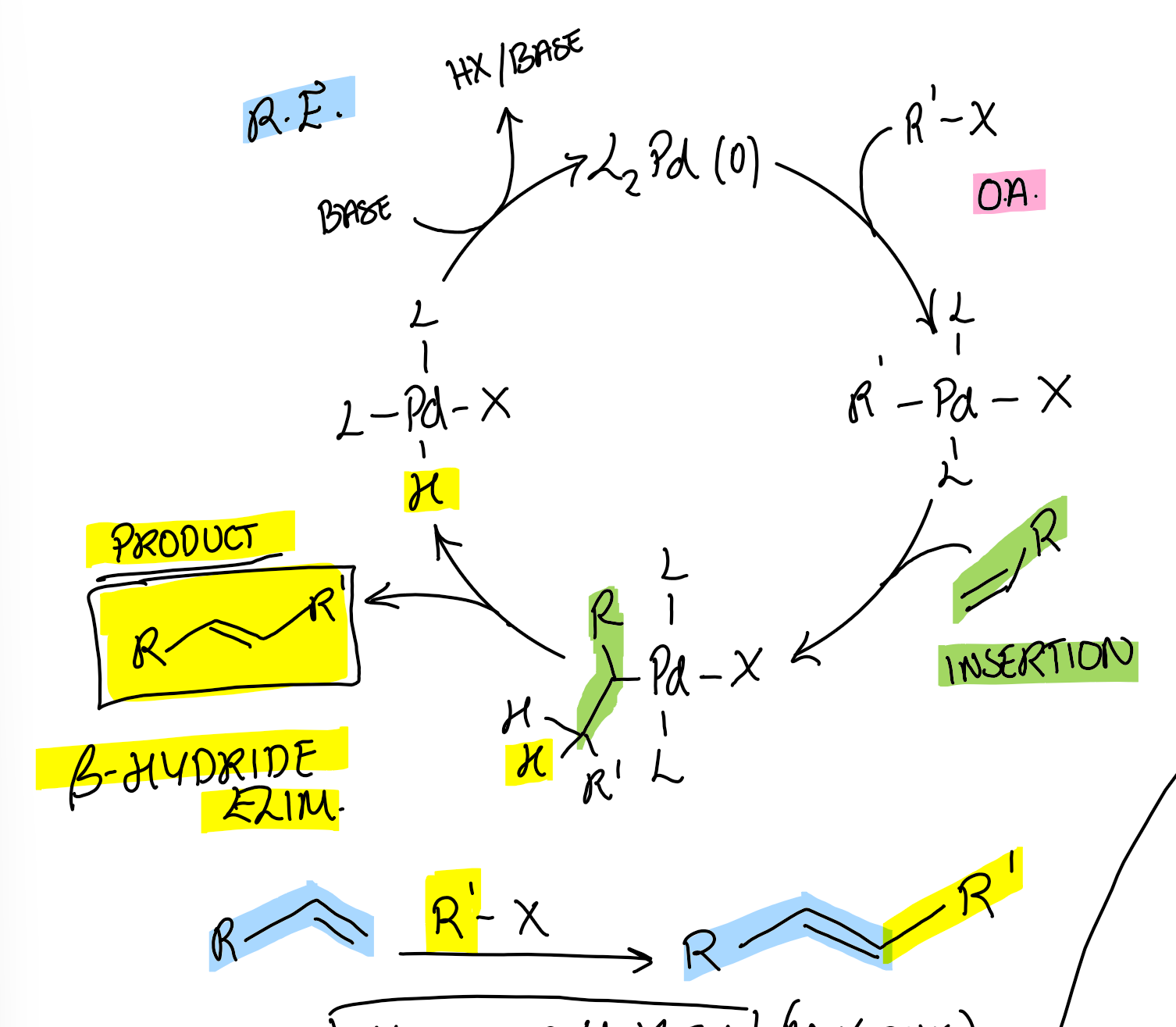
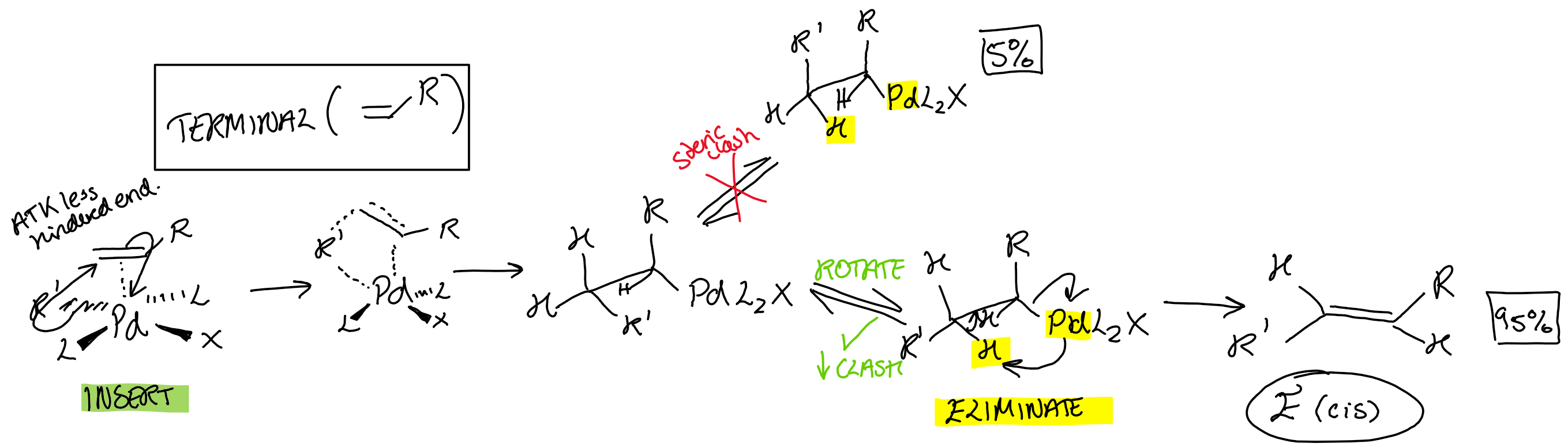
What is the selectivity of the Mizoroki-Heck reaction for terminal alkenes, H2C=CHR?
TS goes via R’ attack on least substituted side
Force E (trans) due to steric clash when eliminating β hydride
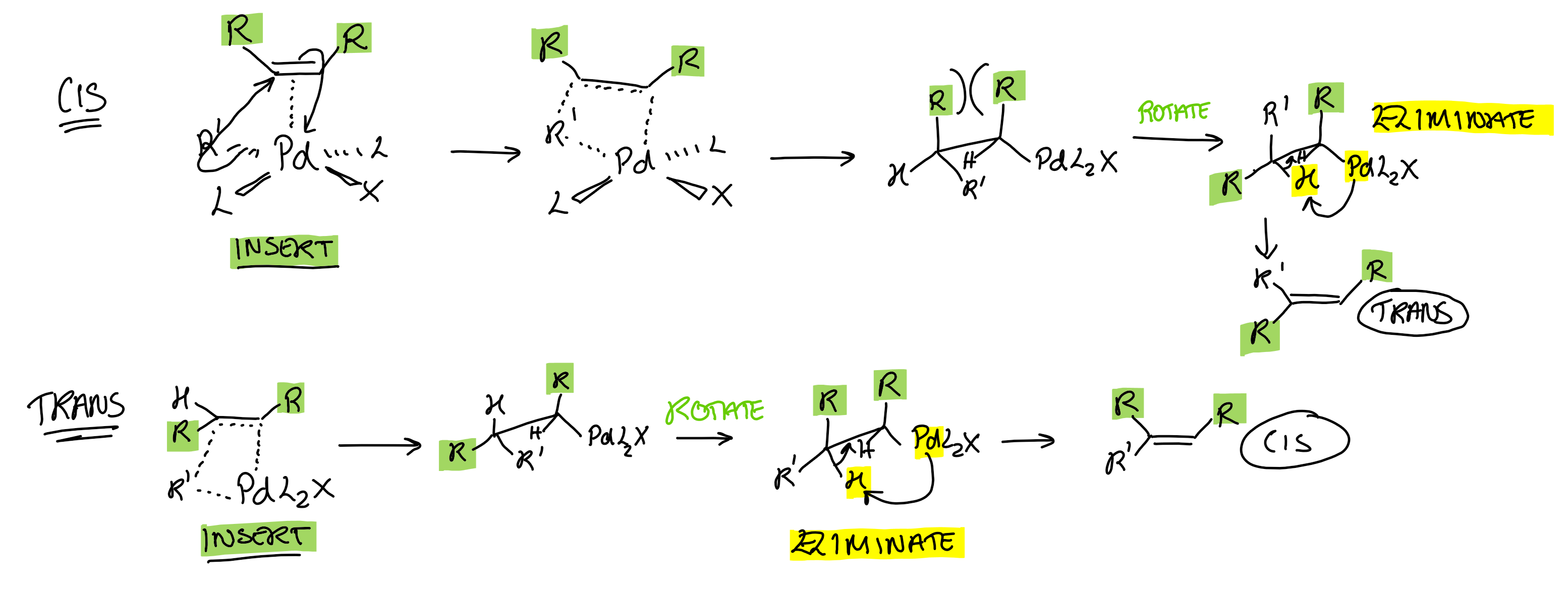
What is the selectivity of the Mizoroki-Heck reaction for 1,2 disubstituted alkenes, RHC=CHR?
INVERSION cis ↔ trans
Only 1 elimination position possible
What reaction is Suzuki-Miyaura used for?
join 2 aromatic rings, via BORONIC ACID
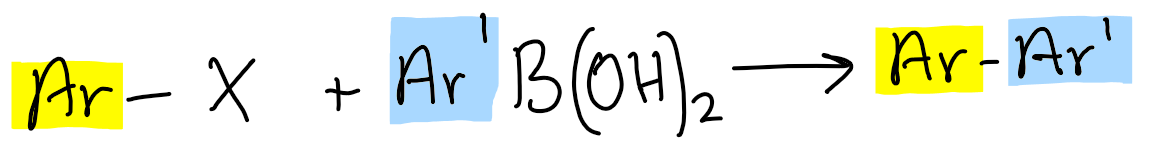
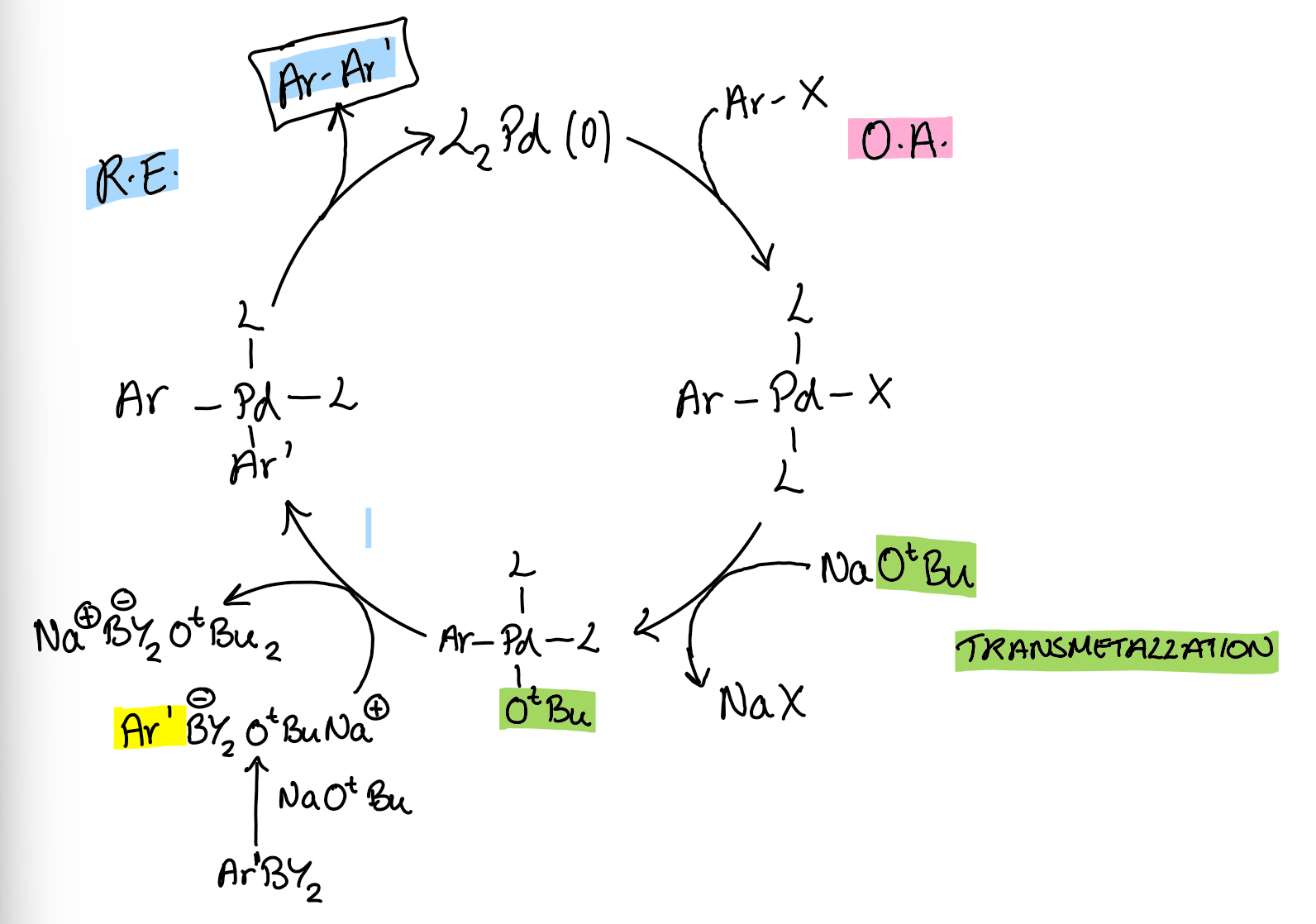
Suzuki cross-coupling cycle
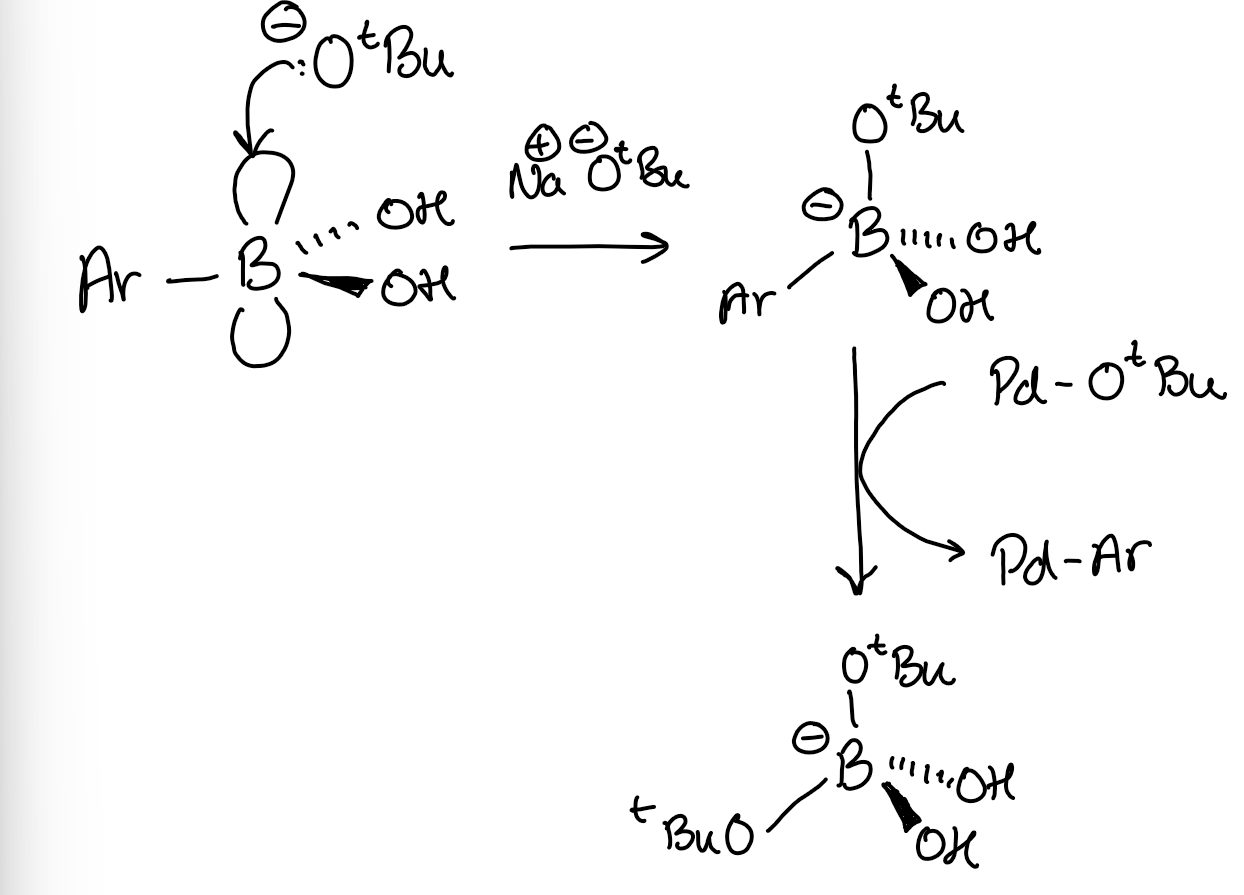
Mechanism for Suzuki ligand exchange step, starting from Boronic acid
What reaction is Buchwald-Hartwig used for?
Phenylamines by adding to aromatic ring
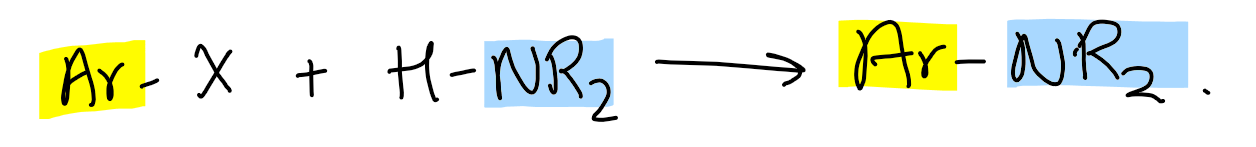
Buchwald-Hartwig cross-coupling cycle
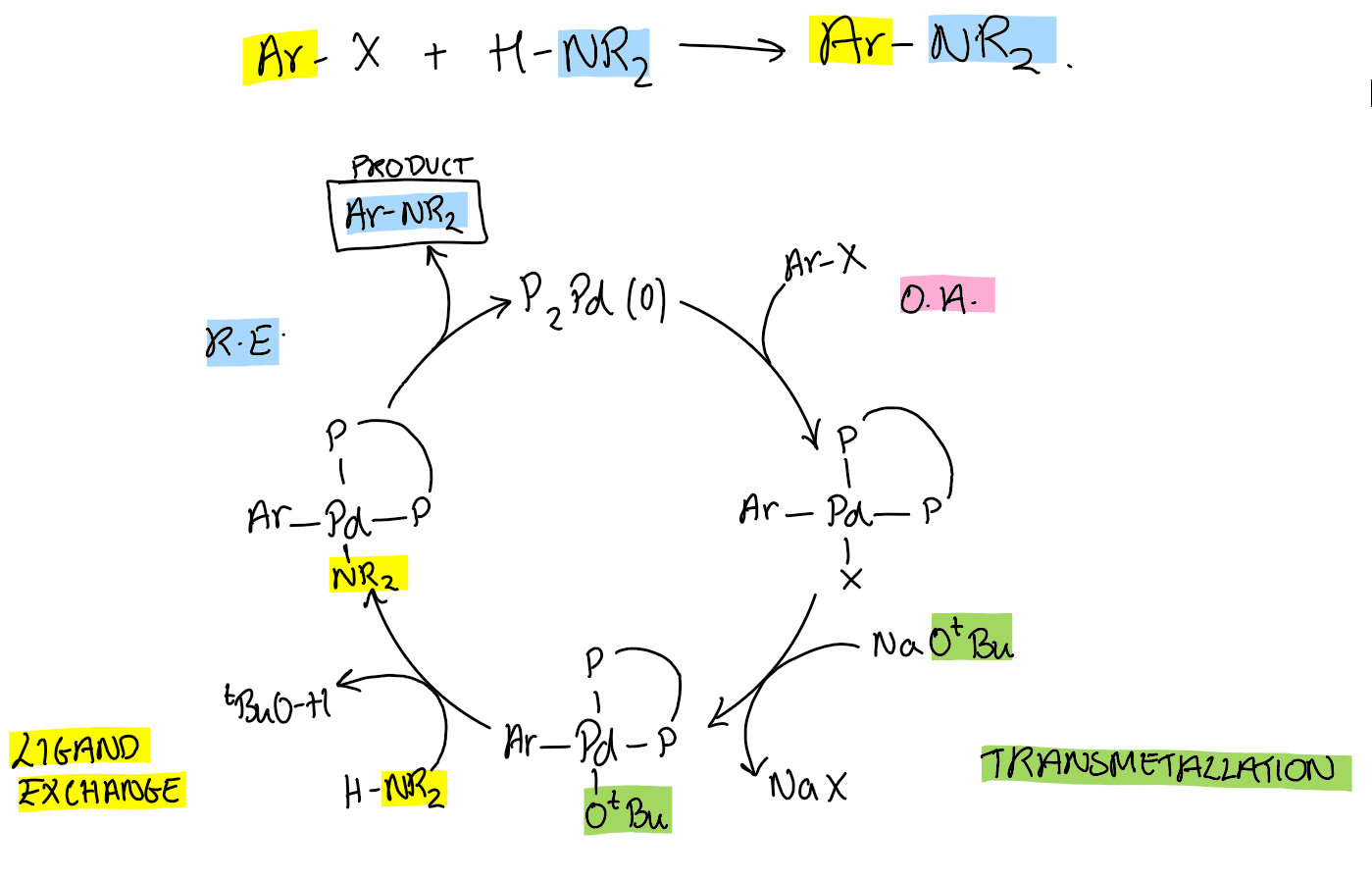
General Pd catalytic cycle