Cybil Board review flashcards
1/103
Earn XP
Description and Tags
Flashcards from Cybil's review powerpoint/review sesssion
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
104 Terms
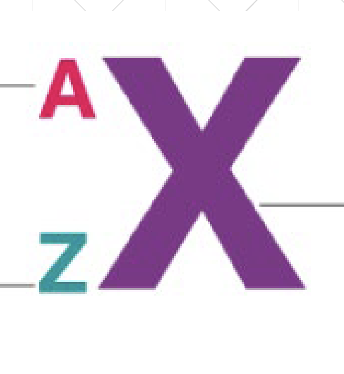
“A” stands for
Mass #
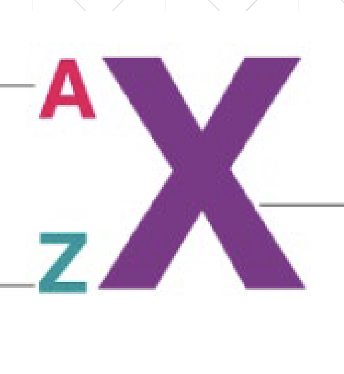
“Z” in atomic notation stands for
Atomic number
Isotopes are
nuclides with the same # protons, but different # mass
Isotones are:
nuclides with the same # neutrons, different atomic #
Isobars are
Nuclides with same mass, different atomic #
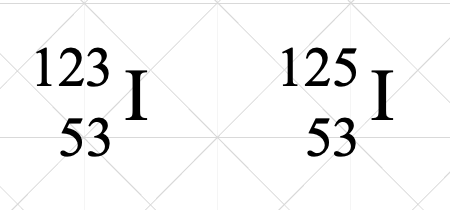
This is an example of
isotopes
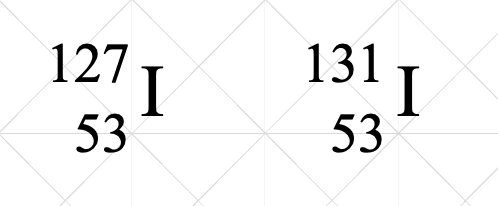
This is an example of
Isotopes

This is an example of
isotones
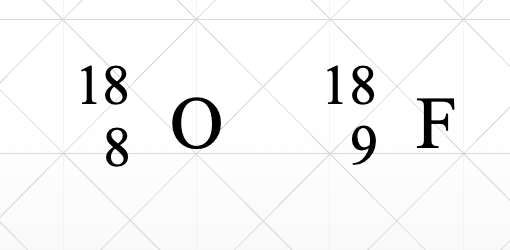
These are an example of
isobars
What are the three types of isobaric decay?
electron capture
beta postiive decay
beta negative decay
In beta negative decay,
A neutron is converted into a proton
If two elements have the same atomic number, but different mass numbers, how are they related?
isotopes
If two elements have the same number of neutrons, but different atomic numbers, how are they related?
isotones
If two elements have the same mass numbers, but different atomic numbers, how are they related?
isobars
Cyclotron produced radionuclides will always decay via:
beta positive decay
F-18 decays via
beta positive decay
indium-111 decays via
electron capture
Tl-201 decays via
electron capture
A nuclide that decays via alpha decay will result in
the mass number decreasing by four
What type of interaction occurs when a photon interacts with an inner shell electron, and all of the energy is absorbed and given to that electron?
photoelectric
During beta negative decay,
a neutron is converted into a proton, and a negatively charged electron is released
Gamma emission is a type of ________ transition/decay
isomeric
The consequence of an unstable nuclei with excess energy is:
emission of a gamma emission
The consequence of an unstable neutron-rich nucleus is:
production of a beta negative particle
describe the process of internal conversion
An unstable nucleus releases gamma ray that interacts with and is absorbed by an inner shell electron
That electron is ejected from the nucleus.
The atom reorganizes: a higher energy outer-shell electron fills the gap, and releases excess energy as a characteristic X-ray
A characteristic x-ray is produced as a result of
the ejection of an inner-shell electron from an atom, and the subsequent filling of that vacancy by an outer-shell electron
Ionization is defined as
the removal of electrons from an atom
What are the two types of particulate radiation
alpha and beta
Bremsstrahlung ionization occurs as a result of
A charged particle (beta negative electron) is rapidly decelerated and deflected by another charged particle (typically an electron near a nucleus
The lost kinetic energy is emitted as an X-ray
a photon is produced by _______, and has ____ mass, and has __ charge, and ___ electrons
gamma and X-ray
no mass
no charge
does not attract or repel electrons
particulate radaition is produced by _______ decay, and has ____ mass, and __ charge, and ___ electrons
alpha and beta
has mass
has charge
attracts or repels
The photoelectric effect occurs when
What type of interaction occurs when a photon interacts with an inner shell electron, and all of the energy is absorbed and given to that electron
resulting in the emission of a characteristic X-ray
Compton scatter occurs when
a photon interacts with an outer shell electron, and only part of its energy is absorbed and given to that electron
The annode within a gas filled detector is:
positively charged
The cathode within a gas filled detector is
negatively charged
Accuracy quality control for a dose calibrator is checking for:
How close the reading is to the true value?
What is the acceptable % error for a dose calibrator readings?
10%
During a linearity test, a dose calibrators calculated % error is 11.1. The technologist should
apply a correction factor
What is the half life of Cs-137
30 years
What is the energy of Cs-137
662 keV
What is the half life for Co-57
271 days
What is the energy of Co-57
122 keV
A technologist performed a constancy test for a dose calibrator and got these results:
Expected Activity: 78.5 uCi
Actual reading: 70.1 uCi
What is the percent error? Can this calibrator be used?
10.7, no
Determine if this dose calibrator needs a correction factor applied for 5 mL syringes based on these results:
5mL syringe
Expected reading: 2.95 mCi
Actual reading: 2.45 mCi
What is the percent error? Calculate the correction factor if needed.
17%; 1.2
What can cause a low constancy reading
incorrect high voltage
incorrect settings
source not placed properly in detector
What can cause a high constancy reading for well-counters or thyroid probes
contamination
outside sources placed near by
What is high-voltage checking in a non-imaging scintillation device
aka peaking, done if constancy fails
How can a technologist improve both sensitivity and resolution of a gamma camera at the same time
reducing distance of detector-patietn
______ counts should be collected for a daily uniformity flood on a gamma camera
5-15 million counts
The UFOV is considered to be ___ of the FOV
95%
The CFOV is ____ of the ___
75%; UFOV
What is the half life of F-18
110min
F-18 is produced via
cyclotron
What is the half. life of Cu-64
12.7hrs
Cu-64 is produced via
cyclotron
Ga-68 is produced via
generator
The half life of Ga-68 is
68min
The half life of Rb-82 is
75 sec
Rb-82 is produced via
generatro
The half life of C-11 is
20min
The half life of O-15 is
2 min
The half life of N-13 is
10 min
C-11, O-15, and N-13 are all produced via
cyclotron
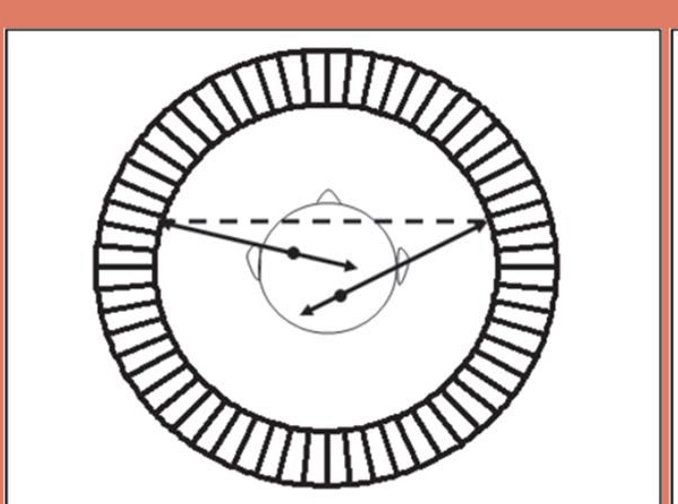
This is an example of
true coincidence
This is an example of a
random/accidental event
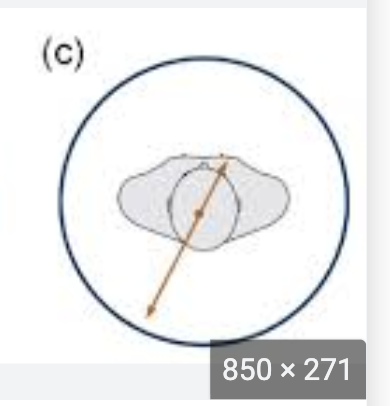
This is an example of
scatter
Time-of-flight in PET
improves signal to noise ratio
mA’s in CT and X-ray represents _____, whereas kVp represents ______
mA’s = amount; kVp = energy
NRC regulation 10 CFR 19 pertains to
notices and reports to employees
NRC regulations 10 CFR 20 pertains to
protection against radiation
NRC regulation 10 CFR part 35 pertains
radioactive materials license (usage, and regulations)

This sign should be posted in areas where a personc can recieve more than _____ mrem in 1 hr
5 mrem

This sign should be posted in areas where a personc can recieve more than _____ mrem in 1 hr
100 mrem

This sign should be posted
to indiciate any area in which certain quantities of radioactive materials are used or stored
Radiation safety education is required for all workers likely to receive greater than
100 mrem
T/F: radiation safety education is universal
false
T/F: radiation safety education is specific to the license
true
Unrestricted areas are considered
Anywhere a person would recieve less than 2mrem in any hour
less than 100mrem in any year
does not include contribution from patients
Restricted areas:
are areas where you can receive greater than 2mrem in any hour
signs should be posted
access controlled
Sealed source leak testing should be done:
every 6 months
radionuclidic purity testing must be performed
after every elution
Sealed source innovatory should be done
every 6 months
Radioactive waste should be decayed in storage until
it measures at or below background
Removable contamination surveys (wipe tests) should be performed
on a resonable schedule
Which of the following is NOT required by the NRC to be posted?
NRC form 5
NRC form three is
notice to emloyees
NRC form 4 is
Cumulative occupational dose history
NRC form 5 is
occupational dose record for a monitoring period
An occupationally exposed radiation worker has declared pregnancy. What is the maximum allowable radiation dose to the fetus?
500 mRem
What is the annual Whole-body (TEDE) occupational limit?
5 REM
What is the annual lens of the eye occupational limit?
15 REM
What is the annual dose to any organ occupational limit?
50 REM
What is the annual dose occupational limit to skin or extremities?
50 REM
What is the exposure limit to the public?
0.1 REM
What is the exposure limit to minors?
0.5 REM
What is the exposure limit for family members of therapy patients?
0.5 REM
All of the following are considered members of the public except
A person taking public transport
Non-stochiastic effects of radiation are:
deterministic
Stochastic effects are
probalistic
All of the following are early effects of acute radiation syndrome except
cancer