Week 1 - Electrical Signals
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
2 types of Electric signals travel down neurons
Voltage-changes across the neuron’s cell membrane can trigger 2 types of electrical signals:
Graded potentials
Action Potentials
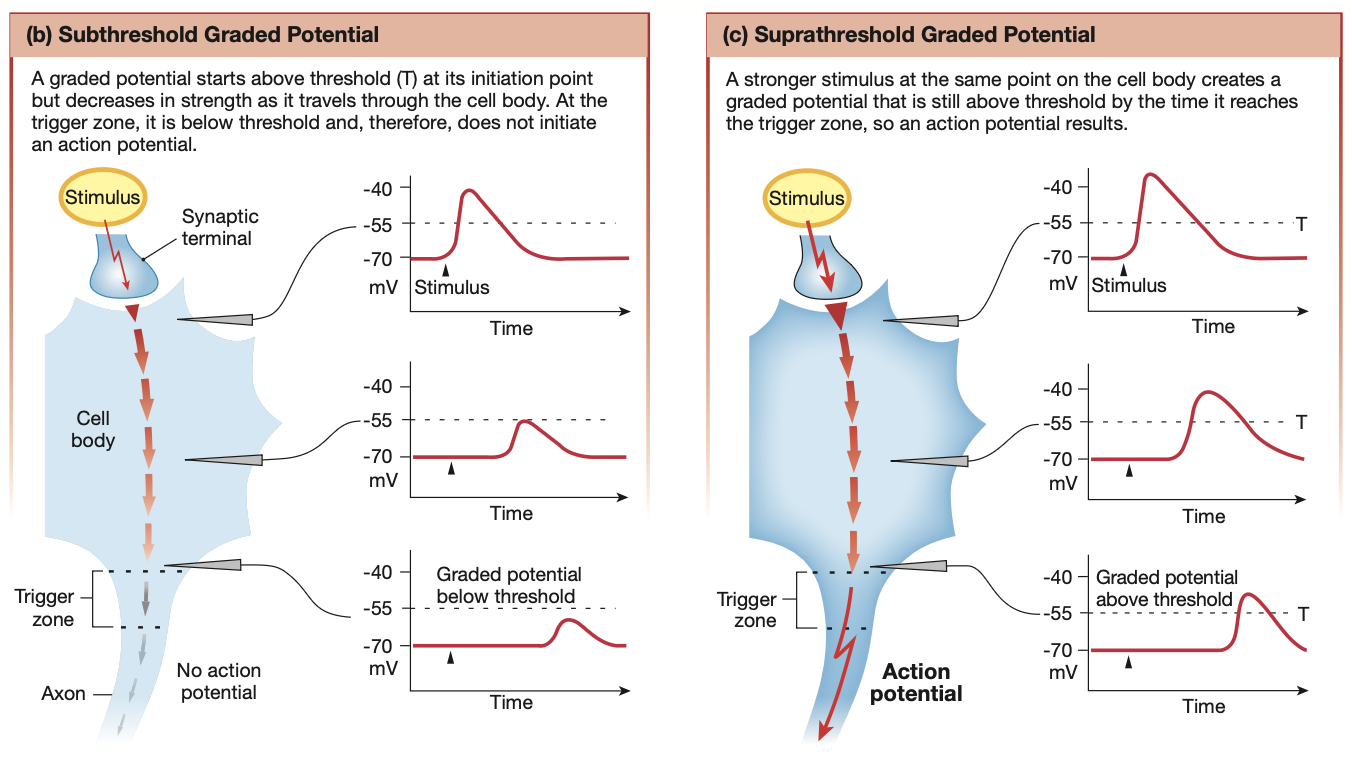
What are graded potentials?
What?
Vary in strength, but cause changes to the neuron’s membrane potential.
Travel over short distances and lose strength as they travel through the cell
Strength is proportional to distance travelled within the neuron
If strong enough to pass threshold potential, becomes an action potential
Used for short distance communication
Dendrite, through to cell body. Stops at trigger zone
If strong enough via signal summation, can initiate an action potential
Where and how do graded potentials occur?
Where?
Dendrites & cell body
How?
mechanical, chemical or voltage-gated ion channels
Ions involved: Na+, K+, Ca2+ & Cl-
Trigger Signal
Ions entering through gated channels
Signal type
More positive: Depolarising (e.g Na+ entering) - excitatory
More negative: Hyperpolarising (e.g Cl- entering or Na+ leaving) - inhibitory
What are action potentials?
What?
Brief, large depolarisations that travel the entire length of the neuron
The electrical signal is strong, All-or-nothing
Where and how do action potentials occur?
Where?
Trigger zone of axon / axon hillock
How?
Voltage-gated ion channels
Ions involved: Na+ & K+
Trigger Signal
A graded potential that pushes the membrane potential above the threshold potential in the axon hillock
This opens ion-channels, letting Na+ and/or K+ rush in
Signal type
Depolarising (e.g Na+ entering)
Threshold potential
-55mv
Action potential: Depolarisation
Graded potential reaches threshold potential of -55mv
All voltage-gated Na+ channels along the axon hillock open quickly, voltage-gated K+ channels start opening slower
Na+ rushes in
Membrane potential becomes extremely positive (+30mv)
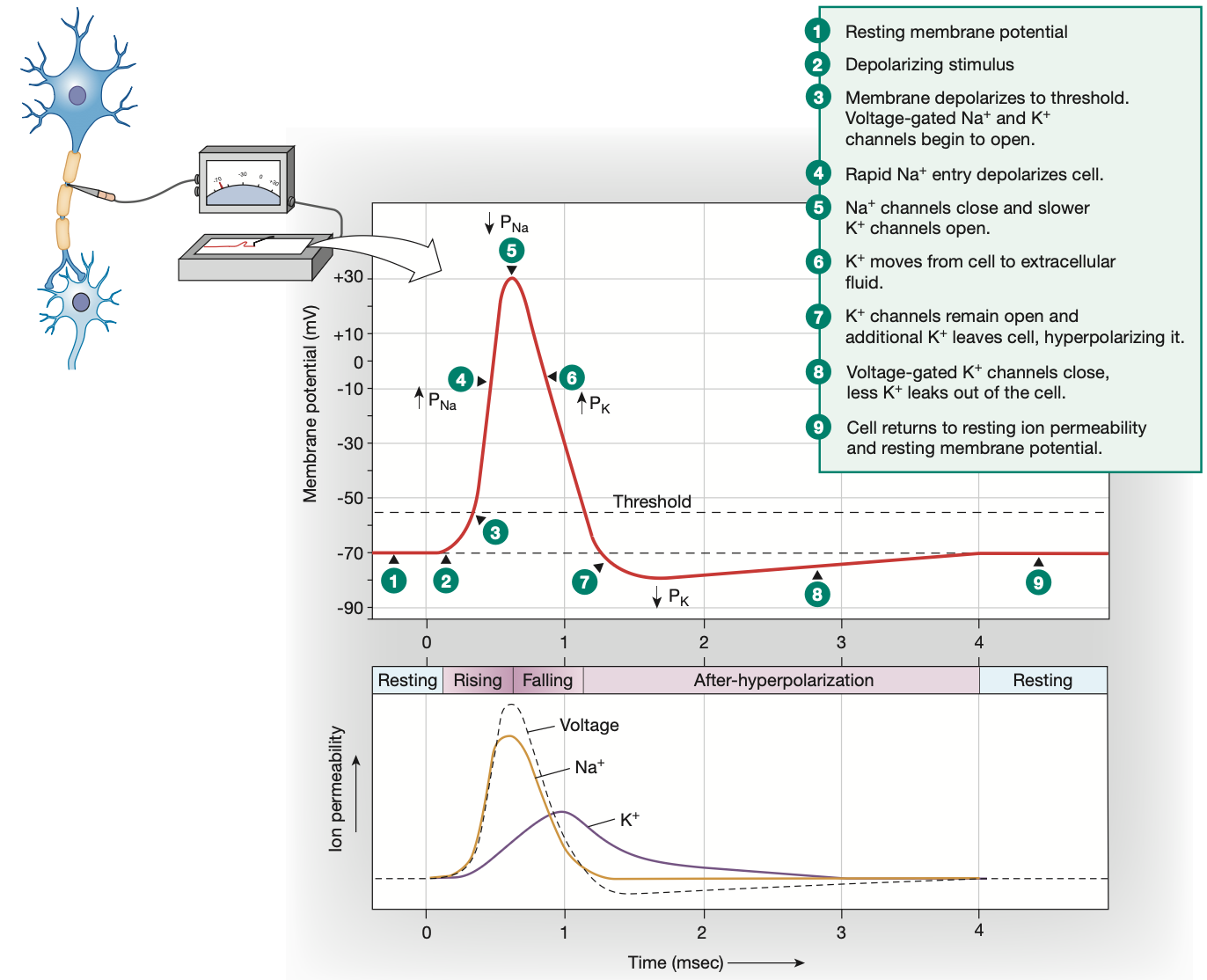
Action potential: Repolarisation
Voltage-gated Na+ channels shut quickly to stop Na+ from flooding in further
At the same time, slow voltage-gated K+ channels fully open
K+ ions move out of the cell to counteract the high positive charge brought in by the excess Na+ ions
Voltage-gated K+ channels remain open
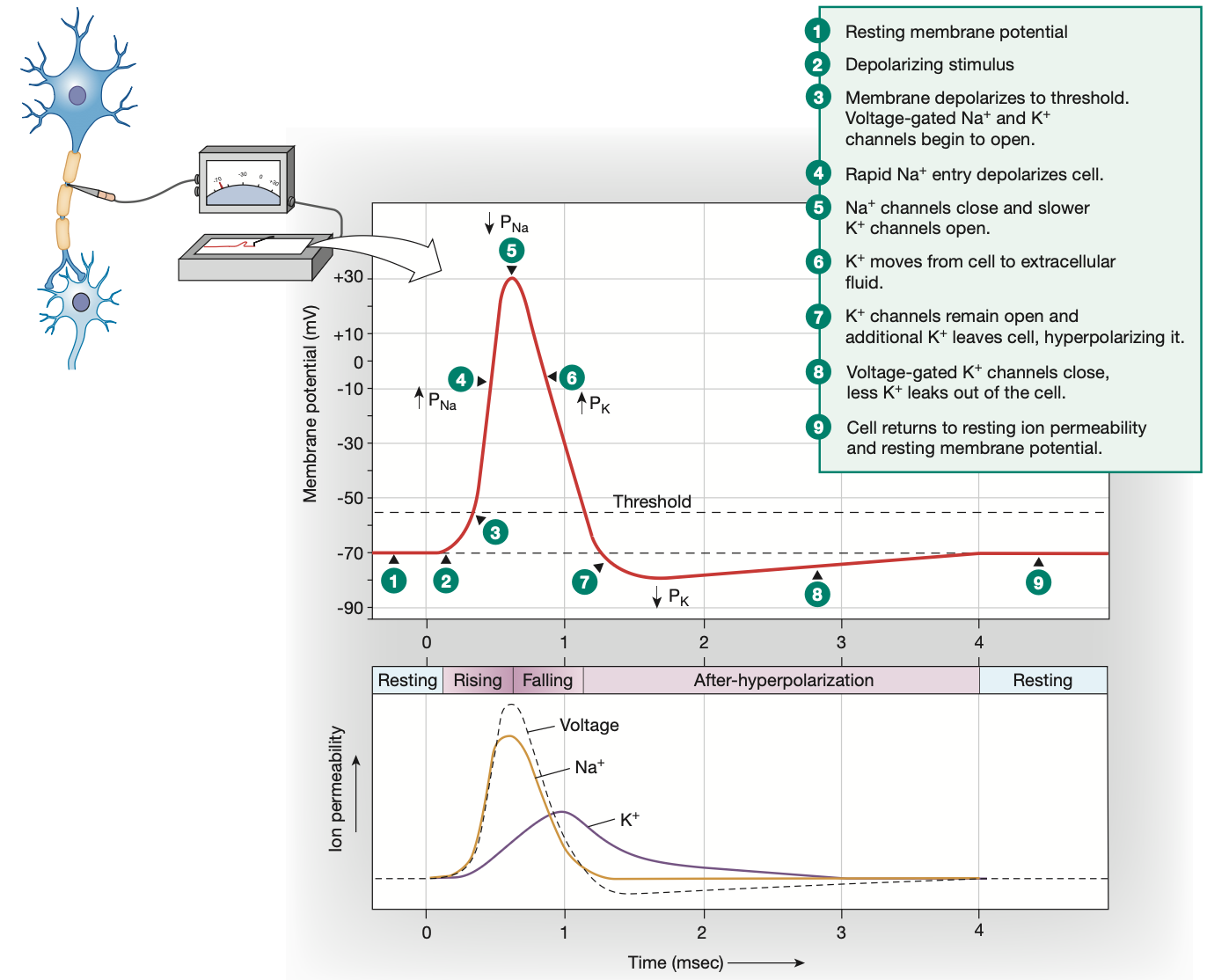
Action potential: Hyperpolarisation
Neuron overreacts, leaving voltage-gated K+ channels open, causing excess K+ to leave the cell, alongside leaky K+ channels working overtime
Voltage-gated K+ channels are slow to close
Excess K+ leaves the cell, overshooting the resting membrane potential and making the cell membrane too negative
This hyperpolarisation is the reason for the absolute & relative refractory periods lasting so long, preventing another action potential from happening too soon
Leaky channels and 3Na-2K pump work to have neuron return to resting membrane potential (a.k.a homeostasis)
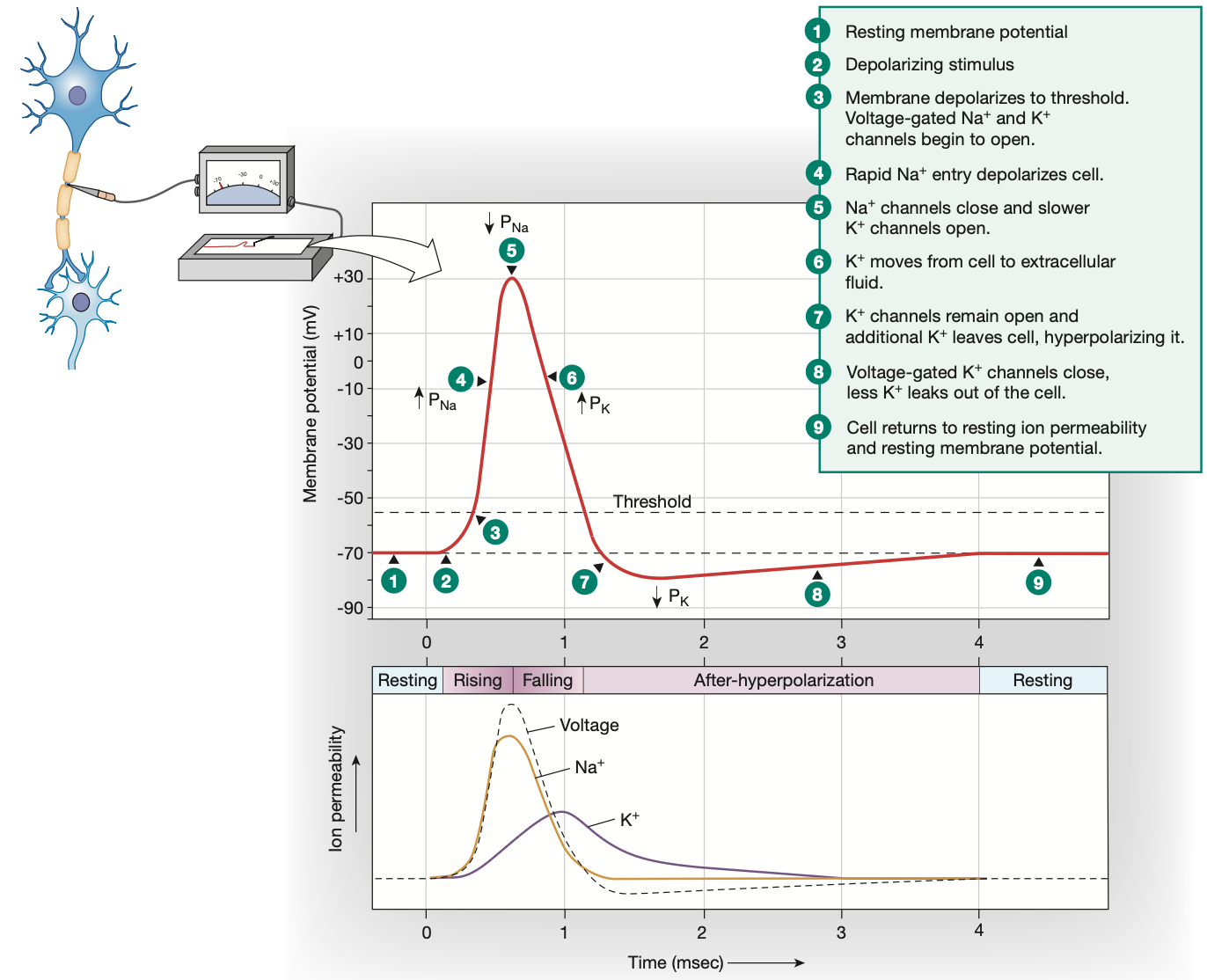
4 states of Voltage-gated Na+ channels
Closed: Resting membrane potential, Na+ can only enter via leak channels
Open: During depolarisation phase of action potential, Na+ rushes into cell
Inactivated: During an action potential, when the membrane potential reaches its peak of +30mv, the inactivation gate quickly closes the voltage-gated channel, preventing further entry of Na+. This inactivation gate also lengthens the absolute refractory period.
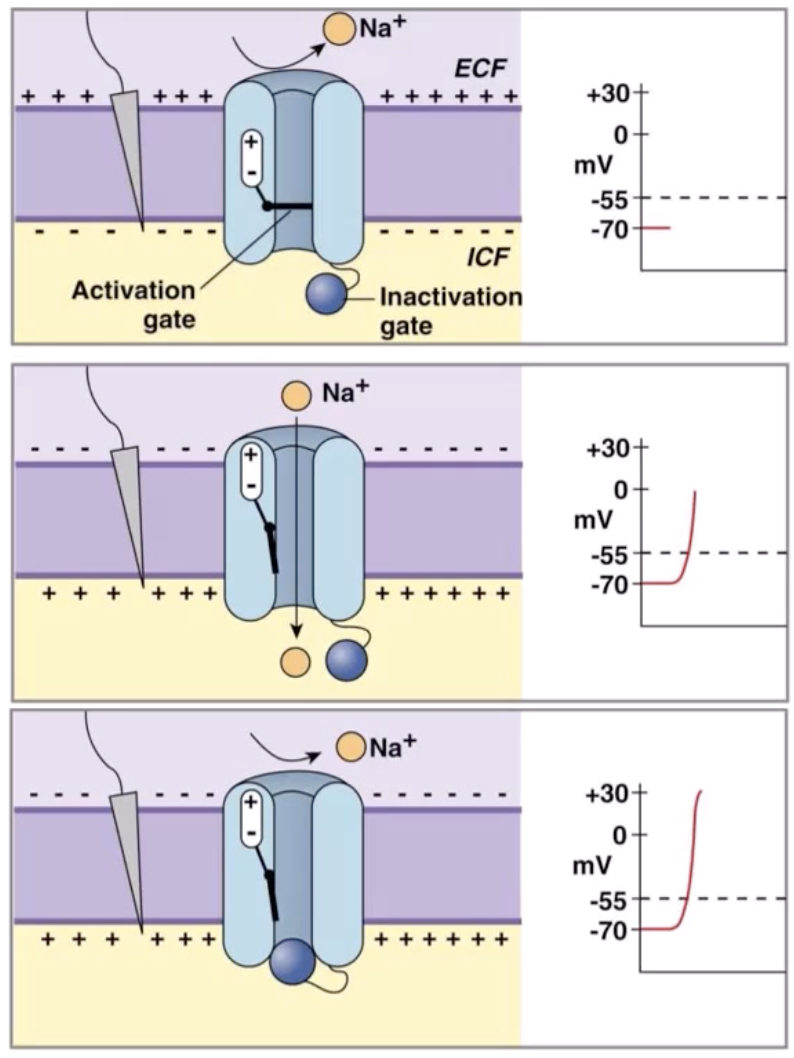
Absolute vs Relative refractory periods
Absolute refractory period
Time taken from the start of an action potential, until after depolarisation phase of action potential where voltage-gated Na+ channels to transition from inactivated (open gate, closed ball), to closed but activated (closed gate, open ball, state found at resting membrane potential).
Absolutely no action potential can occur during this 1-2 millisecond phase
Relative refractory period
Some voltage-gated Na+ channels have returned to their closed, but activated positions.
Voltage gated K+ channels are still open.
Due to the membrane potential still being hyperpolarised, the threshold potential is further away than usual (however it is still -55mv).
Thus, an oddly strong graded-potential will be required to trigger an action potential.
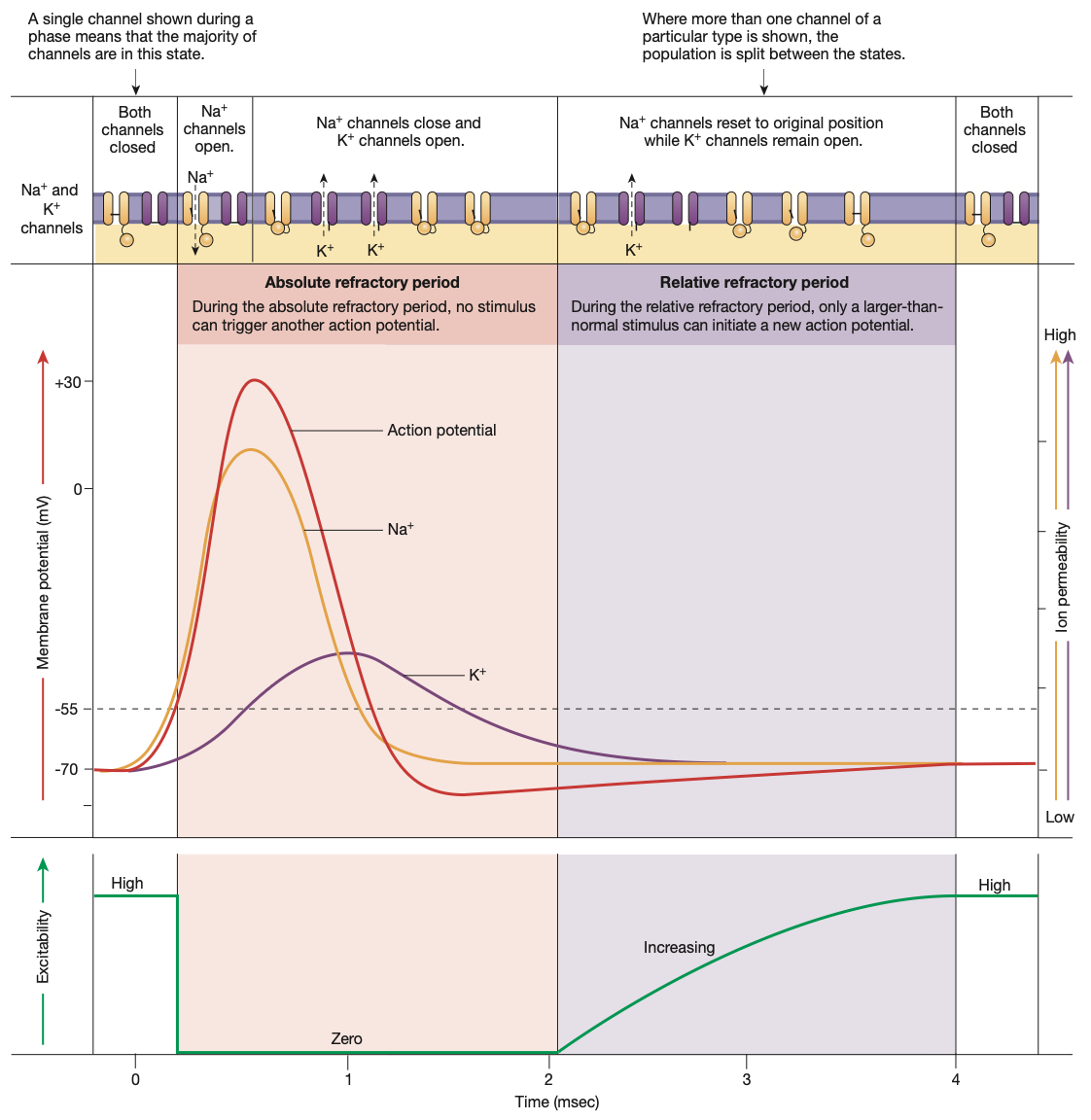
Why action potentials move in one direction? Conduction of Action Potentials
Action potentials are triggered by voltage-gated Na+ ion channels opening
Voltage-gated ion channels are triggered by disturbances in the surrounding cell membrane potential. (change comes from the rushing in of Na+ from said gates opening)
Hence, when an action potential begins in the axon hillock, the depolarisation travels down the axon due to neighbouring voltage-gated Na+ channels.
Much like a ripple on a lake
However, the ripple (action potential) does not travel backwards due to the previous section of the axon being in the refractory stages.
Hence, the axon potential shoots down the axon, towards the axon terminal.
In skeletal muscle cells/fibres, action potentials are recieved and propagated along the sarcolemma a true ‘ripple’ fashion.
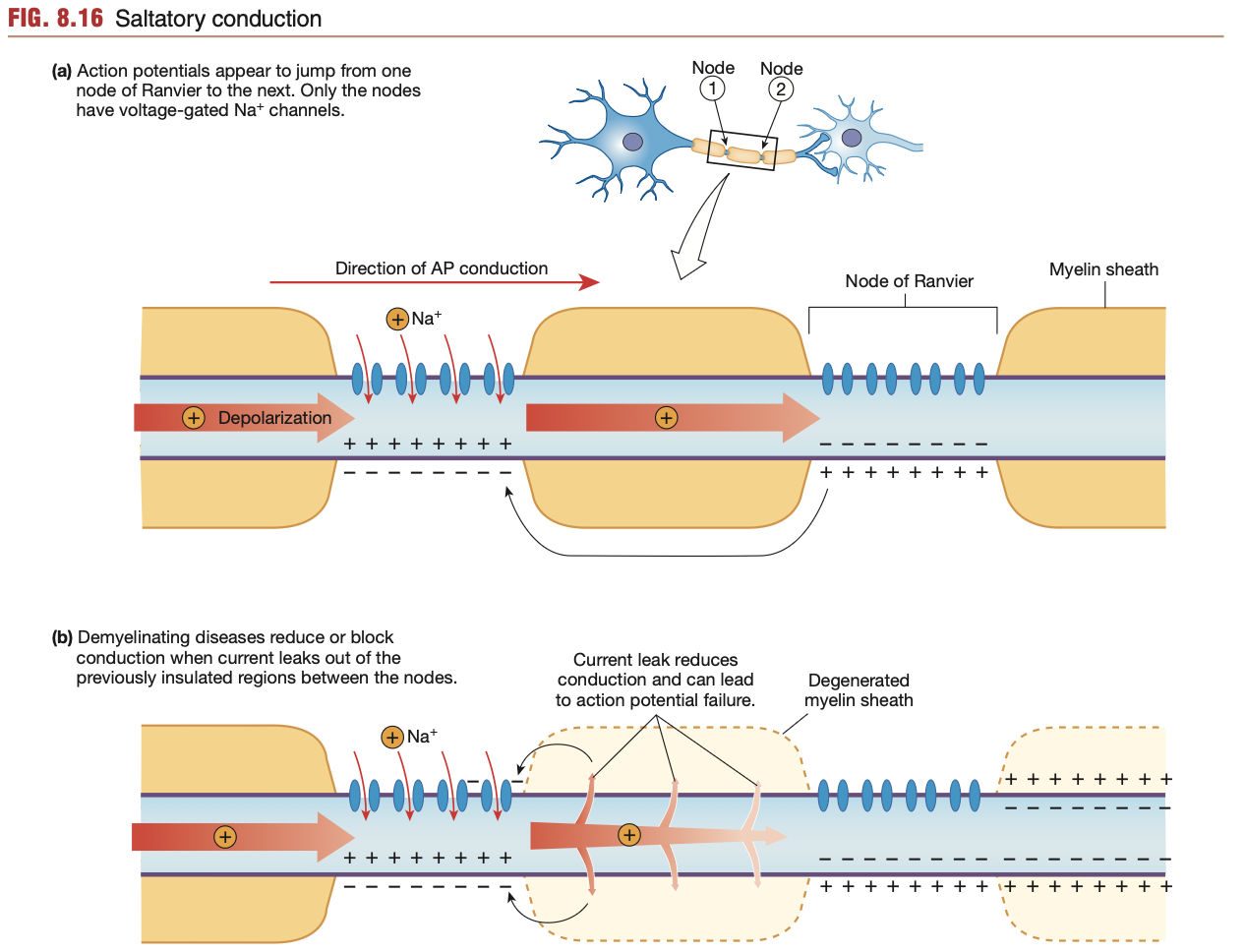
Saltatory conduction: Axon potentials
In unmyelinated axons, opening of voltage-gated Na+ channels along the entire axon takes time, especially in long neurons
Process of Saltatory conduction:
Depolarisation occurs at the trigger zone/axon hillock
Initial batch of voltage-gated Na+ channels open, letting Na+ rush into the cell.
Eventually, the depolarising neuron membrane stops, as a myelin sheath is reached (sometimes Schwann cell)
In myelinated axons, voltage-gated Na+ channels along the axon can only be found along the nodes of Ranvier (refer to diagram)
Hence, the inner steep Na+ gradient (from previously opened voltage-gated Na+ channels is able to rush past myelin sheath segments inside the neuron without risk of Na+ leaking out from leaky channels. (i.e no current leak)
Hence, myelin sheaths allow the axon potential to “skip” along nodes of Ranvier, while also preventing leaks of Na+ ions from leaky channels.
More myelin sheath, the faster an action potential, the less likelihood of an action potential dissipating
Lack of a myelin sheath can hinder action potentials greatly, as found in ailments such as multiple sclerosis (degradation of myelin sheath).