Lecture 12 (Protein Purification, Detection & Mass Spectrometry)
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
What are the 3 major components of protein purification?
Solubilization
Separation
Protein detection
Why is E. coli often used for protein expression?
Engineered E. coli can overexpress high yields of a protein of interest
What is a limitation of protein overexpression?
Engineered cells may lack required post-translational modifications for function
What is cell lysis?
Breaking open cells to release proteins, forming a crude extract/cell lysate
What are common cell lysis methods?
Osmotic shock = Cells in hypotonic solution take up water and burst
Mechanical:
Sonication = Sonication uses ultrasound
French press = Push with high pressure through a small hole
What factors stabilize purified proteins?
Correct pH (can easily denature by changes in pH → need buffers)
Cold temperature
Protease inhibitors (proteases can destroy protein of interest)
Detergents (hydrophobic proteins need to be kept soluble)
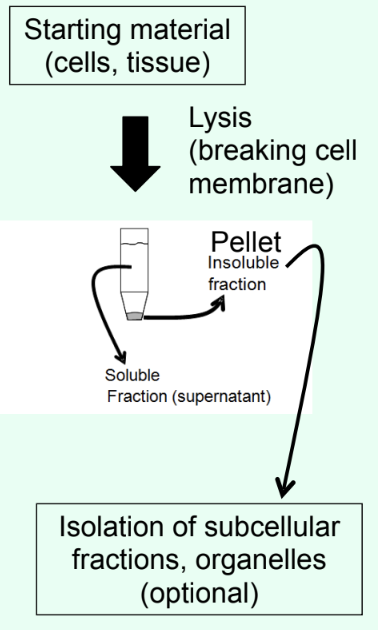
What is the pellet vs supernatant in centrifugation?
Pellet = insoluble material
Supernatant = soluble fraction (where protein of interest resides)
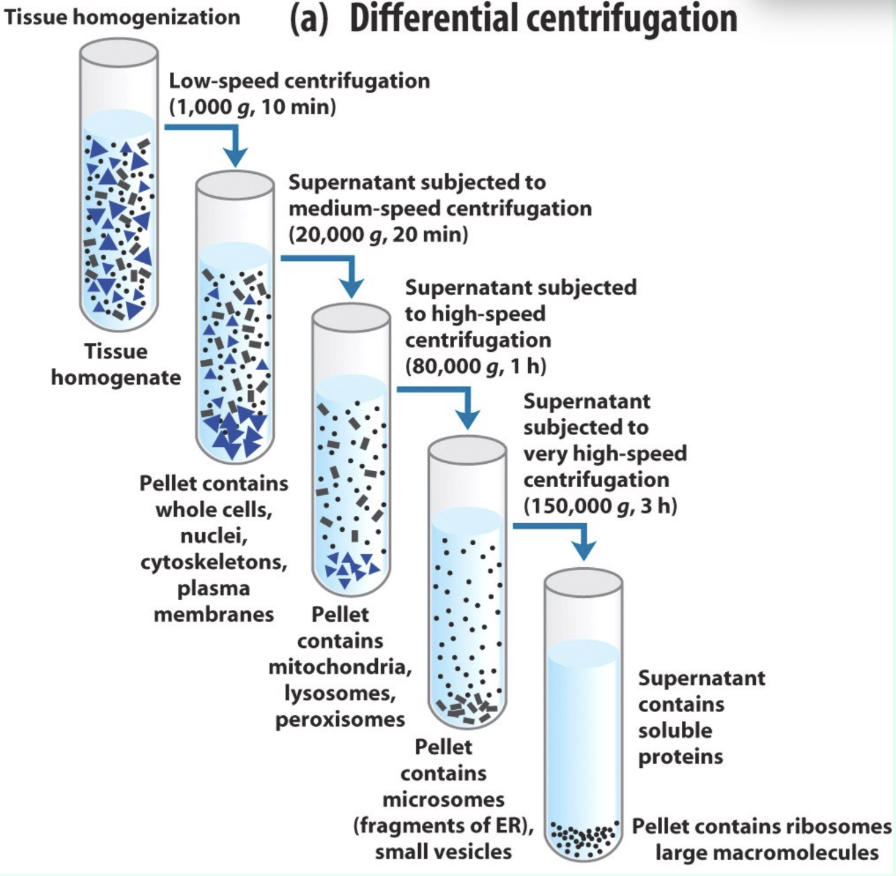
What is differential centrifugation?
Sequential spins at increasing speed to pellet different organelles/fractions
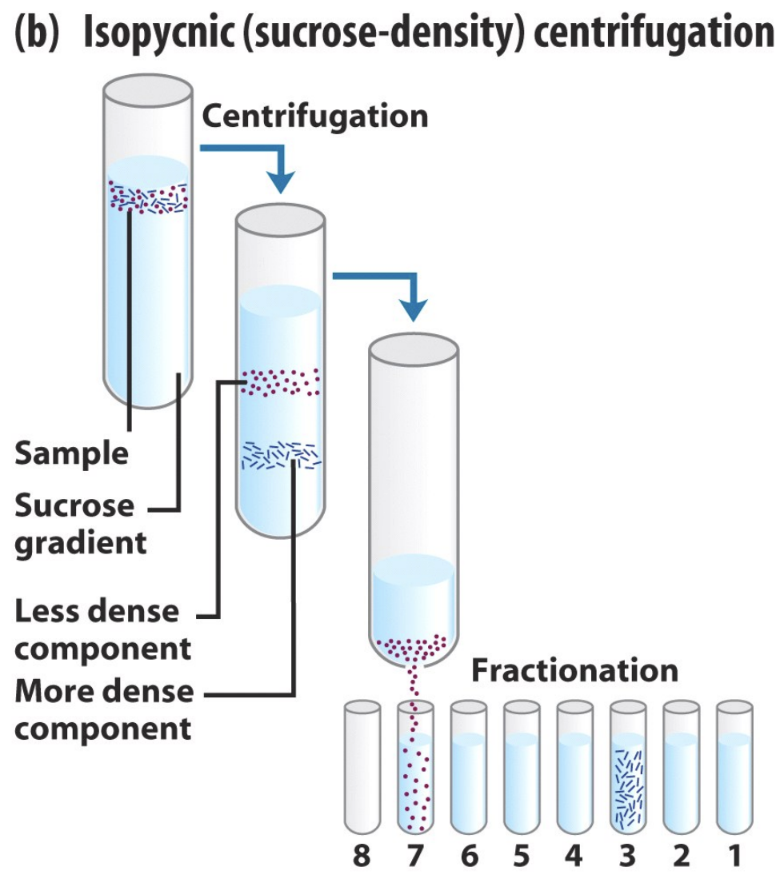
What is isopycnic centrifugation?
Organelles sediment in a density gradient until buoyant density matches
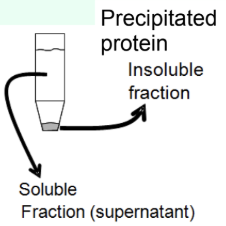
What is protein precipitation used for?
Reduce solubility
Pellet
Concentrate the protein
Re-dissolve proteins
What is salting in vs salting out?
Salting in keeps proteins soluble
Salting out uses high salt to precipitate/concentrate proteins
What is an ammonium sulfate cut?
Protein precipitation/concentration using ammonium sulfate
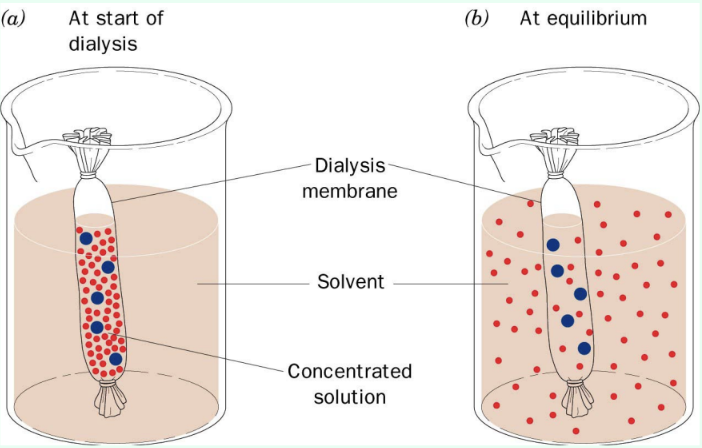
What is dialysis used for?
Semipermeable membrane removes small molecules like salt while retaining proteins
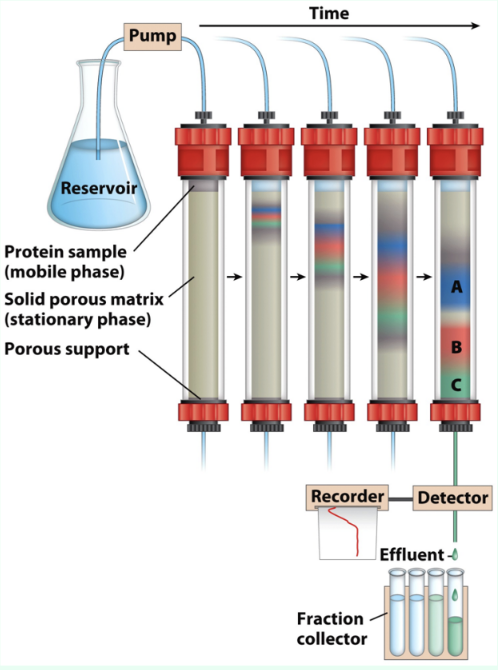
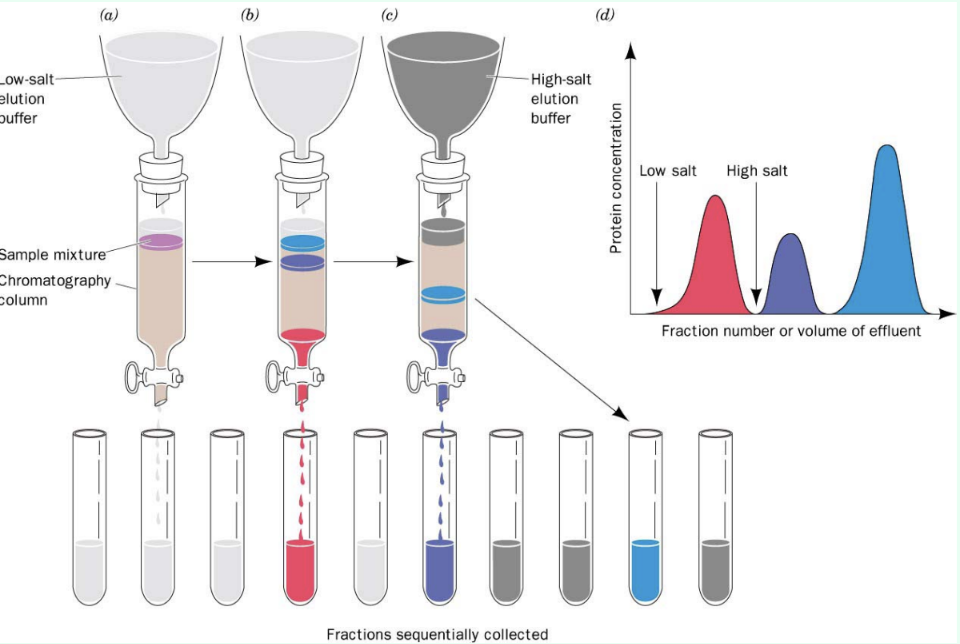
What is chromatography?
Separation/fractionation based on physicochemical properties
What are mobile and stationary phases?
Mobile phase = protein solution (contains protein of interest in solution)
Stationary phase = column matrix (moving solution through column of packed porous solid material)
What is retention time?
Time/fraction when a molecule elutes from a column
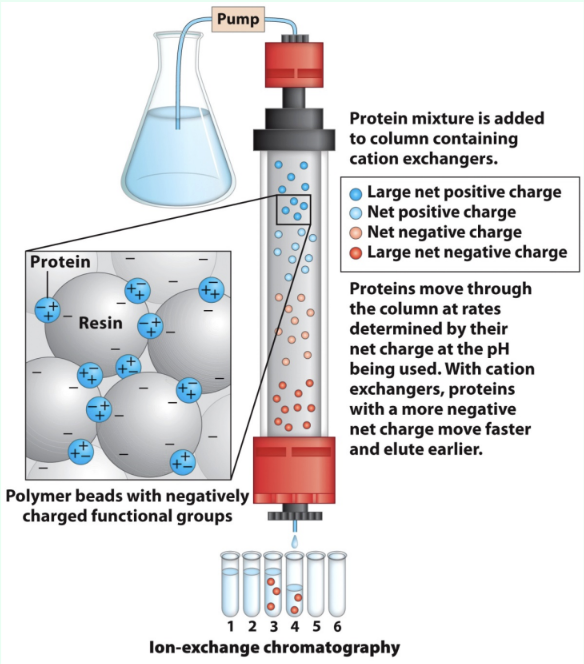
What is ion exchange chromatography?
Separates proteins by charge using charged stationary-phase resin
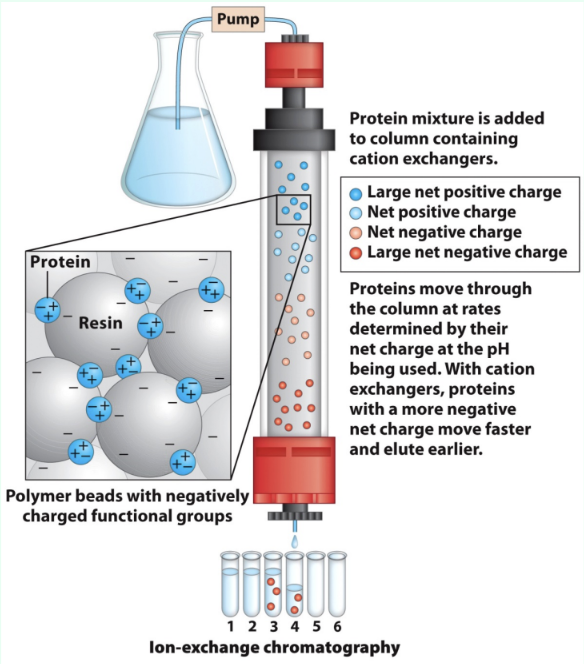
What do anion vs. cation exchangers bind?
Anions exchangers:
Anions
Resin is positively charged
Cation exchangers:
Cations
Resin is negatively charged
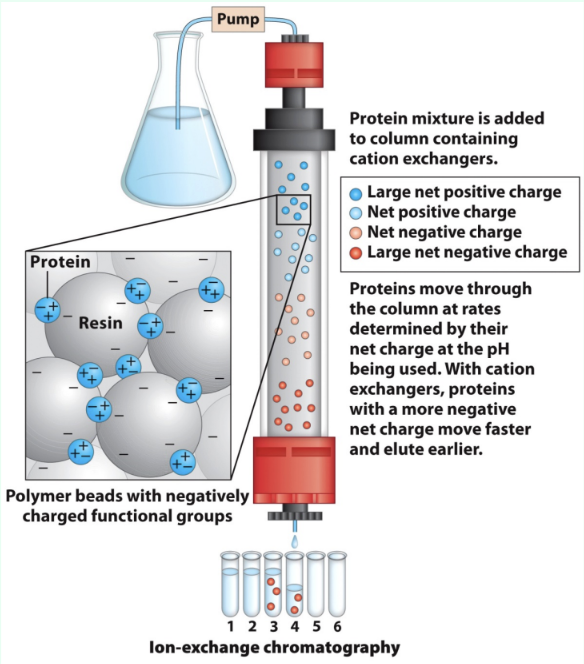
How does protein charge affect ion exchange choice?
Proteins are polyelectrolytes (have both negative/positive charges and their net charge depend on pH)
Acidic proteins → anion exchange
Basic proteins → cation exchange
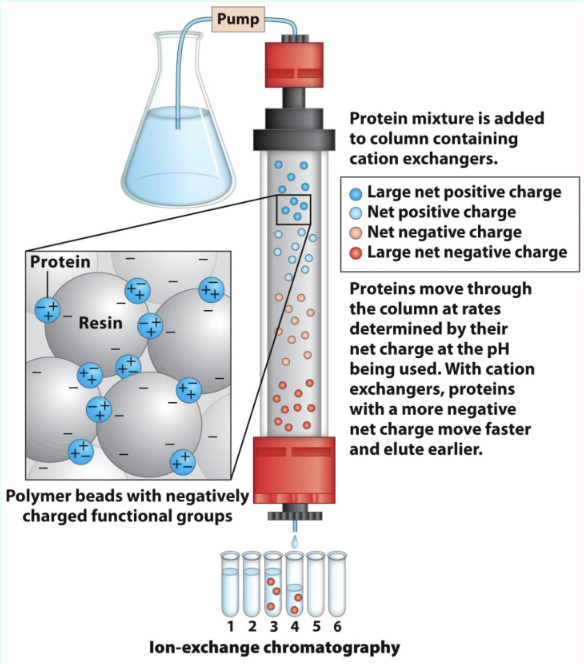
How do proteins elute in ion exchange?
Lower-affinity proteins elute faster
High salt competes off stronger binders
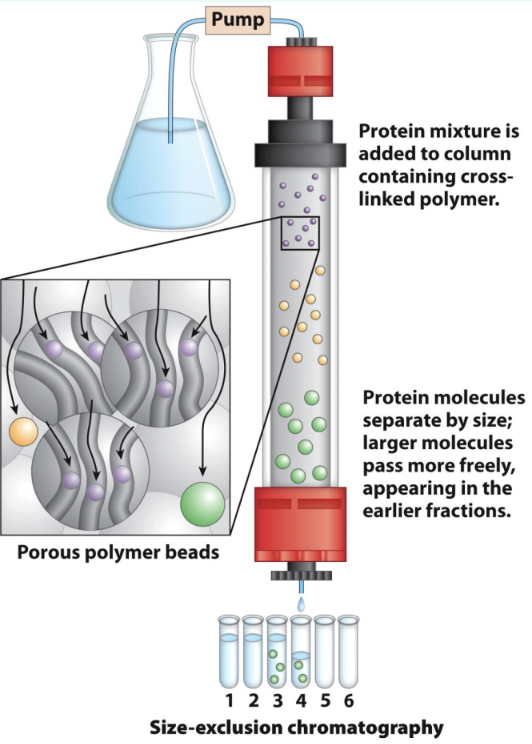
What is gel filtration chromatography?
Size-exclusion chromatography that separates proteins by size
Dilutes the sample
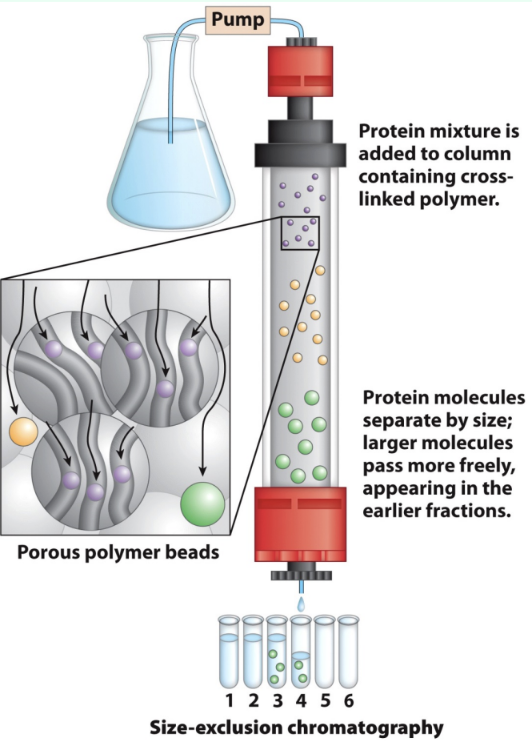
How do large vs small proteins behave in gel filtration?
Large: Cannot enter beads → small void volume → elute first
Small: Enter bead pores → larger volume → elute later
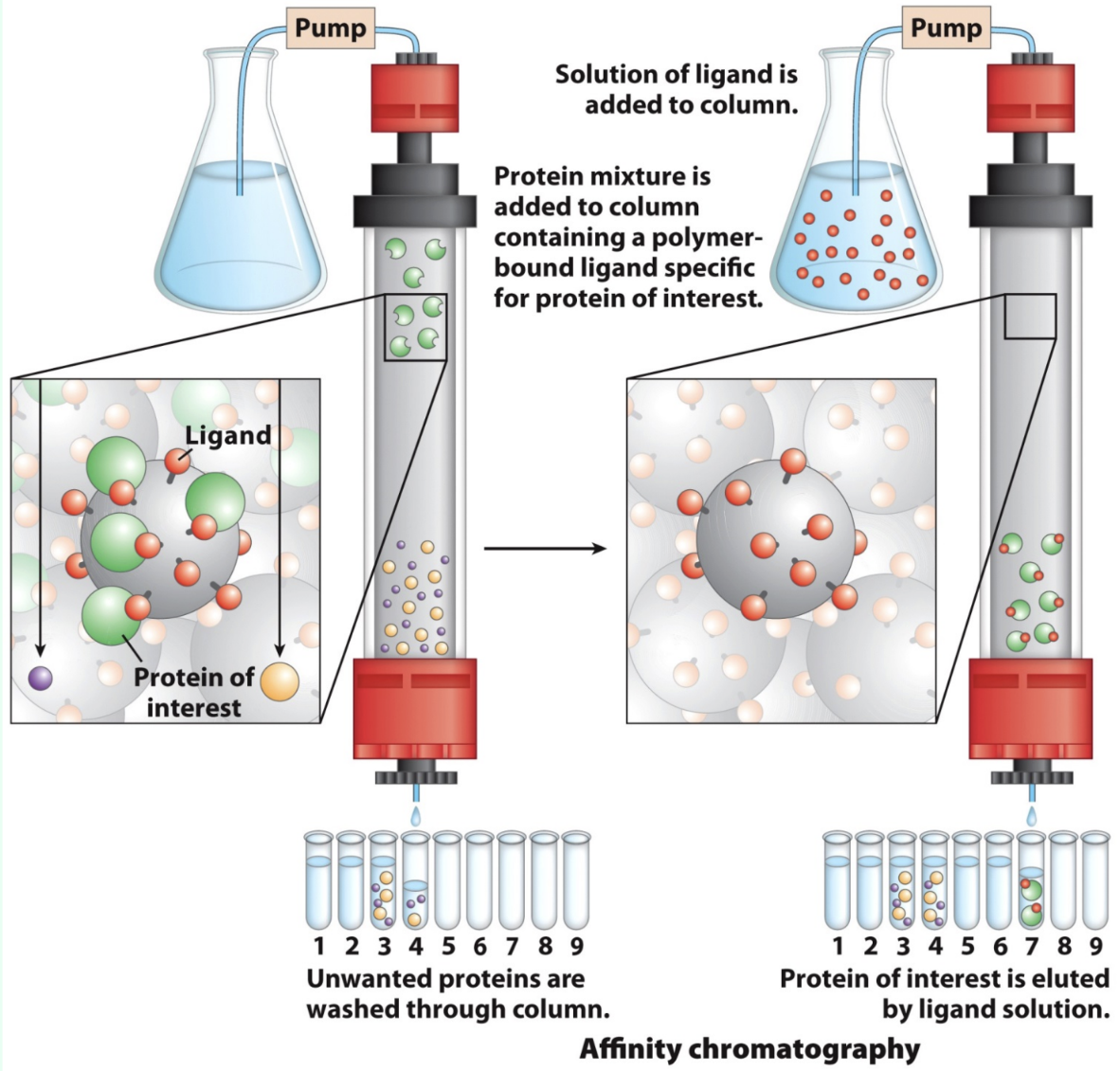
What is affinity chromatography?
Stationary phase binds only the target protein, allowing one-step purification
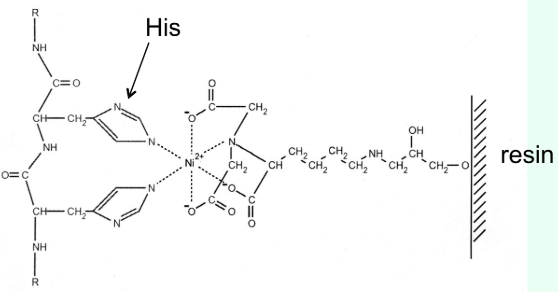
What is the most common affinity tag?
Six-His residue tag at the N- or C-terminus
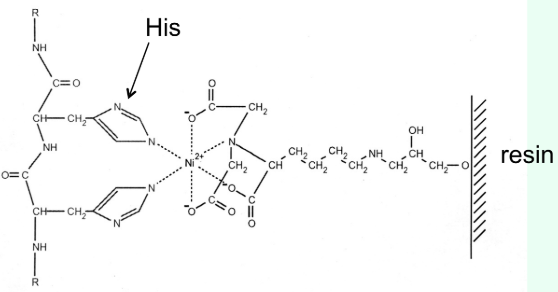
How does nickel affinity chromatography work?
His-tag binds Ni2+ resin (stationary phase)
Imidazole competes off and elutes the protein
How are chromatography fractions collected?
As fractions using a fraction collector
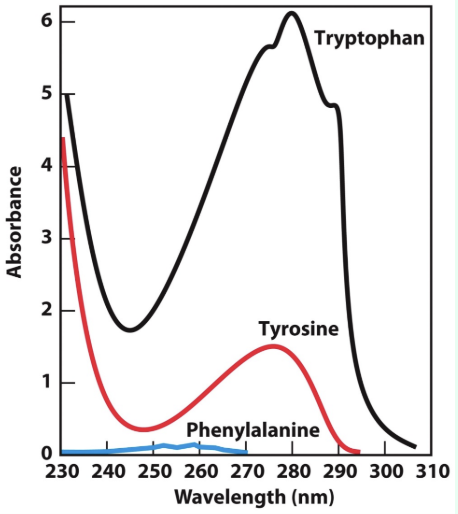
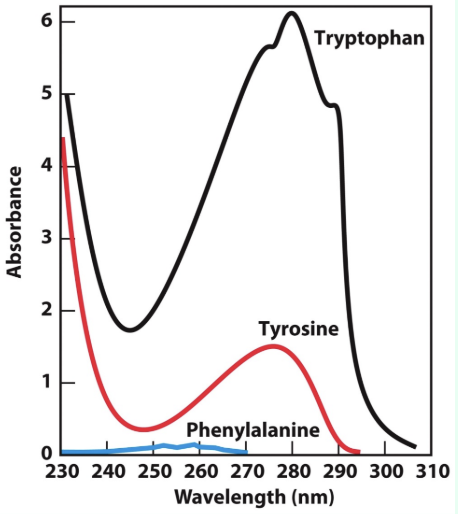
What absorbs at 280 nm?
Mostly Trp and Tyr, plus a little Phe
W > Y > F
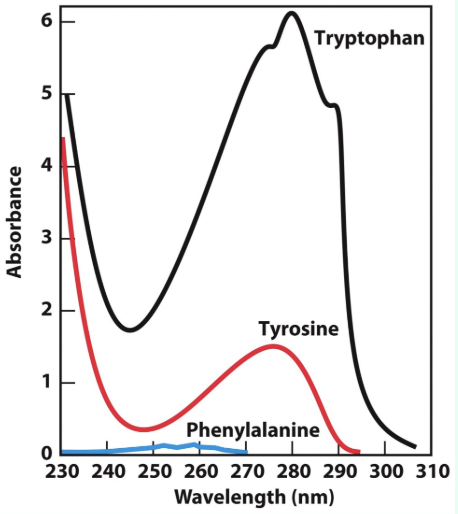
What absorbs at 214 nm?
Peptide bonds
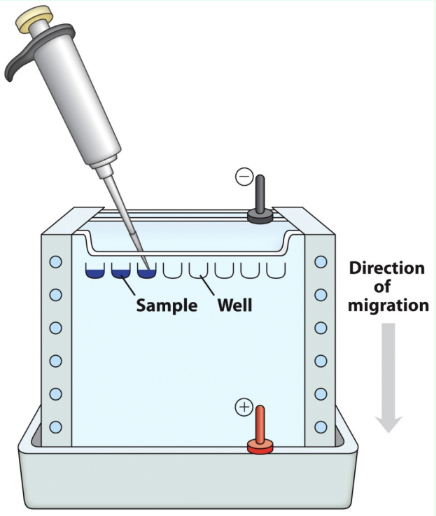
What is electrophoresis?
Separation of charged proteins by migration in an electric field
What is PAGE?
Polyacrylamide gel electrophoresis
Gel acts as a molecular sieve
Slows migration of proteins based on their charge-to-mass ratio and their shape
What can electrophoresis reveal?
Protein purity
Approximate molecular weight
Isoelectric point (pH at which net electric charge = 0)
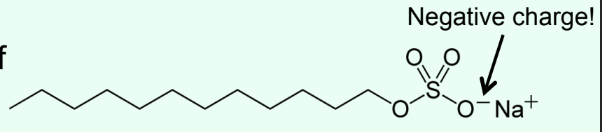
What is SDS-PAGE used for?
SDS = Sosium dodecyl sulfate
Estimating purity and molecular weight
Uses heat to denature proteins and gives large negative charge
How can SDS-PAGE bands be identified?
Excise band → digest into peptides → identify by mass spectrometry
What do proteases and chemicals do in protein ID?
Cleave internal peptide bonds next to specific amino acids
What is mass spectrometry?
Measures mass-to-charge ratio (m/z) of gas-phase ions
What is MS/MS used for?
Peptide sequencing/identity confirmation by tandem mass spectrometry
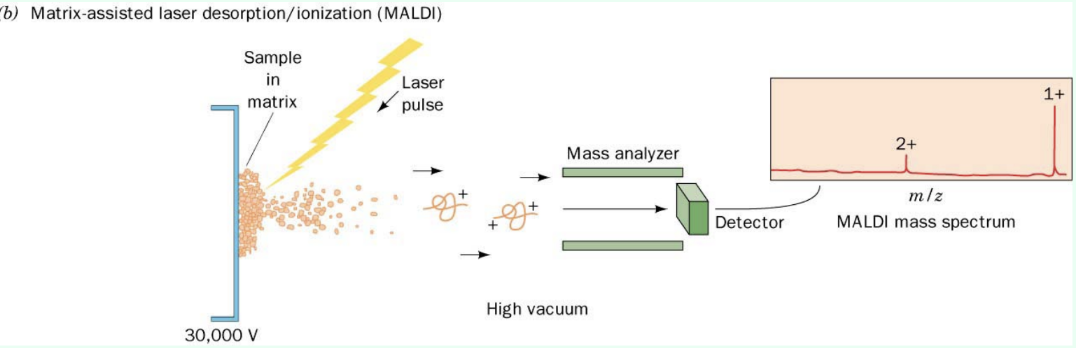
What are the 2 main ionization methods?
MALDI
Protein + matrix is laser-ejected into gas phase, usually +1 charge
Time-of-flight (TOF) depends on mass and charge (m/z)
ESI
High-voltage spray makes charged droplets
Evaporation yields gas-phase protein ions
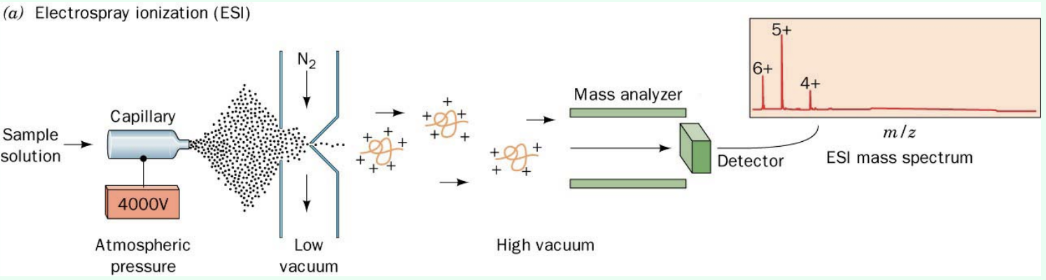
Why is ESI often more accurate than MALDI?
Multiple charge states allow a mass average from many m/z species