503A Pharmacies - Dr. Pond (EXAM 2)
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
What is a 503A pharmacy?
- Pharmacies that compound for individual patients with a valid prescription
- Must adhere to USP 797 and 795
What is the drug quality and security act?
- Its purpose was to differentiate between compounding pharmacies that compound prescriptions for individual patients vs. pharmacies that compound large batches for resale
- 503A is for patients where a prescription is required either when they are getting the product or sometime after whereas with 503B, pharmacies compound large batches for resale
Do 503A pharmacies follow 501(a)(2)(B), 502(f)(1), and 505 of the FD&C?
No xoxo
What are additional stipulations for 503A pharmacies?
- Compounding must be based on the receipt of valid prescription for an identified individual patient
Can you essentially copy a commercially available drug in a 503A pharamcy?
No! 503A states that drugs should not be regularly compounded when there is a market available copy. This prevents unnecessary compounding + risks that come from compounding (mistakes, risk of infection, poor ingredient quality)
*EXCEPTION IS DRUG SHORTAGE, THAT WOULD BE OK
What are examples of 503A pharmacies?
- Independent community compounding pharmacies
- Hospitals/heath systems
- Infusion center
- Home infusion pharmacies
- Long term pharmacies
Who oversees 503A pharmacies?
State Board of Pharmacies
What does the FDA encourage for hospitals/health systems?
Register as 503B pharmacies + look to products that are made by 503B pharmacies
Should hospitals regularly be batching?
No! Acceptable in cases of emergency situations
Should hospitals be delivering compounded products to other health systems outside of their own?
No
The FDA will not take action against a hospital/health system if...
•The compounded drug product is administered only to patients in hospital or health system
•Compounded drug products are discarded within 24 hours of transfer out of the pharmacy
•Products are compounded in accordance with all other FD&C Act and FDA regulations
The FDA will crack down on a hospital/health system if...
•Lack sterility assurance
•Non-patient specific batching products not for emergency use
•Routine large amounts of non-patient-specific compounded drug products
•Distribute interstate large amounts of non-patient specific compounded products
•No procedures to obtain compounded drugs from 503B pharmacies
What situations often require compounded products?
•Allergies to excipients
•Problems with adherence
•Bad tasting medications
•Commercially unavailable dose
•Drug shortage --> this is more of the bread and butter of 503B pharmacies (e.g. for GLP compounding)
503A pharmacies must adhere to USP....
795 (nonsterile) and 797 (sterile)
USP 795 describes....
the minimum standards for compounded nonsterile preparations for humans and animals
What preps fall under USP 795?
•Solid oral preparations
•Liquid oral preparations
•Rectal preparations
•Vaginal preparations
•Topical preparations (i.e., creams, gels, and ointments)
•Nasal and sinus preparations intended for local application (i.e., nasal sprays and nasal irrigation)
•Otic preparations (excluding use in perforated eardrums)
Does reconstitution (e.g. adding water to powder for reconstitution) fall under 795?
No, this is not considered compounding
Does repacking fall under 795?
No
Does splitting tablets fall under 795?
No
Does crushing or opening tablets to facilitate patient dosing fall under 795?
No
What are the responsibilities of the designated personnel?
•Overseeing a training program
•Selecting components
•Monitoring and observing compounding activities and taking immediate corrective action if deficient practices are observed
•Ensuring that standard operating procedures (SOPs) are fully implemented. The designated person(s) must ensure that follow-up is carried out if problems, deviations, or errors are identified
•Establishing, monitoring, and documenting procedures for the handling and storage of CNSPs and/or components of CNSPs
•Designated personnel must develop training to take place every 12 months
What are key concepts to personnel hygiene and garbing per 795?
- Evaluate whether you have personal risk of contaminating the compounding environment (e.g. rash/ open wound/ respiratory infection)
- Remove outer garments + jewelry + earbuds
- Wash hands for minimum of 30 seconds, dry hands, don globes
- Gowns + face masks + shoe covers + head covers only AS NEEDED (not required)
What are key concepts of building/facilities per 795?
How often do work surfaces, floors, walls, ceilings, and storage shelving be cleaned per 795?
- Work surfaces: beginning and end of each shift, after spills, between products of different components
- Floors: Daily
- Walls: When visibly soiled
- Ceilings: When visibly soiled
- Storage shelving: Every 3 months, after spills, after contamination
How are components chosen per USP 795?
- SDS must be readily accessible
- Designated person decides components
FOR APIs...
•Must comply with the criteria in the USP–NF monograph if one exist
•Must have a COA that includes specifications (e.g., compendial requirements for quality) and test results for the component that show the API meets expected quality --> this is only a suggestion for excipients
•In the United States, must be manufactured by an FDA-registered facility --> this is only a suggestion for excipients
•Outside of the United States, must comply with the laws and regulations of the applicable regulatory jurisdiction
What is a master formulation?
Master formulation needs to be created which is essentially an instructional guide for each individual prep and the steps required to make it
What must the master formulation include?
•Name, strength or activity, and dosage form of the CNSP
•Identities and amounts of all components; if applicable, relevant characteristics of components (e.g., particle size, salt form, purity grade, solubility)
•Container closure system(s)
•Complete instructions for preparing the CNSP including equipment, supplies, and description of compounding steps
•Physical description of the final CNSP
•Beyond-use date (BUD) and storage requirements
•Reference source to support the assigned BUD
•If applicable, calculations to determine and verify quantities and/or concentrations of components and strength or activity of the API(s)
•Labeling requirements (e.g., shake well)
•Quality control (QC) procedures (e.g., pH testing, visual inspection) and expected results
•Other information needed to describe the compounding process and ensure repeatability (e.g., adjusting pH, temperature)
What is a compounding record?
A compounding record is required which will display information such as internal identifiers (lot), who made it, when product was made. May be physical copy or built into EHR.
What must the compounding record include?
•Name, strength or activity, and dosage form of the CNSP
•Date—or date and time—of preparation of the CNSP
•Assigned internal identification number (e.g., prescription, order, or lot number)
•A method to identify the individuals involved in the compounding process and individuals verifying the final CNSP
•Name, vendor or manufacturer, lot number, and expiration date of each component
•Weight or measurement of each component
•Total quantity of the CNSP compounded
•Assigned beyond-use date (BUD) and storage requirements
•If applicable, calculations to determine and verify quantities and/or concentrations of components and strength or activity of the API(s)
•Physical description of the final CNSP
•Results of quality control procedures (e.g., pH testing and visual inspection)
•Master formulation reference for the CNSP
What are beyond use date requirements?
- Each label on completed preparation must include an expiration date
- BUD should always be conservative
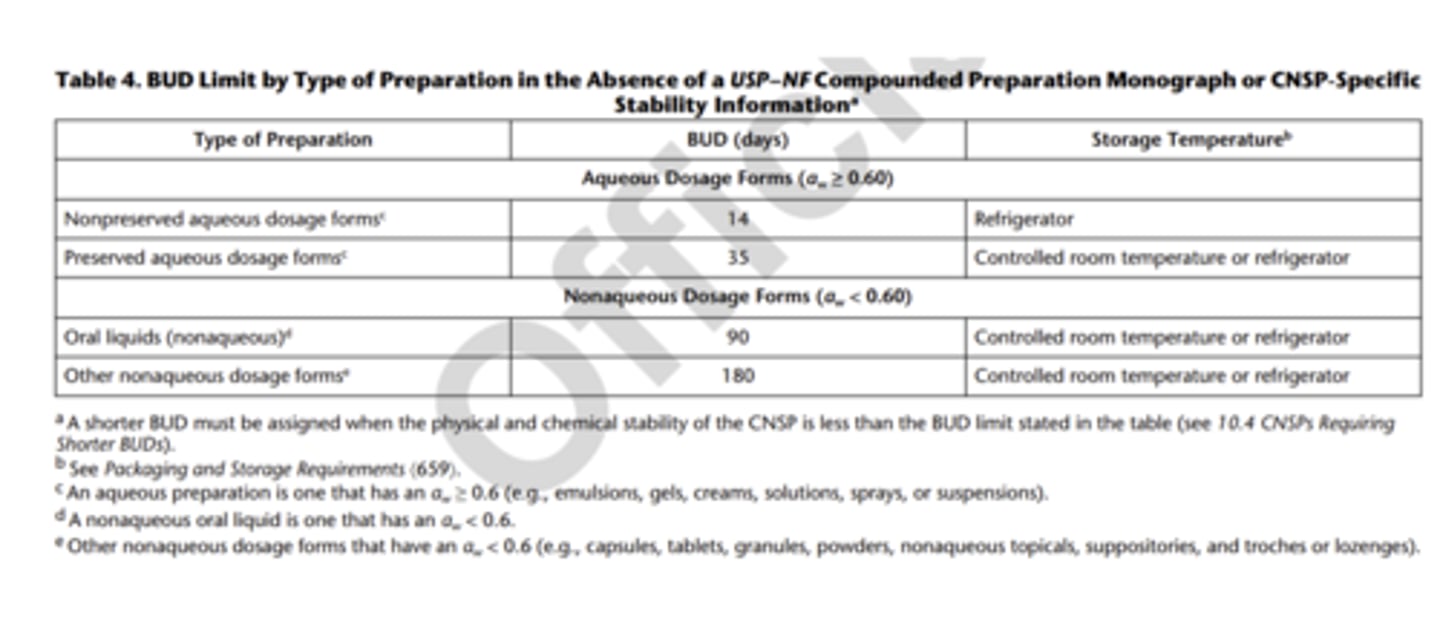
How can we use the water activity table to determine BUD? *EXAM
- Determine what you are making
- Find water activity
- If less than 0.6, you can consider aqueous
- If 0.6 or greater, you can consider aqueous
*you should use this in the absence of USP-NF compounded monograph guidance
What are the BUDs for nonsterile preparations?
What are standard operating procedures?
Determined by each INDIVIDUAL pharmacy NOT USP (but they still need to adhere to 795)
When does quality assurance occur?
BEFORE compounding (e.g. check exp date)
Defined as: System of procedures, activities, and oversight that ensures that the compounding process consistently meets quality standards
When does quality control occur
AFTER compounding
Defined as: Sampling, testing, and documentation of results that, taken together, ensure that specifications have been met before release of the CNSP
QA/QC must cover...
- Adherence to procedures
- Prevention and detection of errors and other quality problems
- Evaluation of complaints and adverse events
- Appropriate investigations and corrective actions
How often does designated personnel need to review QA/QC?
Every 12 months
Does everything need to be documented if it can?
Yas