BIOL/CHEM2300-Midterm
1/28
Earn XP
Description and Tags
Flashcards to review for the midterm. a.a.=amino acid, bc=because, b/tw=between, VDW=van der Waals.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
The peptide bond is between the C of _______ and the N of ______ of two different amino acids.
the carboxyl group (COOH/COO-), the amino group (NH2, amine group)
What is a 1o protein structure?
A sequence of a chain of amino acids; amino acid sequence
What are 3 types of fibrous proteins?
alpha-keratin, fibroin and collagen
alpha-keratin (protein structure, a.a. composition, interactions of structure/structural integrity, uses/where it’s found)
Fibrous protein made of alpha helices, forming a coiled coil of two alpha helices. It is mainly made up of serine, cysteine and glutamine & glutamic acid. It forms this structure due to interactions b/tw leucine along both helices, and disulfide bonds from cysteine b/tw dimers. It is found in hair, fingernails and claws.
What is a dimer?
A dimer is an association of two protein chains.
fibroin (protein structure, a.a. composition, interactions of structure/structural integrity, uses/where it’s found)
Fibrous protein made of stacked beta-sheets. It is rich in glycine, alanine and serine. It is formed by interlocking side chains with neighbouring beta-sheets, and has weak VDW interactions that allow it to be flexible. It is found in spider and silkworm silk, and parachutes.
collagen (protein structure, a.a. composition, interactions of structure/structural integrity, uses/where it’s found)
Fibrous protein made of triple helix; the tropocollagen triple helix. It is rich in glycine, alanine and proline. Proline allows tropocollagen triple helix shape to form, and hydroxyproline strengthens interactions of helices through H-bonding. It is found in skin, tendons and connective tissue.
Tropocollagen Triple Helix
Structure of collagen. Long protein chains made up of three chains to form a triple helix. Each chain has repeats of GXY, where X and Y are usually proline, and proline/hydroxyproline, respectively. G (glycine) is there bc it is the only side chain small enough to fit in the triple helix center. Proline favours extended helices; hydroxyproline helps strengthen interactions b/tw collagen chains.
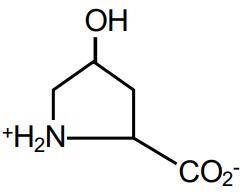
True or False: the amino acid shown is proline
False. It is hydroxyproline.
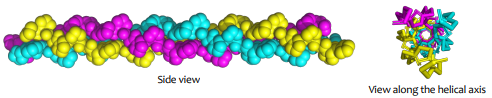
What structure is shown?
A triple helix; tropocollagen triple helix
Why do fibrous proteins have a repeat structure?
Fibrous proteins have repeating structures bc they are only made up of a few a.a. This means that they have repeat sequences of the few residues they use, which makes a repeating structure.
What forms between a carboxyl group and an amino group?
An amide, or amide bond; which is the peptide bond. The reactants (COOH & NH2) become carboxyl (C=O) and NH, forming the amide.
Cryo-electron microscopy (cryo-EM) of proteins uses the sample frozen in vitreous ice. What is vitreous ice?
Vitreous ice is a solid form of water with the structure of liquid water
Select all of the correct statements regarding the hydrophobic effect.
The hydrophobic effect maximizes the number of hydrogen bonds formed between water molecules, the hydrophobic effect is the reason that water-soluble proteins have polar groups on their surface and nonpolar groups within their interiors, the hydrophobic effect minimizes the number of water molecules in clathrates
What advantage do membrane proteins have over soluble proteins with respect to cryo-electron microscopy?
Membrane proteins can tolerate vacuum conditions better than soluble proteins, as the membrane will not evaporate
Electron microscopes have been around for nearly a century, but have only reached the point where they are useful for solving protein structures within the last decade or so when certain problems had been overcome. Below is one problem; what was the solution?
Problem: ice crystals in the sample, which diffract the electron beam
Solution: rapid freezing of the sample
Electron microscopes have been around for nearly a century, but have only reached the point where they are useful for solving protein structures within the last decade or so when certain problems had been overcome. Below is one problem; what was the solution?
Problem: different orientations of protein molecules with the electron beam
Solution: pooling images of similar orientation
Electron microscopes have been around for nearly a century, but have only reached the point where they are useful for solving protein structures within the last decade or so when certain problems had been overcome. Below is one problem; what was the solution?
Problem: fuzzy images owing to weak interactions of the electron beam with single protein molecules
Solution: signal averaging of images
Which one of the following non-covalent interactions will always favor folding of water-soluble protein?
Hydrogen bonding, Ion-dipole interactions, The hydrophobic effect, Van der Waals interaction
The hydrophobic effect (HPE)
True/False: The types non-covalent interactions that give a protein its tertiary structure are the same as those that are involved in giving a protein quaternary structure
True
Quaternary structure
Association of two or more protein chains
Tertiary structure
Interactions involving side chains
secondary structure
backbone hydrogen bonding
True/False: The types of covalent and noncovalent interactions that are observed in proteins with quaternary structure are the same as those observed in the secondary and tertiary structures of proteins
True
A beta turn consists of four residues. A hydrogen bond occurs between residues _____ and four, while residue _____ is usually a small residue, which avoids steric clash.
one, three
True/False: Under physiological conditions the side chain of arginine will always be protonated.
True
Which type of covalent interaction involving amino acid side chains is known to occur in some proteins?
A covalent bond between the sulfur atoms of pairs of cysteine side chains
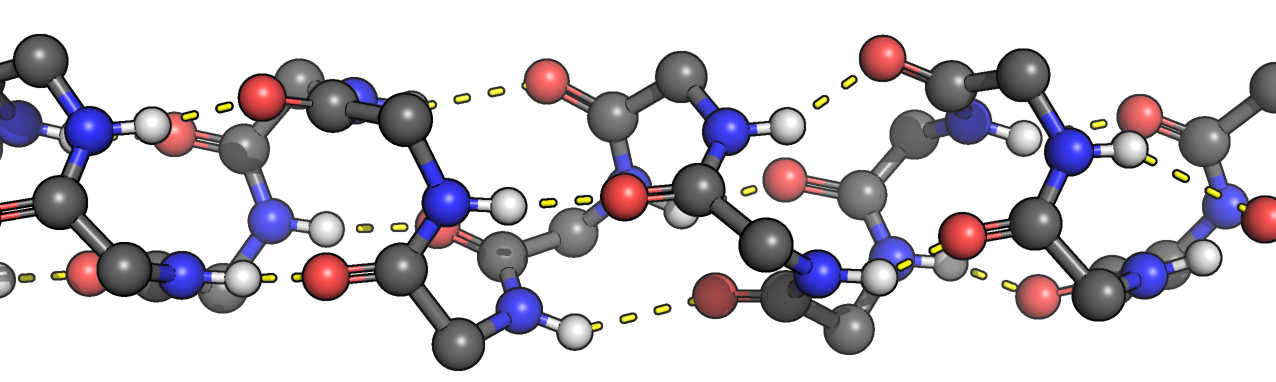
What type of secondary structure is shown below? Side chains have been omitted for clarity. Hydrogen bonds are in yellow.
alpha helix